Stem Cell Biology Jim Huettner 11172015 suggested readings
Stem Cell Biology Jim Huettner 11/17/2015

suggested readings • Solter D. (2006) From teratocarcinomas to embryonic stem cells and beyond: a history of embryonic stem cell research. Nat Rev Genet. 7: 319 -27. • Buganim Y, Faddah DA, Jaenisch R. Mechanisms and models of somatic cell reprogramming. Nat Rev Genet. 2013 Jun; 14(6): 427 -39. • Buganim Y, Markoulaki S, van Wietmarschen N, et al. (2014) The developmental potential of i. PSCs is greatly influenced by reprogramming factor selection. Cell Stem Cell. 15: 295 -309. . • De Los Angeles et al. (Daley GQ). (2015) Hallmarks of pluripotency. Nature 525: 469 -478. • Fox IJ, Daley GQ, Goldman SA, Huard J, Kamp TJ, Trucco M. (2014) Stem cell therapy. Use of differentiated pluripotent stem cells as replacement therapy for treating disease. Science 345(6199): 1247391. • Schwartz SD, Regillo CD, Lam BL et al. , (2014) Human embryonic stem cellderived retinal pigment epithelium in patients with age-related macular degeneration and Stargardt’s macular dystrophy: follow-up of two open-label phase 1/2 studies. Lancet. e-pub October 15.

Stem Cells: definition • Self Renewal - undifferentiated cells that can divide repeatedly while maintaining their undifferentiated state. • Pluripotency – ability to differentiate into a variety of different cell types
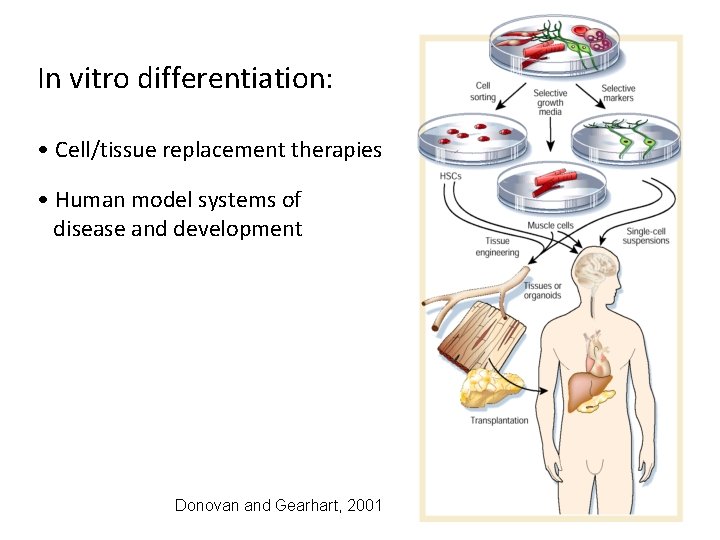
In vitro differentiation: • Cell/tissue replacement therapies • Human model systems of disease and development Donovan and Gearhart, 2001

Types of Stem Cells Embryonic – from the inner cell mass of preimplantation embryos, prior to formation of the 3 germ layers (ectoderm, mesoderm, endoderm) Somatic – undifferentiated cells found in specific locations in “mature” tissues i. PS cells – induced pluripotent stem cells generated by reprogramming differentiated cells (or cell nuclei, i. e. therapeutic cloning)

