Stem cells Ahmad Sh Silmi Msc FIBMS Terminology

Stem cells Ahmad Sh. Silmi Msc, FIBMS

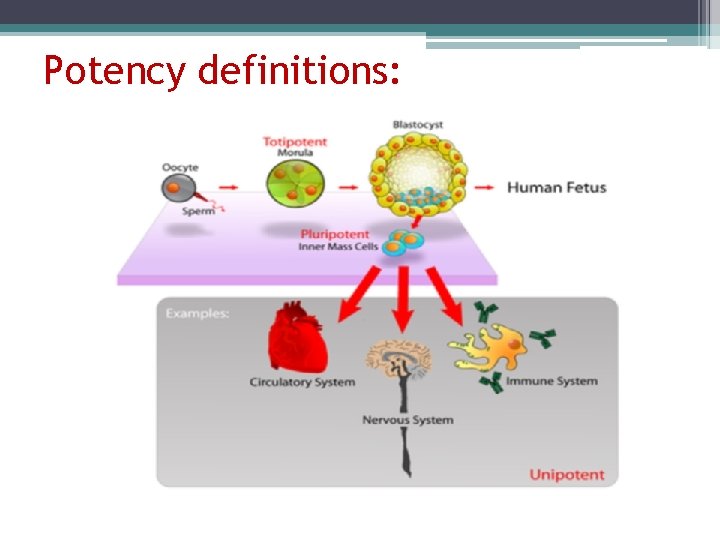
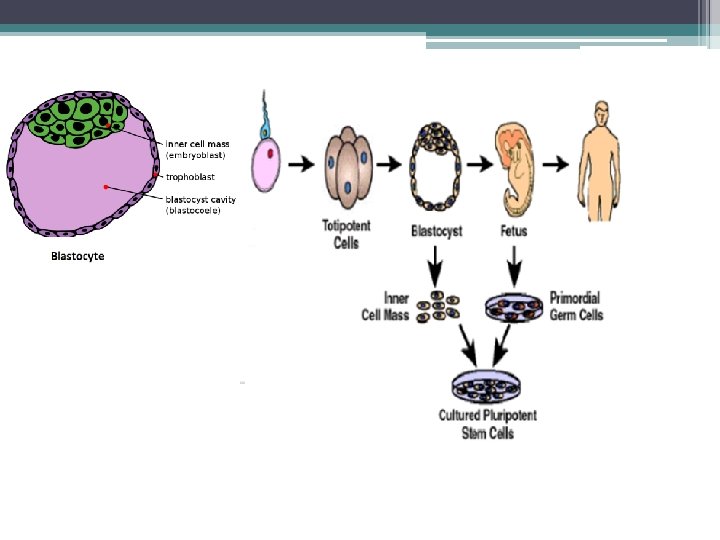
Terminology: q Totipotent cells: These cells have the potential to become any type in the adult body; any cell of the extraembryonic membranes (e. g. , placenta). The only totipotent cells are the fertilized egg and the first 4 or so cells produced by its cleavage (as shown by the ability of mammals to produce identical twins, triplets, etc. ).

q Pluripotent stem cells: These are true stem cells, with the potential to make any differentiated cell in the body (but probably not those of the placenta which is derived from the trophoblast).
Potency definitions:

Three types of pluripotent stem cells have been found: q Embryonic Stem (ES) Cells. These can be isolated from the inner cell mass (ICM) of the blastocyst — the stage of embryonic development when implantation occurs. For humans, excess embryos produced during in vitro fertilization (IVF) procedures are used. q Embryonic Germ (EG) Cells. These can be isolated from the precursor to the gonads in aborted fetuses. q Embryonic Carcinoma (EC) Cells. These can be isolated from teratocarcinomas, a tumor that occasionally occurs in a gonad of a fetus. Unlike the other two, they are usually aneuploid

All three of these types of pluripotent stem cells q can only be isolated from embryonic or fetal tissue; q can be grown in culture, but only with special methods to prevent them from differentiating.

Multipotent stem cells: q These are true stem cells but can only differentiate into a limited number of types. § For example, the bone marrow contains multipotent stem cells that give rise to all the cells of the blood but not to other types of cells. q Multipotent stem cells are found in adult animals; perhaps most organs in the body (e. g. , brain, liver) contain them where they can replace dead or damaged cells. q These adult stem cells may also be the cells that — when one accumulates sufficient mutations — produce a clone of cancer cells.

q Oligopotent stem cells can differentiate into only a few cells, such as lymphoid or myeloid stem cells q Unipotent cells can produce only one cell type, their own but have the property of self-renewal which distinguishes them from non-stem cells (e. g. muscle stem cells).

