Stereoisomerism Isomerism The term isomerism refers to compounds














- Slides: 28

Stereoisomerism

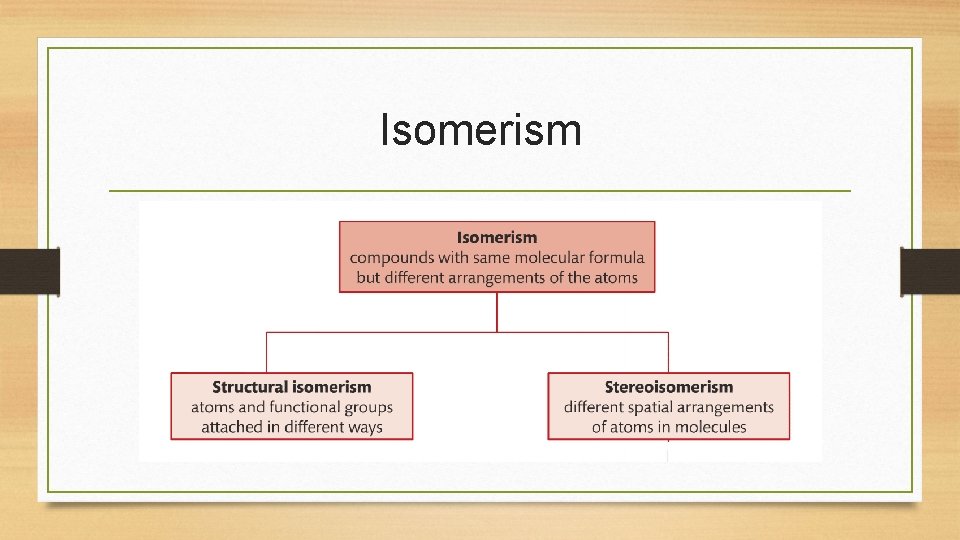
Isomerism • The term isomerism refers to compounds that have the same molecular formula but different arrangement of atoms. • Structural isomerism- isomers in which atoms and functional groups are attached in different ways. • Stereoisomerism- isomers in which the atoms are attached in the same order, but different in their spatial or three dimensional arrangement.

Isomerism

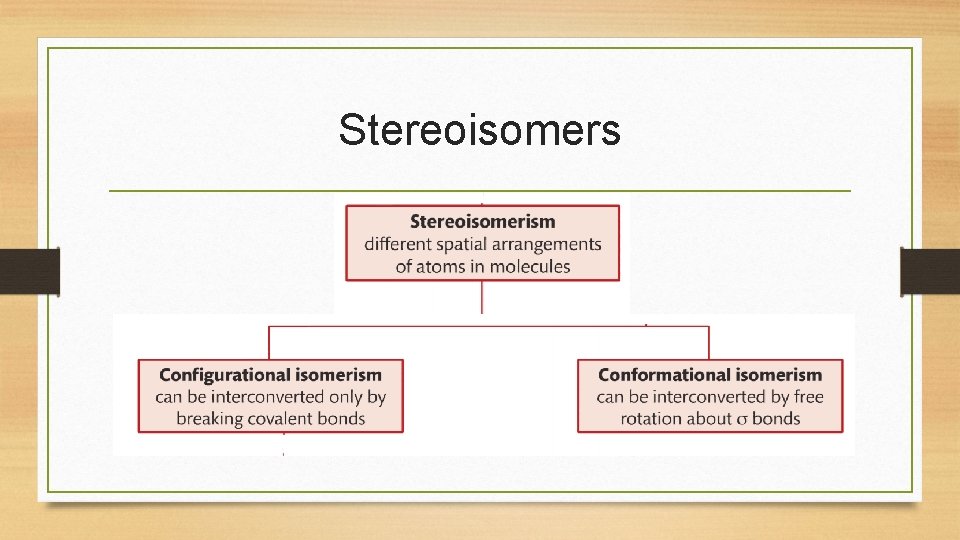
Stereoisomers • Stereoisomers can be classified as: 1. Conformational isomers- can be interconverted by free rotation about single (sigma) bonds. 2. Configurational isomers- can’t be interconverted, so they exist as separate compounds with distinct properties