Potency • Totipotent – able to generate every cell type including extraembryonic tissues • Pluripotent – able to generate cells from all three embryonic germ layers • Multipotent – able to generate a variety of cells from a particular somatic structure • Unipotent – only generate one cell type
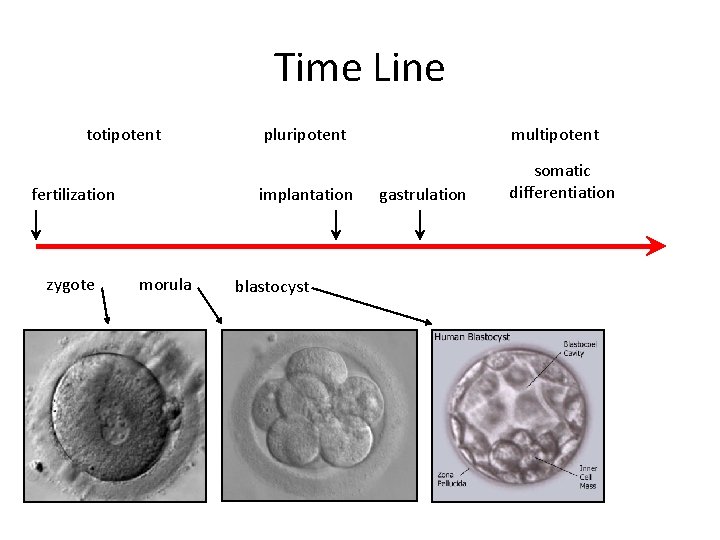
Time Line totipotent fertilization zygote morula pluripotent multipotent implantation somatic differentiation blastocyst gastrulation
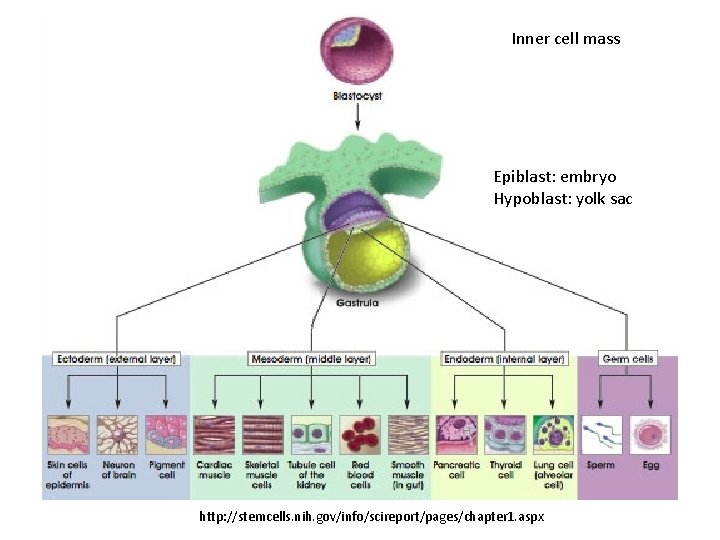
Inner cell mass Epiblast: embryo Hypoblast: yolk sac http: //stemcells. nih. gov/info/scireport/pages/chapter 1. aspx
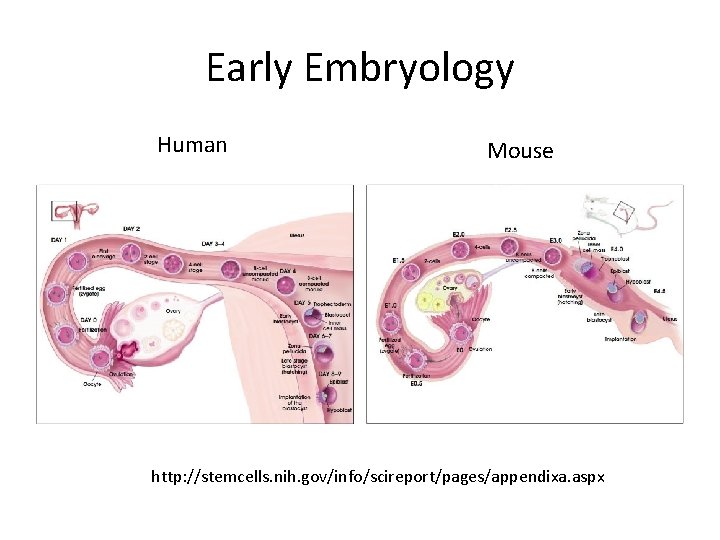
Early Embryology Human Mouse http: //stemcells. nih. gov/info/scireport/pages/appendixa. aspx
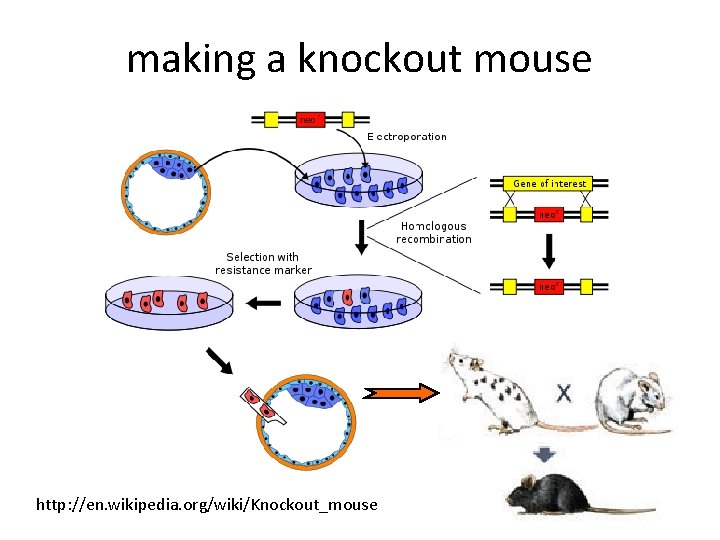
making a knockout mouse http: //en. wikipedia. org/wiki/Knockout_mouse