What are stem cells? Sources. Culture. Haematopoietic stem cell transplantation. Problems with stem cell transplantation and their possible solutions. q Future trends. q Key research events. q q q

What are stem cells? : q Stem cells are a special kind of cell that have the ability to divide indefinitely and have the potential to give rise to specialized cells (that is, any cell of the body). q Stating it in scientific terminology, the defining properties of stem cells are § They are not terminally differentiated but determined. § They can divide indefinitely. § When they divide, each daughter cell has a choice: it can either remain a stem cell or take on a course leading to terminal differentiation. The job of stem cell is not to carryout differentiation function, but rather to produce cells that will differentiate.

q Stem cells are cells that divide by mitosis to form either § two stem cells, thus increasing the size of the stem cell " pool", § or �one daughter that goes on to differentiate, �and one daughter that retains its stem-cell properties. q How the choice is made is still unknown. However, several genes have been found whose activity prevents a daughter cell from differentiating.

q Stem cells have a non-descript appearance, making them hard to identify. But this is not to say that stem cells are alike. Although not terminally differentiated, they are nevertheless determined.

Unique properties of all stem cells: 1. Stem cells are unspecialized One of the fundamental properties of stem cells is that it does not have any tissue specific structures that allow it to perform specialized function. However under certain physiological and experimental conditions unspecialized cell can give rise to specialized cells such as including heart muscle cells, blood cells or nerve cells required to repair damaged or depleted adult cell population or tissue.

Contd. 2. Proliferation They are capable of dividing and renewing themselves for indefinite periods 3. Differentiation They can give rise to specialized tissue 4. Plasticity 5. Stem cell from one tissue may be able to give rise to cell types of completely different tissue , a phenomenon known as plasticity. e. g. Blood cells becoming neuron, liver cells producing insulin and haematopoietic stem cells, developing into heart muscle.
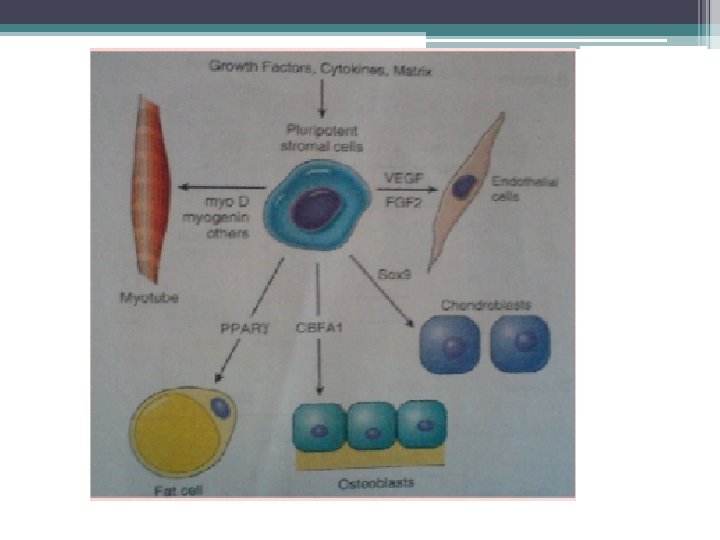

Sources and characteristics q Human embryonic cells : They are derived from the inner cell mass of blastocyts. The isolation and partial characterization first reported in Nov-1998. q ES cells combine three properties not found in other cell lines ▫ They replicate indefinitely ▫ They are genetically normal ▫ They can also differentiate into many cell types in tissue culture.

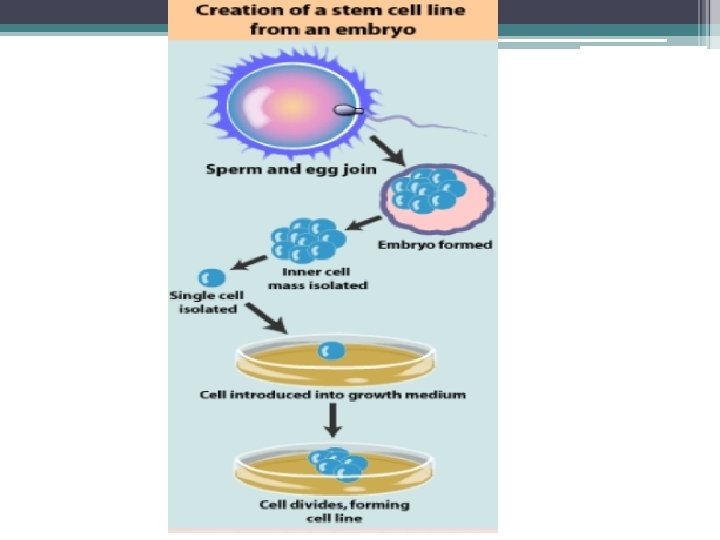
q These are derived from embryos that develop from the eggs that have been fertilized in vitro and donated for research purposes with informed consent of the donors. q They are not derived from eggs fertilized in a woman’s body. The embryos are typically four or five days old and are hollow microscopic ball of cells called the blastocyts.

q Human embryonic germ cells: These cells are collected from fetal tissue at a somewhat later stage of development {from a region called gonadal ridge} and the cell type in which they can develop into may be slightly limited. {First in 1998} q These are capable of forming the three germ layers that make all specific organs of the body.