Stereoisomers

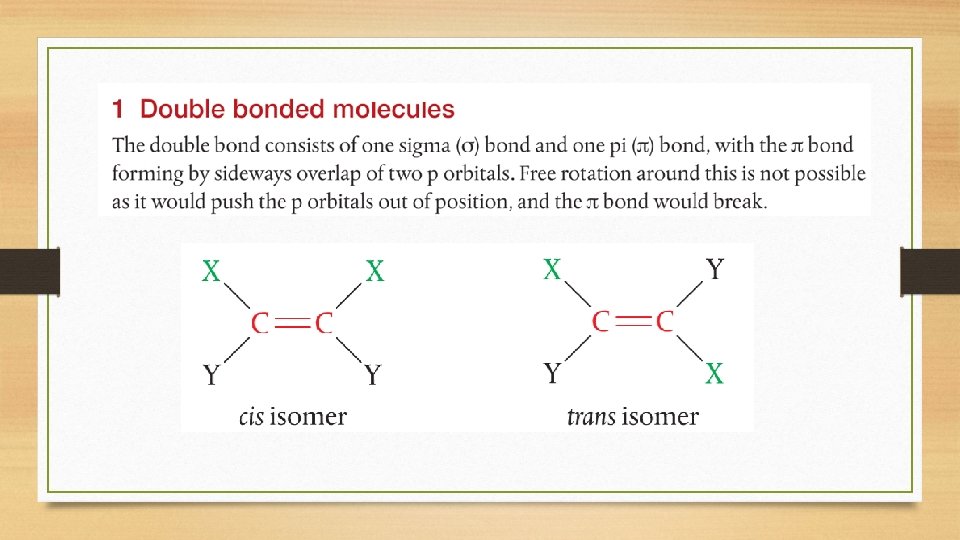
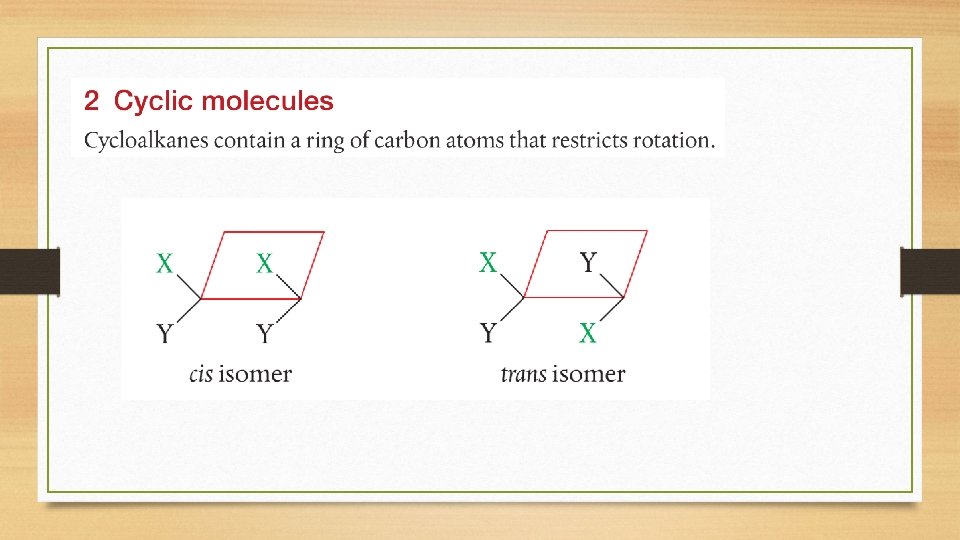
Configurational Isomers • Two main groups of configurational isomers are:

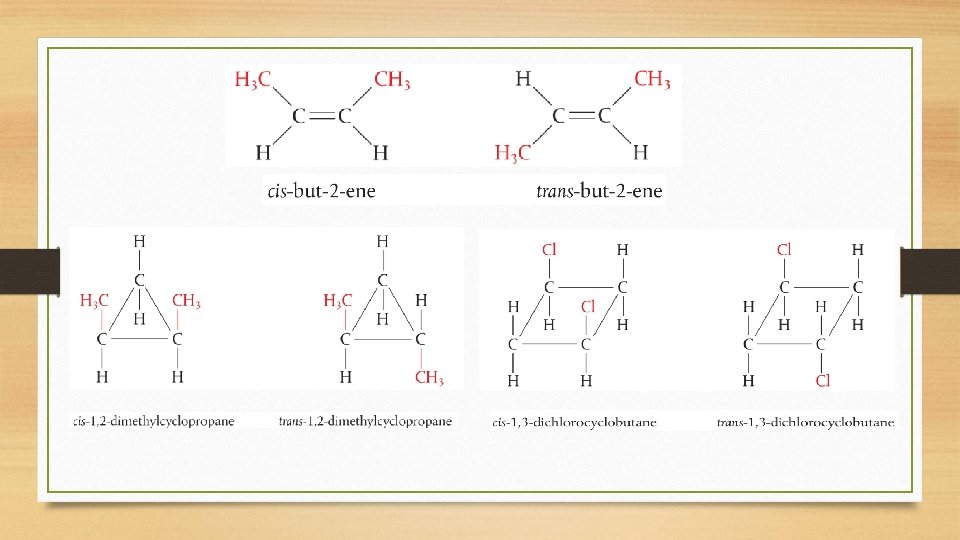
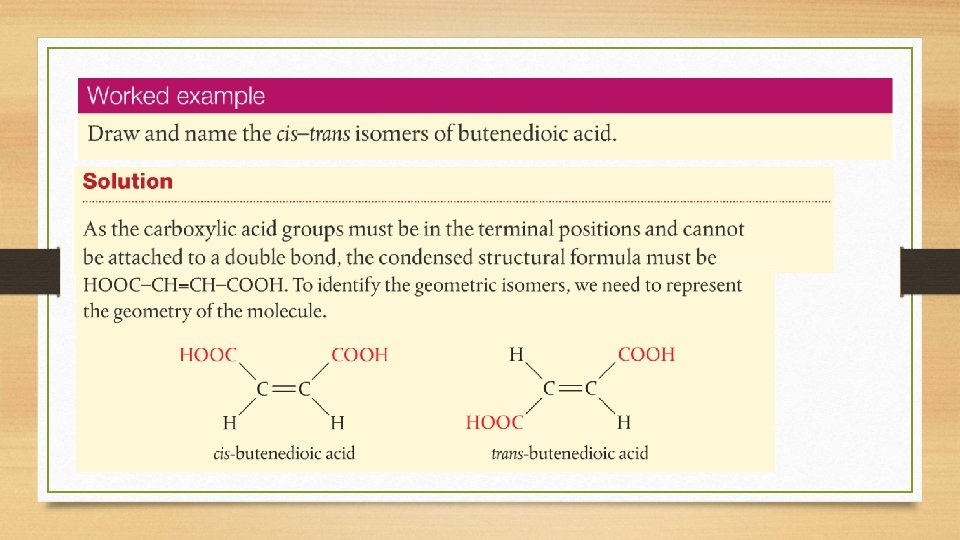
Cis-Trans and E/Z Isomers