First Isolation of ES cells Mouse: Evans MJ, Kaufman MH. (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature. 292: 154 -6. Martin GR. (1981) Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. P. N. A. S. U S A. 78: 7634 -8. Human: Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. (1998) Embryonic stem cell lines derived from human blastocysts. Science. 282: 1145 -7. Genetic and Developmental Normality (140 cycles): Suda Y, Suzuki M, Ikawa Y, Aizawa S. (1987) Mouse embryonic stem cells exhibit indefinite proliferative potential. J Cell Physiol. 133: 197 -201.

Pluripotency markers • Stage-specific antigens: Anti-SSEA 3 and 4 recognize globo-series gangliosides • Tra 1 -60 and Tra 1 -81: keratin sulfate surface antigens • Oct 3/4, Sox 2, Nanog – transcription factors involved with maintaining pluripotency • Normal karyotype, and pre-X-inactivation?

Two types of ES cells? “Naïve” (ICM-like) • • • Blastocyst chimera (+) High cloning efficiency Short doubling time Xa Xa Distal Oct 4 enhancer High Nanog, Klf 2/4, Rex 1 “Primed” (Epi-SC) • • • Blastocyst chimera (-) Low cloning efficiency Long doubling time Xa Xi Proximal Oct 4 enhancer Low Nanog, Klf 2/4, Rex 1 Both types can self renew and give rise to cells from all 3 germ layers in teratomas or following in vitro differentiation

maintenance of pluripotency - 1 • Initial work done on mouse embryonic fibroblast (MEF) feeder cells in medium supplemented with animal serum • One factor produced by feeder cells that helps maintain mouse ES cells in their undifferentiated state is leukemia inhibitory factor (LIF) which activates the Stat 3 pathway. • Good Manufacturing Process (GMP) – guidelines for isolation and propagation of cells that would be used for replacement therapy. Ideally they would be xeno-free. • The push for xeno-free conditions, combined with work to optimize reprogramming, has driven screening of factors that can enable serum-free maintenance of pluripotency

maintenance of pluripotency - 2 “naïve” • • Positive Regulators LIF - Stat 3 BMP 4 - Smad 1/5 Wnt (GSK-3 inhibitors) IGF • • • “primed” TGFb/activin – Smad 2/3 FGF 2 ERK 1/2 Wnt (GSK-3 inhibitors) IGF Negative Regulators • TGFb/activin-Smad 2/3 • FGF 2 • ERK 1/2 • BMP 4 – Smad 1/5

maintenance of pluripotency - 3 “Current Standard” Conditions Mouse (2008) • LIF - Stat 3 • GSK-3 inhibitors (Wnt) • ERK 1/2 inhibitors (2 i/LIF) For serum free growth also need: Insulin, transferrin, progesterone, putrescine, selenium Human (2013) • • • LIF – Stat 3 GSK-3 inhibitors (Wnt) ERK 1/2 inhibitors PKC inhibitor p 38 inhibitor JNK inhibitor ROCK inhibitor FGF 2 TGF-b 1 but see: Takashima et al. , Cell 158: 1254 -1269 (2014)

Attempts to define “Stemness” • Early microarray profiles showed surprising lack of agreement (limitations in microarray technology or platform/lab/primary cell or cell line differences) (Science 302: 393, 2003) • Relatively weak overlap between mouse and human ES cells (~25%) compared to >90% typical for differentiated tissues. (Stem Cell Reviews 1: 111 -118, 2005) but this may reflect confusion between naïve and primed ES cells