Adult stem cells q Contrary to the ES cells, which are pluripotent, adult stem cells have a more restricted differentiation capacity and are usually lineage specific. q Present in bone marrow and other tissues. q Stem cells outside bone marrow are called as tissue stem cells. q In the tissue, stem cells are located in the sites called niches.
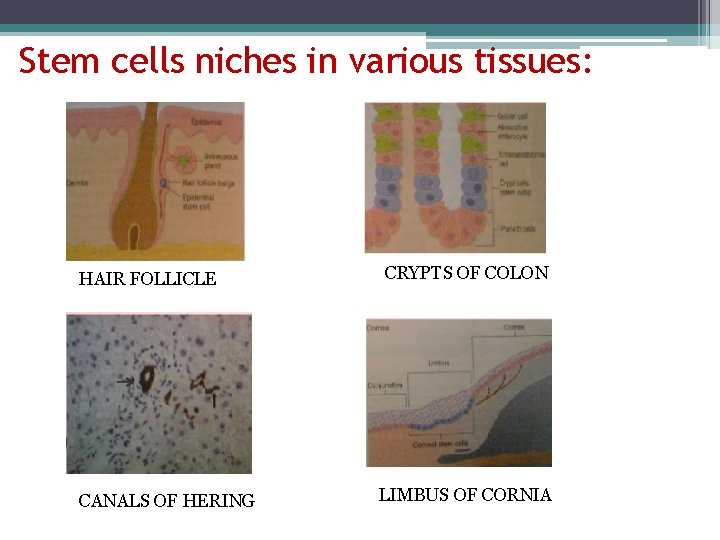
Stem cells niches in various tissues: HAIR FOLLICLE CANALS OF HERING CRYPTS OF COLON LIMBUS OF CORNIA

How are embryonic cells grown in the laboratory? q Growing cells in the laboratory is called as cell culture. The inner surface of the culture dish is coated with mouse embryonic skin cells , called as feeder layer. § It gives the inner cell mass a sticky surface to which they can attach also the feeder cell releases nutrients. q Over the course of several days the inner cell mass proliferate and begin to crowd the culture dish. q They are then gently removed and plated into several fresh culture dishes. § The process of plating the cells is repeated several times and for many months and is called subculturing. § Each cycle of subculturing is referred to as a passage. q After six months or more the inner cell mass yield millions of embryonic stem cells. These cells are pluripotent and appear genetically normal and are referred to as an embryonic stem cell line.
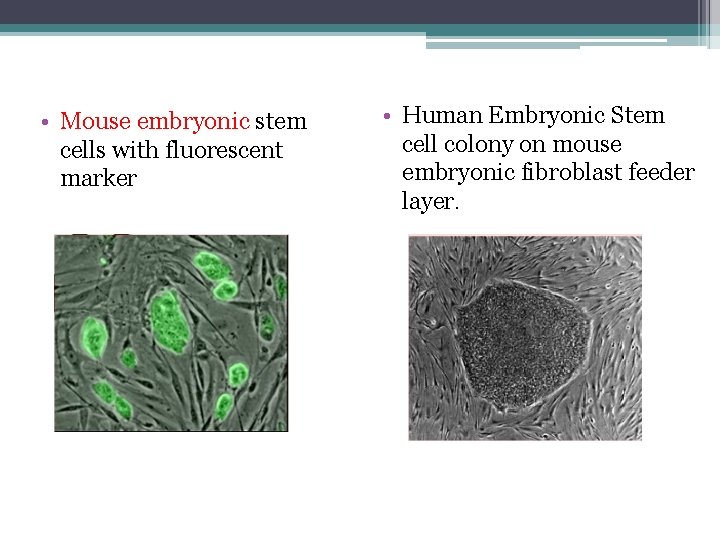
• Mouse embryonic stem cells with fluorescent marker • Human Embryonic Stem cell colony on mouse embryonic fibroblast feeder layer.

q An adult stem cell also called as somatic stem cells are an undifferentiated cells found among differentiated cells in a tissue or an organ which can renew itself and can differentiate to yield the major specialized cell type of the tissue or the organ. q Primary role of adult stem cell is to maintain and repair the tissue in which they are found. q Their origin is unknown § Haematopoietic stem cells – form all type of blood cells § Stromal cells – can generate cartilage, fat, and fibrous connective tissue. § Brain stem cells – astrocytes , oligodendrocytes and neurons
Adult stem cell plasticity and transdifferentiation q The ability to differentiate into multiple cell type is called as plasticity or transdifferentiation. e. g. q Haematopoietic stem cells differentiating into brain cells, skeletal muscle cells, cardiac muscle cells and liver cells q Brain cells may differentiate into blood cells and skeletal muscle cells.