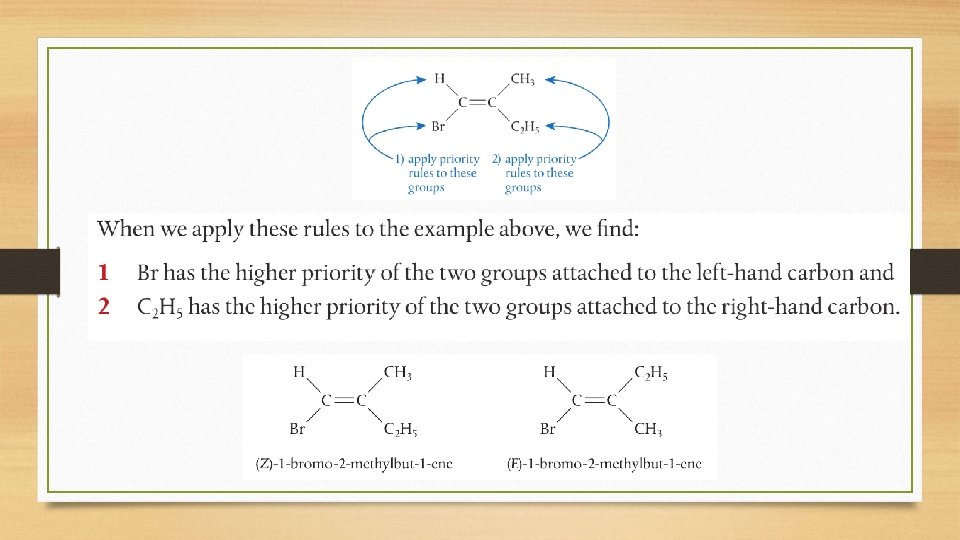
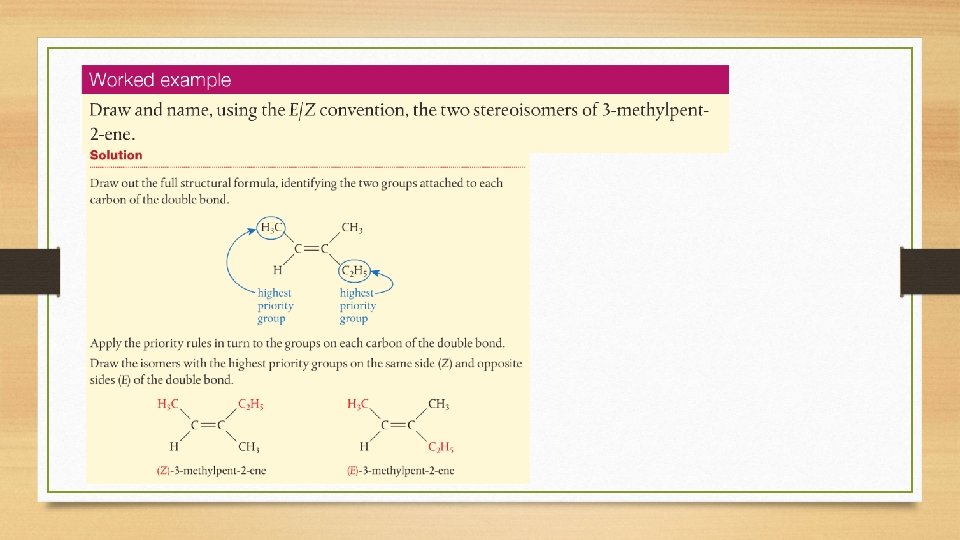
E/Z Isomers • There are cases where E/Z is used instead of cis-trans. • This happens when the carbon atoms in the double bond are bonded to more than two different groups. For example:

E/Z Isomers E/Z naming is based on the rules of priority.