In vitro differentiation • Different culture conditions alter the fate of ES cells in vitro • Protocols exist for all three germ layers • Many, but not all, protocols involve aggregation of ES cells in “embryoid bodies” • Most protocols do not yield a single type of cell • Selection steps can help to remove undesired cell types • Need to ask: How far? & How faithful? Pancreatic β cells: • Pagliuca FW, et al. (2014) Generation of Functional Human Pancreatic β Cells In Vitro. Cell. Oct, 159: 428 -39. • Rezania A, et al. (2014) Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat Biotechnol. Nov, 32: 1121 -33.
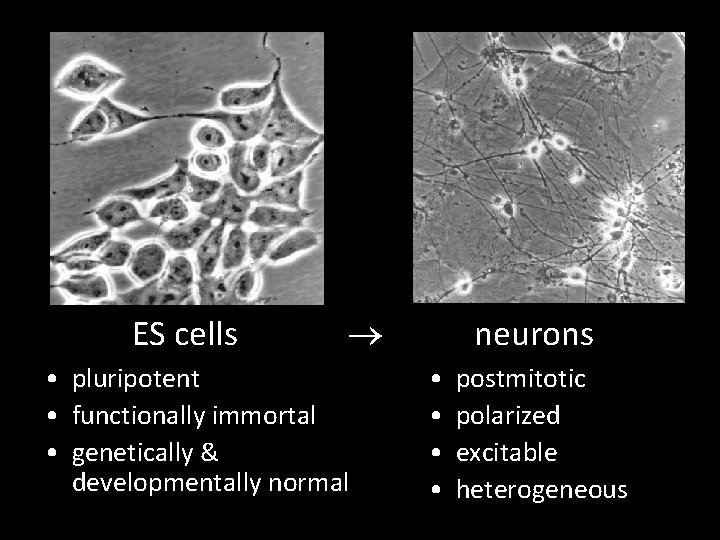
ES cells ® • pluripotent • functionally immortal • genetically & developmentally normal neurons • • postmitotic polarized excitable heterogeneous
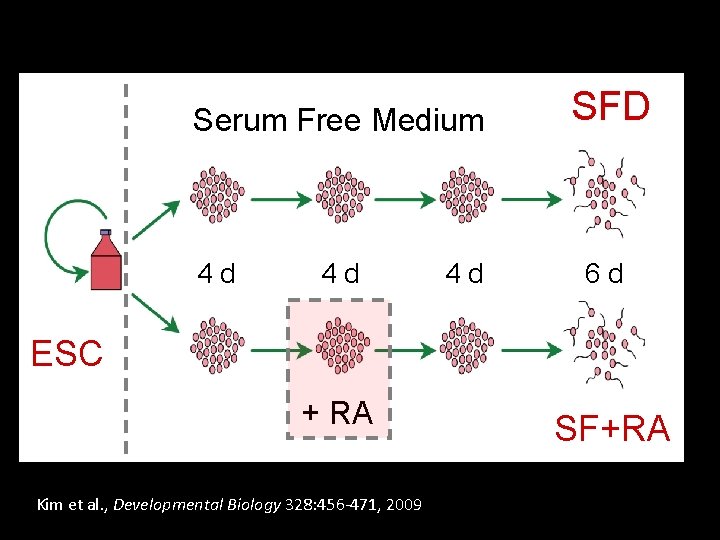
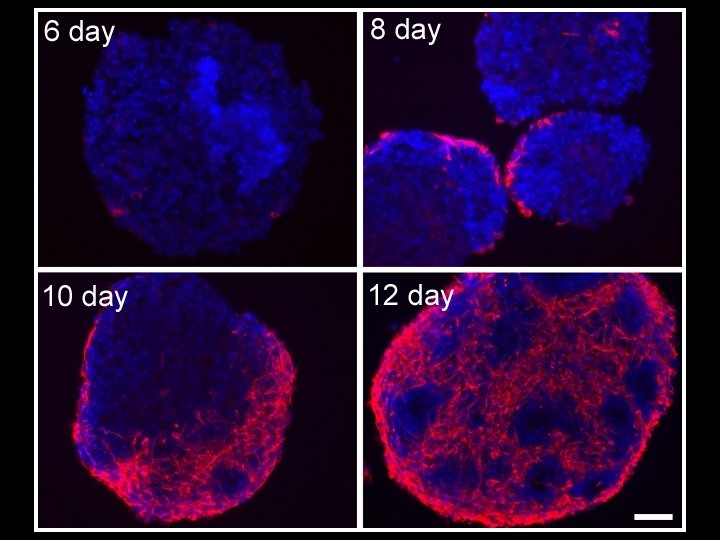
Serum Free Medium 4 d 4 d 4 d SFD 6 d ESC + RA Kim et al. , Developmental Biology 328: 456 -471, 2009 SF+RA
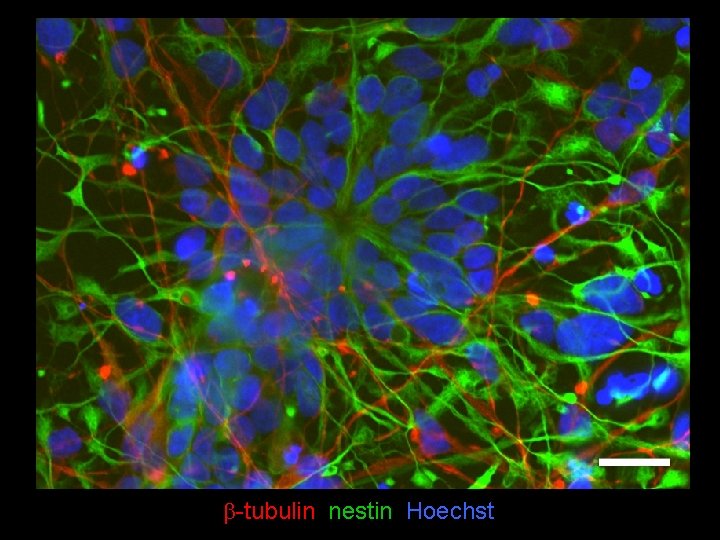
b-tubulin nestin Hoechst
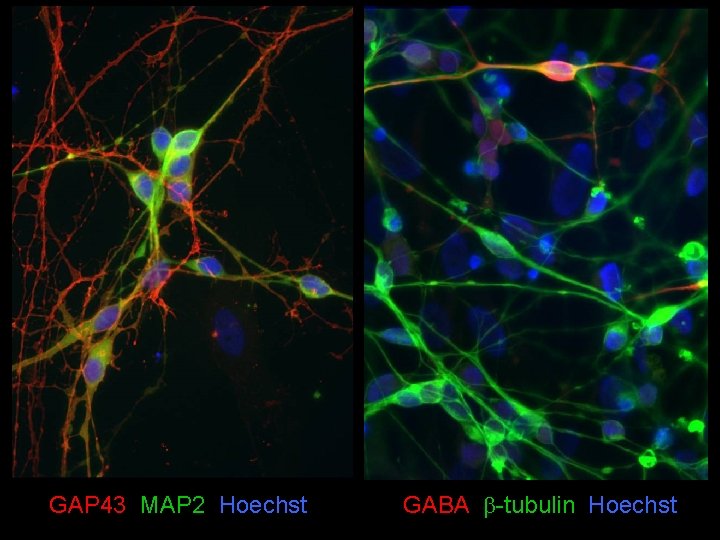
GAP 43 MAP 2 Hoechst GABA b-tubulin Hoechst

