STERILIZATION AND DISINFECTION Ms Dunishiya de Silva DISINFECTION









































- Slides: 43

STERILIZATION AND DISINFECTION Ms. Dunishiya de Silva

DISINFECTION • Disinfection is the elimination of pathogens, except spores, from inanimate objects • Disinfectants are chemical solutions used to clean inanimate objects (physical processes, e. g. , UV radiation, may also be employed to effect disinfection)

• Germicides are chemicals that can be applied to both animate (living) and inanimate objects for the purpose of eliminating pathogens • Antiseptics are formulated for application to living tissue

Qualities of a disinfectant • • Broadly active (killing pathogens) Not poisonous (not harmful) Penetrating (to pathogens) Not damaging to non-living materials Stable Easy to work with Not unpleasant

Factors determine the disinfectant performance • Disinfectant concentrations • Length (time) of administration • Temperature during administration (usual chemical reaction 2 x increase in rate with each 10°C increase in temperature) • Microbe type (e. g. , mycobacteria, spores, and certain viruses can be very resistant to disinfection—in general vegetative cells in log phase are easiest to kill)

• Substrate effects (e. g. , high organic content interferes with disinfection) • It is easier (and faster) to kill fewer microbes than many microbes

Cleansing is the removal of soil or organic material from instruments and equipment & may be done, clinically, in four steps: • Rinsing the object under cold water • Applying detergent and scrubbing object • Rinsing the object under warm water • Drying the object prior to sterilization or disinfection

Sterilization is the total elimination of all microorganisms including spores • Typically the last things to die are the highly heat and chemical-resistant bacterial endospores • Instruments used for invasive procedures must be sterilized prior to use

• Moist heat or steam, radiation, chemicals (e. g. , glutaraldehyde), and ethylene oxide (a gas) are employed for sterilization • Sterilization by autoclaving, which uses moist heat, is used in most hospital and microbiology laboratory settings

Other terms related to disinfection • Sanitization: Lowering of microbial counts to prevent transmission in public setting (e. g. , restaurants & public rest rooms) • Degerming: Mechanical removal of microbes, e. g. , from hands with washing • Sepsis: Bacterial contamination • Antisepsis: Reduction of or inhibition of microbes found on living tissue

• Germincides, Fungicides, Virucides • Physical versus Chemical disinfectants • Static (stasis) versus Cidal (e. g. , bacteriostatic versus bacteriocidal)

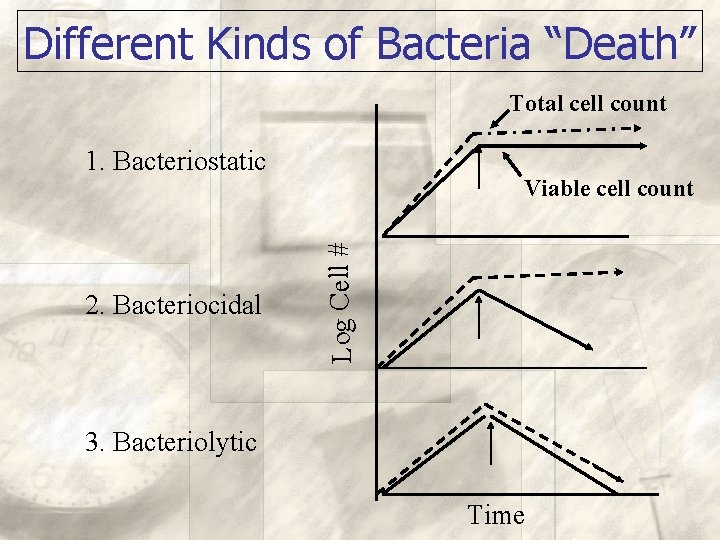
Different Kinds of Bacteria “Death” Total cell count 1. Bacteriostatic Log Cell # 2. Bacteriocidal Viable cell count 3. Bacteriolytic Time