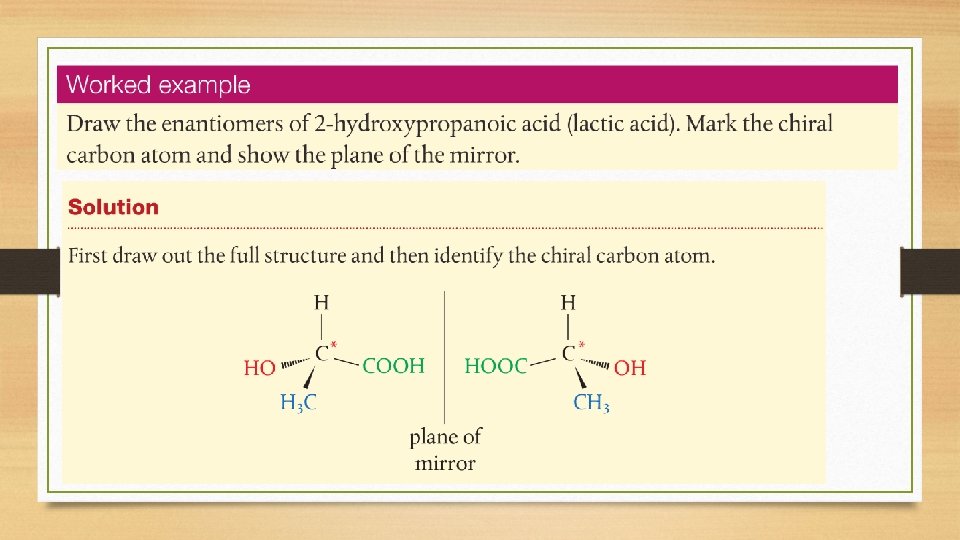
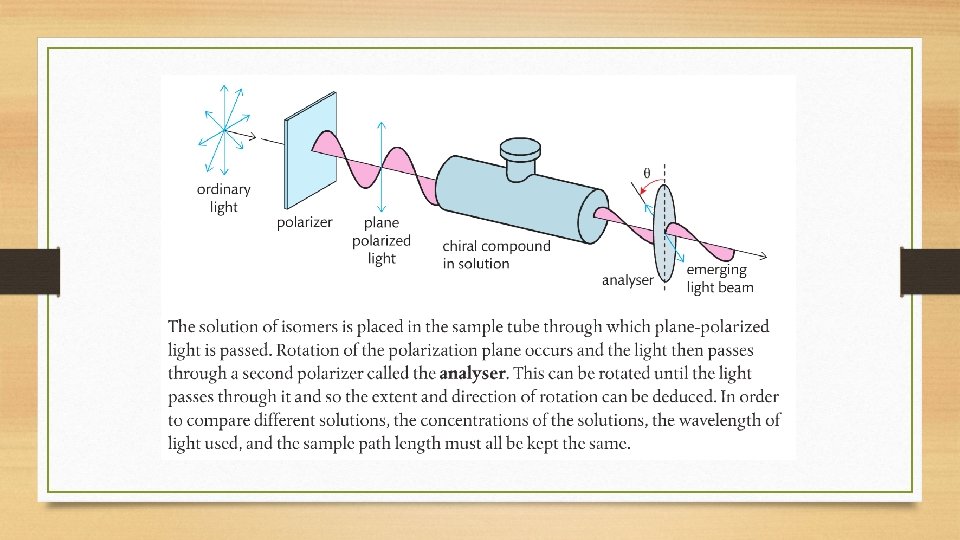
Optical Isomers • Isomers that have different spatial arrangements as a result of there being four different groups tetrahedrally bonded to a single carbon atom, known as a chiral carbon. • These isomers are non-superimposable mirror images of each other and are known as enantiomers. • Optical isomers have the ability to rotate plane-polarized light.