voltage-gated Na+ and K+ currents Developmental Biology 168: 342 -357, 1995
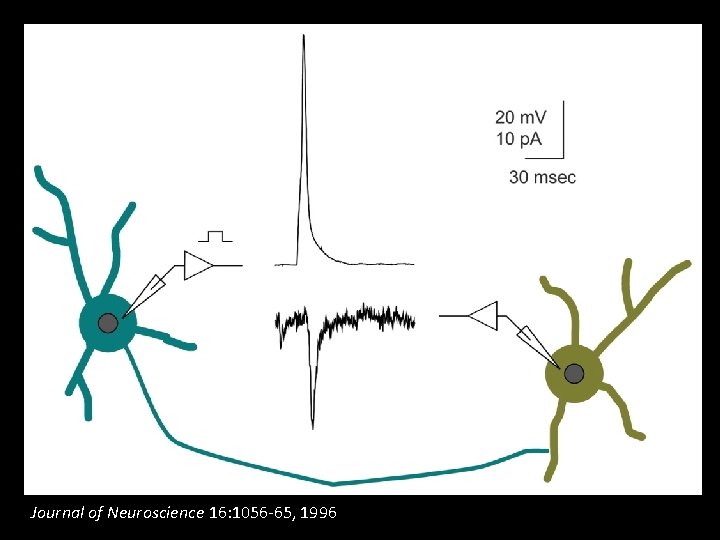
Journal of Neuroscience 16: 1056 -65, 1996
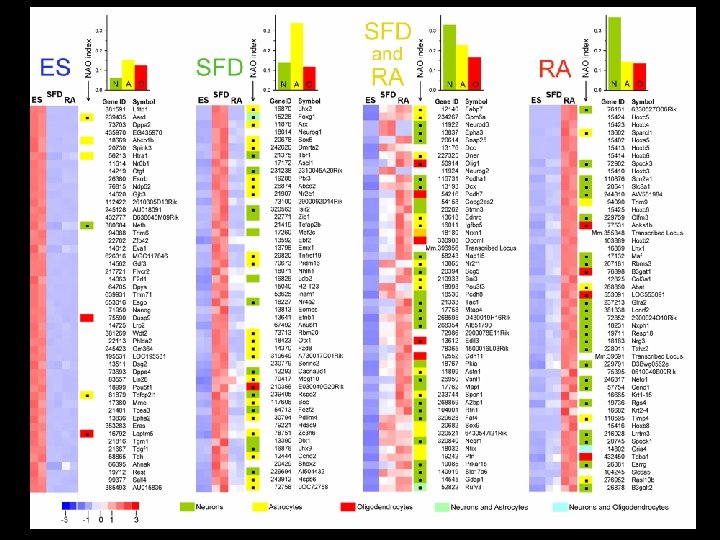
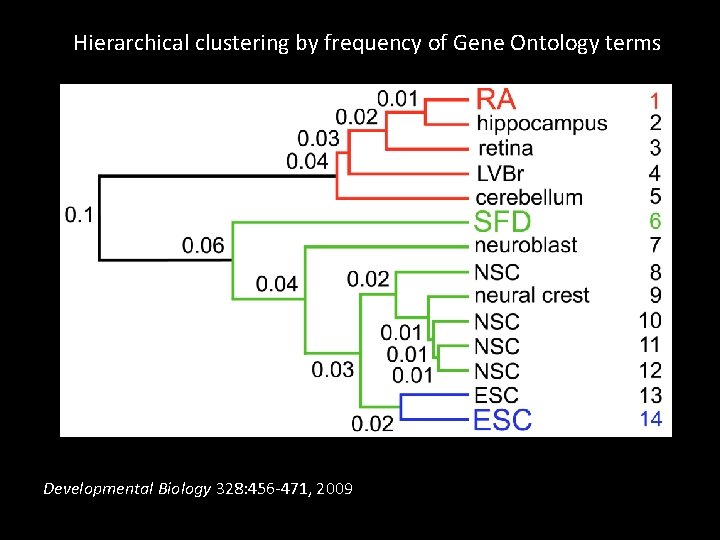
Hierarchical clustering by frequency of Gene Ontology terms Developmental Biology 328: 456 -471, 2009