Haematopoietic stem cell transplantation Introduction: q Original term- Bone marrow transplantation q It involves collection and transplantation of haematopoietic stem cells but Peripheral blood and umbilical cord blood are also useful source of stem cells so Haematopoietic stem cell transplantation has become the preferred generic term. q It was pioneered at Fred Hutchinson Cancer Research Centre from the 1950 s through 1970 s by E. Donnall Thomas {Nobel prize in physiology and medicine} q In 1968, first major landmark : allogenic transplant for an infant with X-linked lymphopenic immune deficiency and for another with Wiskott Aldrich syndrome. q In 1988 cord blood collected stem cell transplantation for a young boy with Fanconi anaemia.

Haematopoietic stem cells q It is defined as the cell with the ability to achieve long term reconstruction of both myeloid and lymphoid lineage. q Features : - 1. Remarkable regenerative capacity 2. Ability to home to marrow spaces following IV injection. {mediated in par by interaction of selectin on bone marrow endothelium and integrin on early hematopoietic cells } 3. Ability to be cryopreserved.

HLA typing q It is essential for patients receiving allogenic transplant. It is performed on blood samples obtained from patient and potential donors. The probability of finding an HLA match in the family {from sibling} is about 25 -35%. q Currently most centers perform class I {HLA –A, B, C} typing. This is followed by molecular typing for class II antigen {HLA –DR, DQ and DP}. q By molecular typing C locus appears to be a significant determinant of the graft failure and graft versus host disease {GVHD}. q When more than one donor appear suitable , the limiting dilution assay of alloreactive cytotoxic T-lymphocytes precursors can aid donor selection. The donor choice involves donors ABO type, donors age, transfusion history and (for female) donors parity.

HLA testing q In HLA antigen compatibility test 6 -of-6 match refers to testing of HLA-A, HLA-B and HLA-DR, each of which has 2 alleles. q If donor and recipient are not 6 -of-6 match, they are termed as mismatched and when only 3 of 6 mismatch, the term haploidentical donor applies. q These grafts must be manipulated in vitro to reduce the number of immunocompetent T cells to lessen the likelihood and severity of GVHD. q One advantage of using Haploidentical donor is that multiple donor are available within a family. q Another advantage is equal availability of donor for all ethnic and racial groups. q In haploidentical transplantation mismatching of maternal antigen is better tolerated.

Donor selection q The donor must be in good health without other co morbid conditions. The donor must have a performance status that permit safe collection of cells , be able to tolerate anesthesia and have adequate cardiac , pulmonary, hepatic and renal function. q Paediatric donor are used only for autologous collection or donation to siblings. Donors with ongoing or history of a malignancy other than minor skin cancer {e. g. basal cell carcinoma } are generally excluded. q The following studies are performed on hematopoietic cell donors: § § § § § History and physical examination CBC count and platelet count Serum creatinine, electrolyte and liver function studies Serological studies for cytomegalo virus, herpes virus, HIV RNA, Anti -HIV antibodies and Hepatitis B and C viruses ABO blood typing Chest radiography Electrocardiography Stem cell sources: - Bone marrow {BM}

Stem cell sources: Bone marrow {BM} q Marrow is obtained by multiple aspirations from the posterior iliac crest under general or epidural anesthesia. q For larger quantity anterior crest or sternum can be the site. Several skin puncture on each iliac crest and multiple bone puncture are usually required. q Target volume is 10 -15 mg/kg of recipient or donor weight , whichever is less. q Marrow is collected in heparinized syringe and filtered through 0. 3 -0. 2 mm screen to remove the fat and bony spicule.

q Further processing depend on the clinical situation, such as § Removal of RBCs to prevent hemolysis in ABO incompatible transplants {In BMSCT ABO mismatch is the most common} § Removal of immunocompetent donor T cells to prevent GVHD § Attempts to remove possible contamination of tumour cells in autologous transplantation. q Marrow is usually transfused immediately after harvesting, but delay of upto 24 hrs may occur without adverse consequences.