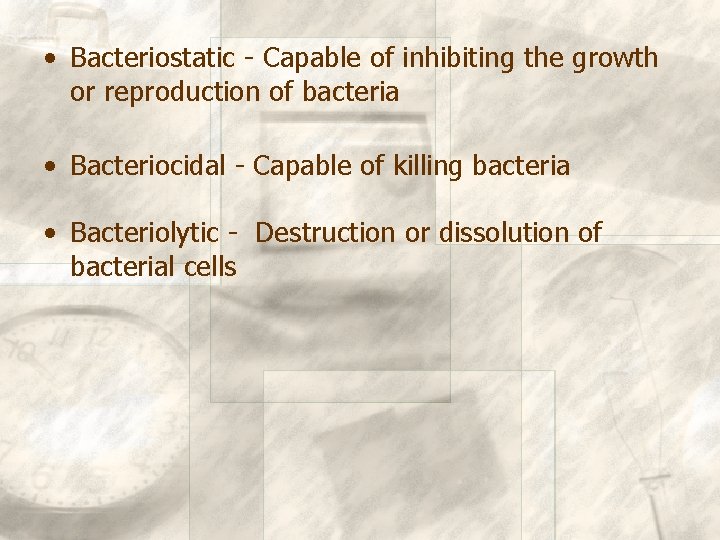
• Bacteriostatic - Capable of inhibiting the growth or reproduction of bacteria • Bacteriocidal - Capable of killing bacteria • Bacteriolytic - Destruction or dissolution of bacterial cells

Methods of Disinfection and Sterilization • Heat: Kills microorganisms by denaturing their enzymes and other proteins. Heat resistance varies widely among microbes.

MOIST HEAT • Moist Heat: Kills microorganisms by coagulating their proteins.

• Boiling: Heat to 100 o. C or more. Kills vegetative forms of bacterial pathogens, almost all viruses, and fungi and their spores within 10 minutes or less. Endospores and some viruses are not destroyed this quickly. However brief boiling will kill most pathogens. • Hepatitis virus: Can survive up to 30 minutes of boiling. • Endospores: Can survive up to 20 hours or more of boiling

• Autoclave: Chamber which is filled with hot steam under pressure. Preferred method of sterilization, unless material is damaged by heat, moisture, or high pressure. • Temperature of steam reaches 121 o. C at twice atmospheric pressure. • Most effective when organisms contact steam directly or are contained in a small volume of liquid. • All organisms and endospores are killed within 15 minutes.

• Pasteurization: Developed by Louis Pasteur to prevent the spoilage of beverages. Used to reduce microbes responsible for spoilage of beer, milk, wine, juices, etc.

Dry Heat • Dry Heat: Kills by oxidation effect • Direct Flaming: Used to sterilize inoculating loops and needles. Heat metal until it has a red glow. • Incineration: Effective way to sterilize disposable items (paper cups, dressings) and biological waste.

• Hot Air Sterilization: Place objects in an oven. Require 2 hours at 170 o. C for sterilization. Dry heat is transfers heat less effectively to a cool body, than moist heat.

Filtration • Filtration: Removal of microbes by passage of a liquid or gas through a screen like material with small pores. Used to sterilize heat sensitive materials like vaccines, enzymes, antibiotics, and some culture media.

• High Efficiency Particulate Air Filters (HEPA): Used in operating rooms and burn units to remove bacteria from air. • Membrane Filters: Uniform pore size. Used in industry and research. Different sizes: • 0. 22µm and 0. 45µm Pores: Used to filter most bacteria. Don’t retain spirochetes, mycoplasmas and viruses. • 0. 01µm Pores: Retain all viruses and some large proteins.

Low Temperature • Low Temperature: Effect depends on microbe and treatment applied • Refrigeration: Temperatures from 0 to 7 o. C. Bacteriostatic effect. Reduces metabolic rate of most microbes so they cannot reproduce or produce toxins

• Freezing: Temperatures below 0 o. C. • Flash Freezing: Does not kill most microbes. • Slow Freezing: More harmful because ice crystals disrupt cell structure. • Over a third of vegetative bacteria may survive 1 year. • Most parasites are killed by a few days of freezing.

Desiccation • Desiccation: In the absence of water, microbes cannot grow or reproduce, but some may remain viable for years. After water becomes available, they start growing again. Susceptibility to desiccation varies widely: u. Neisseria gonnorrhea: Only survives about one hour. u. Mycobacterium tuberculosis: May survive several months. u. Viruses are fairly resistant to desiccation. u. Clostridium spp. and Bacillus spp. : May survive decades.

Osmotic Pressure • Osmotic Pressure: The use of high concentrations of salts and sugars in foods is used to increase the osmotic pressure and create a hypertonic environment.