Reprogramming • SCNT – somatic cell nuclear transfer (reproductive and therapeutic cloning) – deterministic and fairly rapid • i. PS – induced pluripotent stem cells – slow and stochastic (until recently) • Transdifferentiation – conversion of one terminally differentiated cell type into another without dedifferentiation to an immature phenotype. Must rule out cell fusion or other explanations.
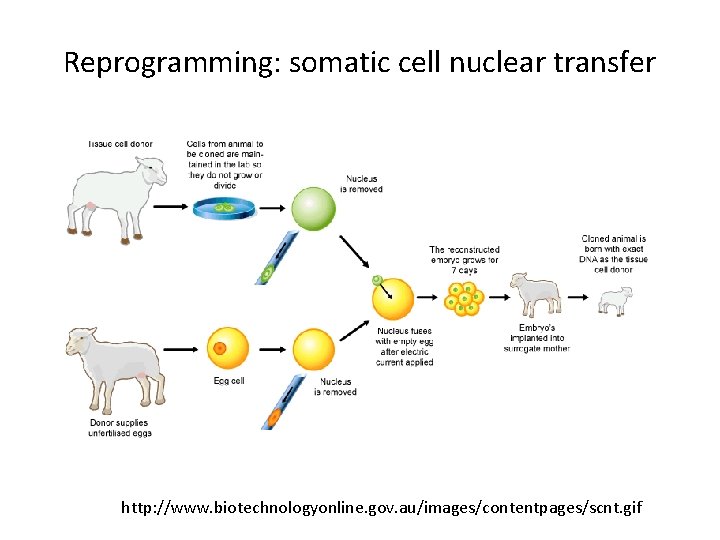
Reprogramming: somatic cell nuclear transfer http: //www. biotechnologyonline. gov. au/images/contentpages/scnt. gif