Peripheral blood {PB } q These cells are now the most common source of stem cells for HSCT. q Peripheral blood stem cells {PBSC} are collected by leokopheresis after the donor has been treated with hematopoietic growth factors or a combination of chemotherapy and growth factors. For pts. with malignancy cyclophosphamide based chemotherapy and G –CSF are used. q The donors are typically treated with growth factor for 4 -6 days, following which the stem cells are collected in one to two 4 hrs sessions. q In autologous setting transplantation of >2. 5 X 106 CD 34+ cells / kg leads to rapid and sustained engraftment.
Peripheral Blood Stem Cell Transplant PBSCs are easier to collect than bone marrow stem cells, which must be extracted from within bones. This makes PBSCs a less invasive treatment option than bone marrow stem cells.

Umbilical cord {UC} q UC blood is a rich source of primitive stem cells as compared to adult stem cells. Therefore these are able to expand rapidly and reconstitute haematopoiesis after myeloablative chemotherapy. q Another advantage is the relative immaturity of the immune system at birth resulting in significant lower risk of acute GVHD compared to adult BM/PB stem cells. q Since the total yield is limited presently UC blood is being used mainly for children weighing upto 25 kg.

q Immediately after a baby is delivered umbilical cord is clamped. After delivery of placenta , the placenta is placed on supporting frame , the cord is cleaned and needle is inserted into the umbilical vein. The umbilical cord blood is collected in a closed system and blood drained as a “standard gravity phlebotomy” into CPD anticoagulant.

q Second method involves collecting the cord blood while the placenta is still in the mother’s womb. q This method has theoretically two advantages § collection begins earlier before the blood has a chance to clot. § It uses the contraction of the uterus to enhance the blood drainage in addition to the gravity. Disadvantage: it is more intrusive and has the potential to interfere with after-delivery care for the mother and infant. The cord blood collected from single placenta is called a cord blood unit ranging from 60 -120 ml

Storage of haematopoietic stem cell q Bone marrow cells can be cryopreserved for prolonged time. This is necessary for autologous HSC because the cells must be harvested months in advance of the transplant treatment. q In allogenic transplants fresh HSC are preferred in order to avoid cell loss that might occur during the freezing and thawing process. q The graft undergoes HLA typing, cell counts and testing for viruses. q Allogenic cord blood is stored frozen at a cord blood bank because it is only obtainable at the time of child birth. q To cryopreserved HSC a preservative (dimethyl sulfoxide) DMSO , must be added and cells must be cooled very slowly in a control rate freezer to prevent osmotic cellular injury during ice crystal formation. q HSC may be stored for years in a cryofreezer.

Types of hematopoietic stem cell transplantation q I. Syngeneic - From an identical twin q II. Allogenic – 1. Sibling (HLA identical Or HLA- A, B, DR matched donor) 2. Matched unrelated q III. Autologous – 1. Bone Marrow of patient himself is injected. 2. PBSC are usually used in autologous BMT. 3. GM-CSF & G-CSF have been used successfully for collection of PBSC’s. Since these drugs result in at least 10 fold rise in CD 34+ progenitor cell population in peripheral blood q Haemopoietic Stem Cell Marker is CD 34+ q Stem cell dose – § BMSC : - 3 x 108/kg – § PBSC : - 5 x 108/kg – § CD 34+ : - 2. 5 x 106/kg
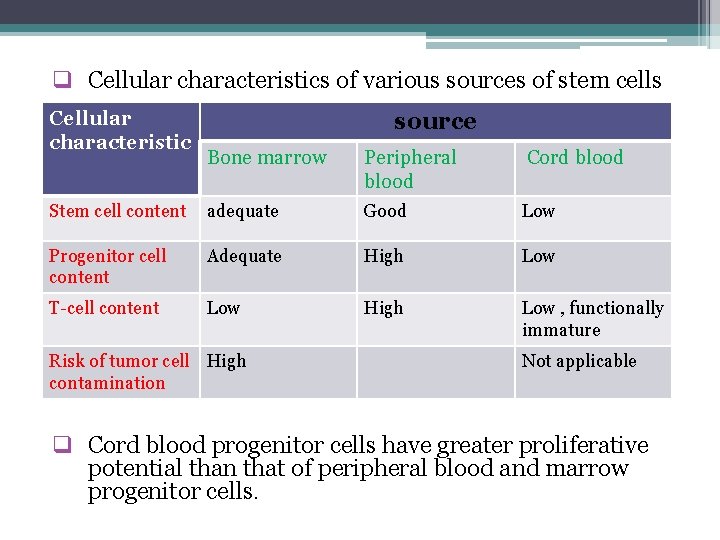
q Cellular characteristics of various sources of stem cells Cellular characteristic source Bone marrow Peripheral blood Cord blood Stem cell content adequate Good Low Progenitor cell content Adequate High Low T-cell content Low High Low , functionally immature Risk of tumor cell High contamination Not applicable q Cord blood progenitor cells have greater proliferative potential than that of peripheral blood and marrow progenitor cells.