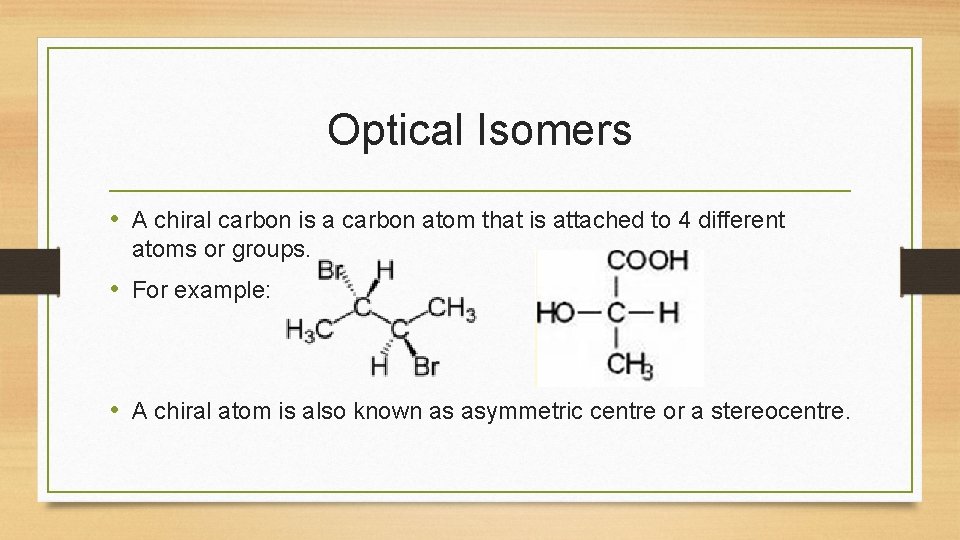
Optical Isomers • A chiral carbon is a carbon atom that is attached to 4 different atoms or groups. • For example: • A chiral atom is also known as asymmetric centre or a stereocentre.

Optical Isomerism

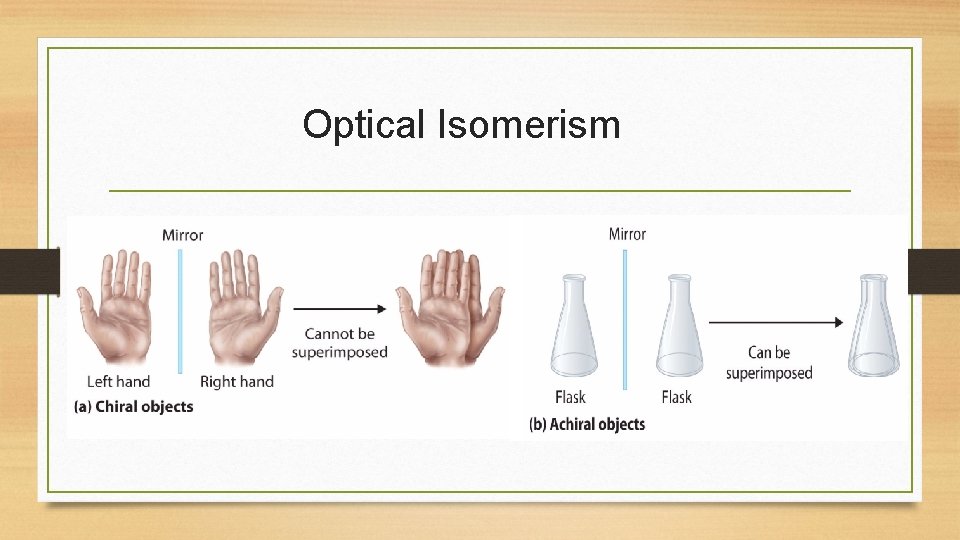
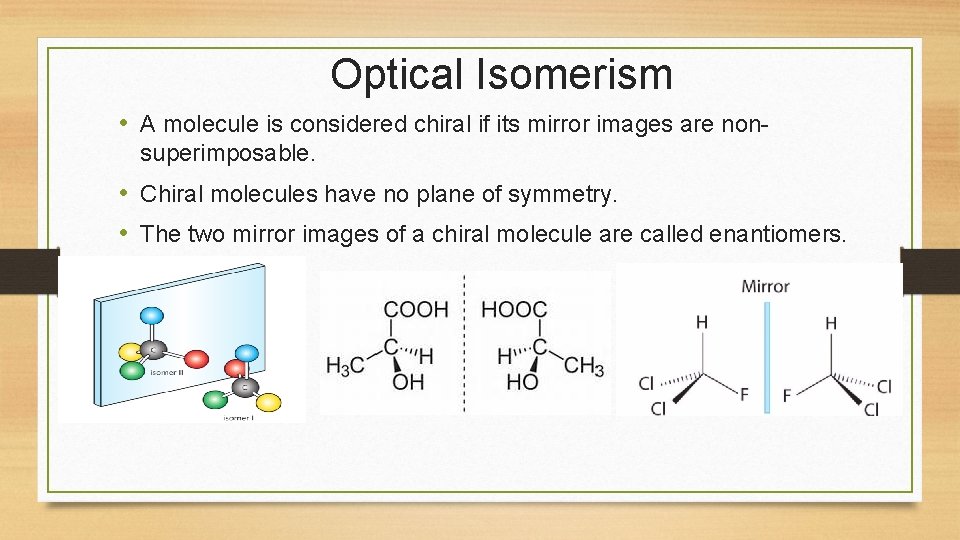
Optical Isomerism • A molecule is considered chiral if its mirror images are nonsuperimposable. • Chiral molecules have no plane of symmetry. • The two mirror images of a chiral molecule are called enantiomers.