• Plasmolysis: As water leaves the cell, plasma membrane shrinks away from cell wall. Cell may not die, but usually stops growing. • Yeasts and molds: More resistant to high osmotic pressures. • Staphylococci spp. that live on skin are fairly resistant to high osmotic pressure

Radiation: Three types of radiation kill microbes: 1. Ionizing Radiation: Gamma rays, X rays, electron beams, or higher energy rays. Dislodge electrons from atoms and form ions. Cause mutations in DNA

• Used to sterilize pharmaceuticals and disposable medical supplies. Food industry is interested in using ionizing radiation. Disadvantages: Penetrates human tissues. May cause genetic mutations in humans.

2. Ultraviolet light (Nonionizing Radiation): Wavelength is longer than 1 nanometer. Damages DNA Used to disinfect operating rooms, nurseries, cafeterias. Disadvantages: Damages skin, eyes. Doesn’t penetrate paper, glass, and cloth.

3. Microwave Radiation: Wavelength ranges from 1 millimeter to 1 meter. Heat is absorbed by water molecules. May kill vegetative cells in moist foods. Bacterial endospores, which do not contain water, are not damaged by microwave radiation.

Types of Disinfectants

1. Phenols and Phenolics: u Phenol (carbolic acid) was first used by Lister as a disinfectant. u Rarely used today because it is a skin irritant and has strong odor. u Acts as local anesthetic.

u Phenolics are chemical derivatives of phenol u Cresols: Derived from coal tar (Lysol). u Biphenols (p. Hiso. Hex): Effective against grampositive staphylococci and streptococci. Used in nurseries. Excessive use in infants may cause neurological damage. Destroy plasma membranes and denature proteins. u Advantages: Stable, persist for long times after applied, and remain active in the presence of organic compounds. u

2. Halogens: Effective alone or in compounds. A. Iodine: u Tincture of iodine (alcohol solution) was one of first antiseptics used. u Combines with amino acid tyrosine in proteins and denatures proteins. u Stains skin and clothes, somewhat irritating. u Iodophors: Compounds with iodine that are slow releasing, take several minutes to act. Used as skin antiseptic in surgery. Not effective against bacterial endospores. u u Betadine Isodine

B. Chlorine: u When mixed in water forms hypochlorous acid: Cl 2 + H 2 O ------>H+ + Cl+ HOCl Used to disinfect drinking water, pools, and sewage. u Chlorine is easily inactivated by organic materials. u Sodium hypochlorite (Na. OCl): Is active ingredient of bleach. u Chloramines: Consist of chlorine and ammonia. Less effective as germicides.

3. Alcohols: u Kill bacteria, fungi, but not endospores or naked viruses. u Act by denaturing proteins and disrupting cell membranes. u Evaporate, leaving no residue. u Used to mechanically wipe microbes off skin before injections or blood drawing.

u Not good for open wounds, because proteins to coagulate. Ethanol: Drinking alcohol. Optimum concentration is 70%. u Isopropanol: Rubbing alcohol. Better disinfectant than ethanol. Also cheaper and less volatile. u

4. Heavy Metals: u Include copper, selenium, mercury, silver, and zinc. A. Silver: u 1% silver nitrate used to protect infants against gonorrheal eye infections until recently. B. Mercury u Organic mercury compounds like merthiolate and mercurochrome are used to disinfect skin wounds. C. Copper u Copper sulfate is used to kill algae in pools and fish tanks.

D. Selenium u Kills fungi and their spores. Used for fungal infections. u Also used in dandruff shampoos. E. Zinc u Zinc chloride is used in mouthwashes. u Zinc oxide is used as antifungal agent in paints.

5. Quaternary Ammonium Compounds (Quats): Widely used surface active agents. u Cationic (positively charge) detergents. u Effective against gram positive bacteria, less effective against gram-negative bacteria. u Also destroy fungi, amoebas, and enveloped viruses. u

6. Aldehydes: u Include some of the most effective antimicrobials. u Inactivate proteins by forming covalent crosslinks with several functional groups.

A. Formaldehyde gas: u Excellent disinfectant. u Commonly used as formalin, a 37% aqueous solution. u Formalin was used extensively to preserve biological specimens and inactivate viruses and bacteria in vaccines. u Irritates mucous membranes, strong odor. u Also used in mortuaries for embalming.