Clinical characteristics with various sources of stem cells Cellular characteristic source Bone marrow Peripheral blood Cord blood HLA Matching Close matching required Less restrictive than other Engraftment Fastest Intermediate Slowest Risk of acute GVHD Same as in bone marrow Same as in peripheral blood Lowest Risk of chronic GVHD Highest Lower than peripheral blood Lowest

The transplantation process q Conditioning regimen § The chemotherapy or irradiation given immediately prior to the transplantation is called the conditioning or preparative regimen. § Purpose: to eradicate pt`s residual malignant cells and to cause immunosuppression.

Classification q Myeloablative q Non myeloablative q Reduces intensity

Myeloablative q Myeloablative regimen is designed to kill all residual cancer cells and to cause immunosuppression. q Total body irradiation {TBI} and cyclophosphamide are the commonly used myeloablative therapies. q Used especially in aggressive malignancies - leukemias

Non myeloablative q Non myeloablative regimen uses doses lower than those of myeloablative regimen. q These regimens are only immunosuppressive and rely on graft versus tumor effect to kill tumor cells with donor T lymphocytes. q Used in older and morbid pts. for slow growing tumors.

q Reduced intensity regimens are between myeloablative and non-myeloablative regimen. q Tandem Transplant § A type of autologous transplant. § Pts receives two sequential courses of high dose chemotherapy with stem cells transplant which are several weeks to months apart. § This prevents the recurrence of cancer at a later time. q Purging § It removes some cancer cells from the harvested cells and minimize the chances of recurrence of cancer.
Stem cell processing and infusion • Infusion is performed over about an hour {varies depending upon volume to be infused}. • Processing is performed if indicated. Depletion of T cells can be performed to decrease GVHD. Stem cell CD 34+ selection may be performed either for depletion of T cells or for tumor purging process. • Before infusion pt is premedicated with acetaminophen and diphenylhydramine to prevent reaction. • Stem cells are infused through central venous catheter.

q Neutropenic Phase § During this period of 2 -4 wks pt has no effective immune system. Healing is poor and the pt is susceptible to infection. § Supportive care and empiric antibiotic therapy are the mainstay. § HSV is an important pathogen {reactivation of previous infection}. § Endogenous flora {skin and gut flora } are most frequently observed organisms. § Hospital acquired nosocomial infection poses the biggest risk {Drug resistance}

q Typically fever manifests 5 -7 days following the start of broad spectrum antibiotics. It is treated empirically with antifungal agents {amphotericin}. q Oral intake is markedly reduced { severe mucositis}. Total parenteral nutrition {TPN} is provided especially for children. q Recombinant hematopoietic growth factors {e. g. filgrastim or sargramostin} reduces the time of blood neutrophils recovery. q Transfusion of platelet to prevent bleeding.

Engraftment Phase q Successful engraftment results from the circulation, homing and growth of the stem cells. q For stable long term engraftment a nucleated cell dose of 2 x 108 cells / kg is generally considered as adequate. q A 5 yr survival is better when CD 34+ cells ≥ 3 x 108 cells / kg of recipient body weight. q Engraftment has been defined as achievement of peripheral blood neutrophil count >0. 5 x 109 / L. q Rate of engraftment has been depends on: - § Number of haematopoietic stem cells , especially the CD 34+ cell content. § The purging strategies used for autologous marrow. § Use of growth factor § Use of MTX for GVHD prohylaxis.

Post engraftment phase q This period lasts for months to years. q Hallmark: - gradual development of tolerance, weaning off of immunosuppression, management of chronic GVHD and documentation of immune reconstitution. q Pt may show delayed or incomplete immune reconstitution or splenic dysfunction.

q Most pt need reimmunization {1 Yr post transplant} q Pt without chronic GVHD show good response to Pnuemococcal polysaccharide, inactivated polio virus, diphtheria, partussis, tetanus toxoid, H. Influenza type B conjugate vaccine and hepatitis B. q Pt treated with immunosuppressive medication to control chronic GVHD may have inadequate response after revaccination. q All other live virus vaccine should be avoided if at all possible.

q In general favorable outcome is seen in; Pt with stable disease or disease in remission have better outcome. Younger age at time of transplant. CMV negative status q Graft rejection is infrequent {<1 -2 % incidence} after HSCT from HLA matched sibling. q Factors that influence risk of graft rejection § T cell depletion of marrow graft § Increased HLA disparity § Intensity of conditioning regimen and post transplant immunosuppression. § No of stem cells in graft