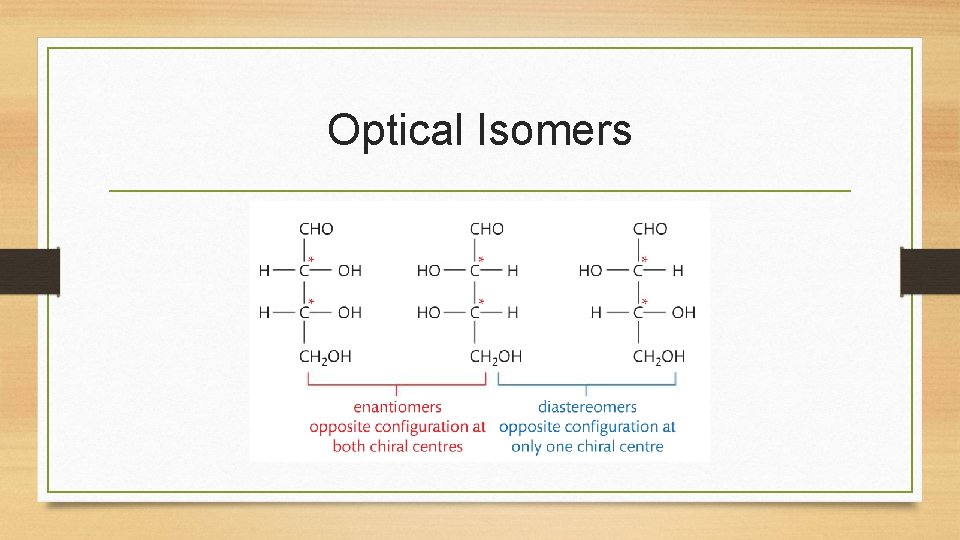
Optical Isomerism • A mixture containing equal amounts of the two enantiomers is known as a racemic mixture. • Many molecules have more than once chiral carbon, and so can give rise to different configurations at each chiral carbon. Enantiomers have opposite configurations at EACH chiral centre, which is why molecules are mirror images of each other. • Molecules that have opposite configurations at some, but not all, chiral carbons are known as diastereomers and are not mirror images of each other. • A single chiral centre in a molecule gives rise to two stereoisomers. In general, a molecule with n number of chiral centres has a maximum of 2 n stereoisomers.

Optical Isomers


Properties of Optical Isomers

Optical Activity



Reactivity With Other Chiral Molecules