Reprogramming Firsts: SCNT Frog: Gurdon JB. (1962) Adult frogs derived from the nuclei of single somatic cells. Dev Biol. 4: 256 -73. Sheep: Campbell KH, Mc. Whir J, Ritchie WA, Wilmut I. (1996) Sheep cloned by nuclear transfer from a cultured cell line. Nature. 380: 64 -6. Human: (2004) – Claim of human SCNT that proved to be unfounded! Tachibana M, et al. (2013) Human embryonic stem cells derived by somatic cell nuclear transfer. Cell. 153: 1228 -38.

Reprogramming Firsts: i. PS cells Mouse: Takahashi K, Yamanaka S. (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 126: 663 -76 Human: Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 131: 861 -72. Yu J, Vodyanik MA, Smuga-Otto K, et al. , (2007) Induced pluripotent stem cell lines derived from human somatic cells. Science. 318: 1917 -20.

Generating i. PS cells • Express transcription factors: Oct 3/4, Sox 2, Klf 4 and c-Myc OR Oct 3/4, Sox 2, Nanog and Lin 28 • Initial de-differentiation and proliferation (day 1 -3, enhanced by Myc); histone modification and chromatin reorganization • 2 nd wave of gene expression - stem cell and development related genes (day 9 -12); DNA demethylation and X reactivation
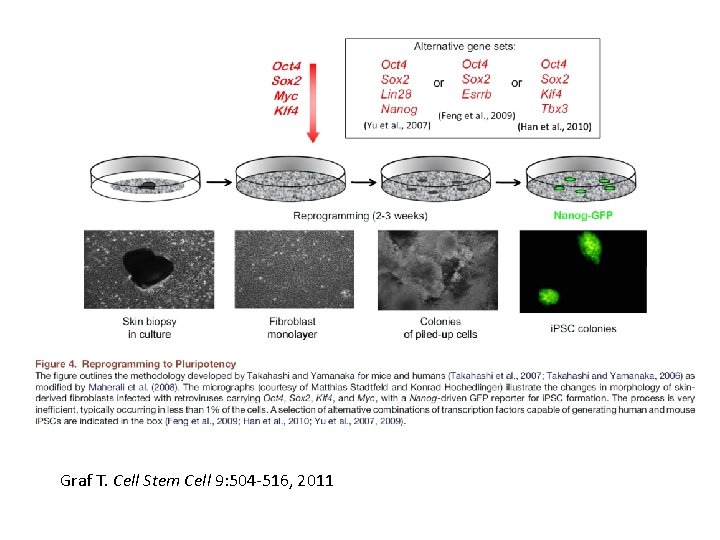
Graf T. Cell Stem Cell 9: 504 -516, 2011
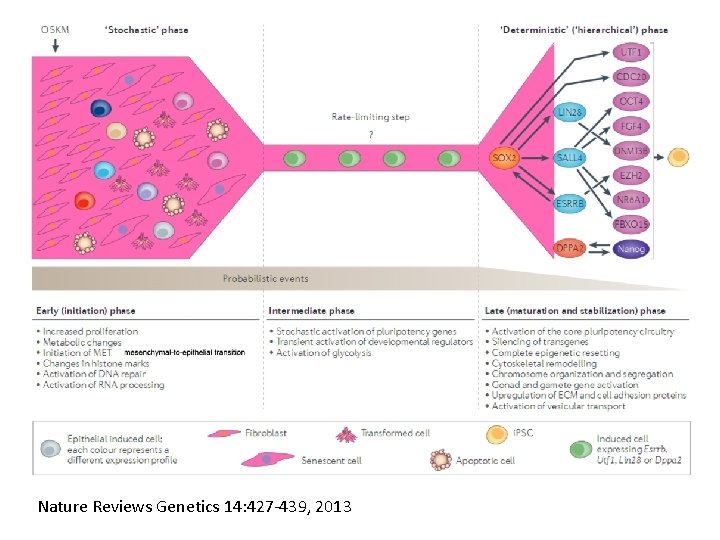
Nature Reviews Genetics 14: 427 -439, 2013