Side effect and complication following HSCT q Regimen Related Toxicity (RRT) The severity of RRT is related to ─ intensity of myoablative therapy used. ─ Type of cytotoxic therapy ─ Medical condition of the pt. before transplantation. ─ Presence of post transplant factor {e. g. amphotericin B , MTX. }

q Early effects- - Mucositis Early GVHD Veno-occlusive disease Transplantation related lung injury Transplantation related infections- § Bacterial {Early after transplantation} § Viral {after engraftment} § Fungal {any time between 7 -10 days after onset of neutropenia}

q Late effects – - Chronic GVHD Ocular effects Pulmonary effects Endocrine effects Musculoskeletal effects Neurocognitive and neuropsychiatric effects Immune effects.
Diseases for which treatment is under trial q Type 1 diabetes q Nervous system diseases § § Parkinson disease Alzheimer disease Amyotrophic lateral sclerosis Spinal cord injury q Primary Immune deficiency diseases § Severe combined immune deficiency disease § Wiscott Aldrich syndrome § SLE q Diseases of bone and cartilage § § Osteogenesis Imperfecta Chondrodysplasia Osteoarthritis Fracture/ surgery q Cancers

Stem cells proved to be a cure forq q q Acute and chronic leukemia Sickle cell disease B- thalasemia Hodgkins disease Stem cell plasma cell disorder Ewing' s sarcoma

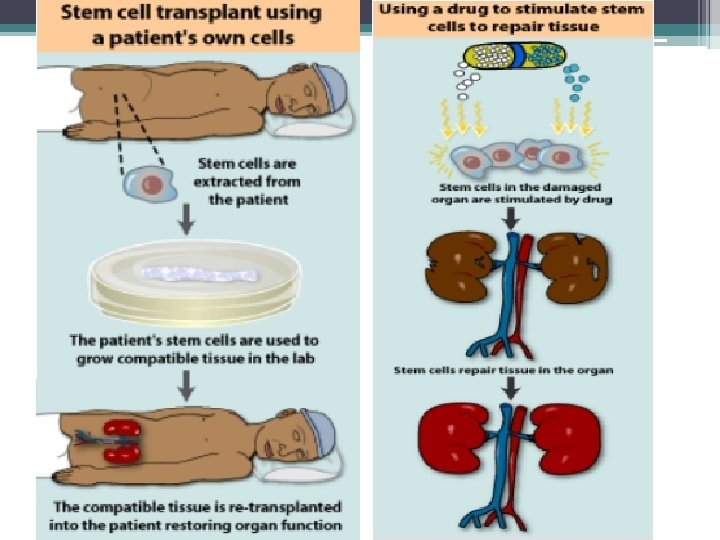
Problems with stem cell transplantation q One major problem that must be solved before human stem cell therapy becomes a reality is the threat of rejection of the transplanted cells by the host’s immune system (if the stem cells are allografts; that is, come from a geneticallydifferent individual). q A Solution? q One way to avoid the problem of rejection is to use stem cells that are genetically identical to the host. q This is already possible in the rare situations when the patient has healthy stem cells in an undamaged part of the body (like the stem cells being used to replace damaged corneas).

q But what about the situation when no "autologous" stems cells are available? q There may be a solution: using somatic-cell nuclear transfer (but with no goal of attempting to implant the resulting blastocyst in a uterus). q In this technique, § A human egg has its own nucleus removed and § replaced by a nucleus taken from a somatic (e. g. , skin) cell of the patient. § The now-diploid egg is allowed to develop in culture to the blastocyst stage when § embryonic stem cells can be harvested and grown up in culture. § When they have acquired the desired properties, they can be implanted in the patient with no fear of rejection. q On 11 November 2007, scientists in Oregon reported success with steps 1– 4 in rhesus monkeys. While this increases the probability of being able to apply the procedure to humans, there are still questions with the method that must be answered.

In the future…. q It may be possible for a person to use a sample of his or her own stem cells to regenerate tissue, which would reduce or even eliminate the danger of rejection. q How might this be done? Some possibilities include: § Collecting healthy adult stem cells from a patient and manipulating them in the laboratory to create new tissue. The tissue would be re-transplanted back into the patient’s body, where it would work to restore a lost function. § Therapeutic cloning, as described in Creating Stem Cells for Research , might enable the creation of embryonic stem cells that are genetically identical to the patient. § One less invasive way to achieve this goal would be to manipulate existing stem cells within the body to perform therapeutic tasks. For example, scientists might design a drug that would direct a certain type of stem cell to restore a lost function inside the patient’s body. This approach would eliminate the need for invasive surgical procedures to harvest and transplant stem cells.

Key research events q 1908 - The term "stem cell" was proposed for scientific use by the Russian histologist Alexander Maksimov (1874 -1928) at congress of hematologic society in Berlin. It postulated existence of haematopoietic stem cells. q 1960 s - Joseph Altman and Gopal Das present scientific evidence of adult neurogenesis , ongoing stem cell activity in the brain; their reports contradict Cajal ‘s "no new neurons" dogma and are largely ignored. q 1963 - Mc. Culloch and Till illustrate the presence of self-renewing cells in mouse bone marrow. q 1968 - Bone marrow transplant between two siblings successfully treats SCID. q 1978 - Haematopoietic stem cells are discovered in human cord blood. q 1981 - Mouse embryonic stem cells are derived from the inner cell mass by scientists Martin Evans , Matthew Kaufman , and Gail R. Martin. Gail Martin is attributed for coining the term "Embryonic Stem Cell". q 1992 - Neural stem cells are cultured in vitro as neurospheres. q 1997 - Leukemia is shown to originate from a haematopoietic stem cell, the first direct evidence for cancer stem cells. q 1998 - James Thomson and coworkers derive the first human embryonic stem cell line at the University of Wisconsin-Madison. [34] q 2000 s - Several reports of adult stem cell plasticity are published

q 2001 - Scientists at Advanced Cell Technology clone first early (four- to six-cell stage) human embryos for the purpose of generating embryonic stem cells. [35] q 2003 - Dr. Songtao Shi of NIH discovers new source of adult stem cells in children’s primary teeth. [36] q 2004 -2005 - Korean researcher Hwang Woo-Suk claims to have created several human embryonic stem cell lines from unfertilised human oocytes. The lines were later shown to be fabricated. q 2005 - Researchers at Kingston University in England claim to have discovered a third category of stem cell, dubbed cord-bloodderived embryonic-like stem cells (CBEs), derived from umbilical cord blood. The group claims these cells are able to differentiate into more types of tissue than adult stem cells. q August 2006 - Rat Induced pluripotent stem cells : the journal Cell publishes Kazutoshi Takahashi and Shinya Yamanaka, "Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors". q October 2006 - Scientists at Newcastle University in England create the first ever artificial liver cells using umbilical cord blood stem cells. [37] [38]

q January 2007 - Scientists at Wake Forest University led by Dr. Anthony Atala and Harvard University report discovery of a new type of stem cell in amniotic fluid. [6] This may potentially provide an alternative to embryonic stem cells for use in research and therapy. [39] q June 2007 - Research reported by three different groups shows that normal skin cells can be reprogrammed to an embryonic state in mice. [40] In the same month, scientist Shoukhrat Mitalipov reports the first successful creation of a primate stem cell line through somatic cell nuclear transfer [41] q October 2007 - Mario Capecchi , Martin Evans , and Oliver Smithies win the 2007 Nobel Prize for Physiology or Medicine for their work on embryonic stem cells from mice using gene targeting strategies producing genetically engineered mice (known as knockout mice ) for gene research. [42] q November 2007 - Human Induced pluripotent stem cells : Two similar papers released by their respective journals prior to formal publication: in Cell by Kazutoshi Takahashi and Shinya Yamanaka , "Induction of Pluripotent Stem Cells from Adult Human Fibroblasts by

q January 2008 - Robert Lanza and colleagues at Advanced Cell Technology and UCSF create the first human embryonic stem cells without destruction of the embryo [43] q January 2008 - Development of human cloned blastocysts following somatic cell nuclear transfer with adult fibroblasts [44] q February 2008 - Generation of Pluripotent Stem Cells from Adult Mouse Liver and Stomach: these i. PS cells seem to be more similar to embryonic stem cells than the previous developed i. PS q Defined Factors" , and in Science by Junying Yu, et al. , from the research group of James Thomson , "Induced Pluripotent Stem Cell Lines Derived from Human Somatic Cells" : pluripotent stem cells generated from mature human fibroblasts. It is possible now to produce a stem cell from almost any other human cell instead of using embryos as needed previously, albeit the risk of tumorigenesis due to cmyc and retroviral gene transfer remains to be determined

q cells and not tumorigenic, moreover genes that are required for i. PS cells do not need to be inserted into specific sites, which encourages the development of non-viral reprogramming techniques. [45] [46] q March 2008 -The first published study of successful cartilage regeneration in the human knee using autologous adult mesenchymal stem cells is published by Clinicians from Regenerative Sciences [47] q October 2008 - Sabine Conrad and colleagues at Tübingen, Germany generate pluripotent stem cells from spermatogonial cells of adult human testis by culturing the cells in vitro under leukemia inhibitory factor (LIF) supplementation. [48] q 30 October 2008 - Embryonic-like stem cells from a single human hair. [49]

THANKS
- Slides: 70