Removing the bottle neck? • Rais et al. , Nature 502: 65 -70, 2013 implicate Mbd 3, a component in the Nu. RD complex that mediates gene repression via histone deacetylation and chromatin remodeling. • Argue that the reprogramming factors recruit both repressive (Mbd 3/Nu. RD) and de-repressive (Wdr 5 and Utx) complexes, and reprogramming only occurs when the Mbd 3/Nu. Rd repression loses. • Achieve nearly 100% reprogramming within 7 days in cells with Mbd 3 reduced or eliminated.
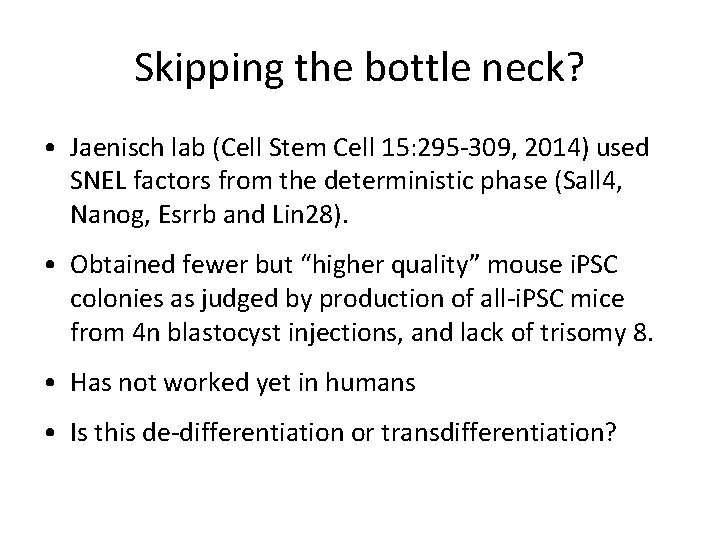
Skipping the bottle neck? • Jaenisch lab (Cell Stem Cell 15: 295 -309, 2014) used SNEL factors from the deterministic phase (Sall 4, Nanog, Esrrb and Lin 28). • Obtained fewer but “higher quality” mouse i. PSC colonies as judged by production of all-i. PSC mice from 4 n blastocyst injections, and lack of trisomy 8. • Has not worked yet in humans • Is this de-differentiation or transdifferentiation?

Transdifferentiation • Conversion from one differentiated cell type to another without evident de-differentiation and re-differentiation • Must not be confused by cell fusion or selection for rare pluripotent cells in the source material. • Induced by expression of transcription factors and micro. RNAs
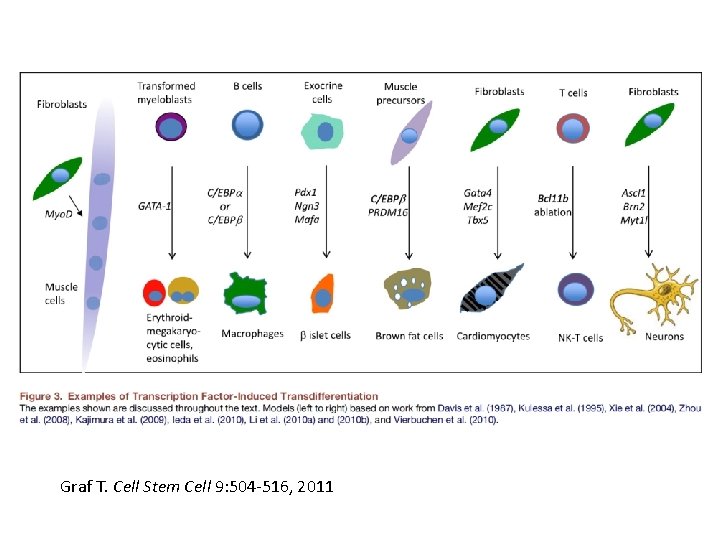
Graf T. Cell Stem Cell 9: 504 -516, 2011

Fibroblasts to neurons • Wernig and colleagues screened 19 transcription factors via lentiviral expression • Found 5 were most critical Asc 1, Brn 2, Olig 2, Zic 1 and Myt 1 l, and 3 were sufficient • 20% conversion within 2 weeks • For human fibroblast conversion also require Neuro. D 1 and it is less efficient (2 -4%) and slower (5 -6 weeks for functional synapses)
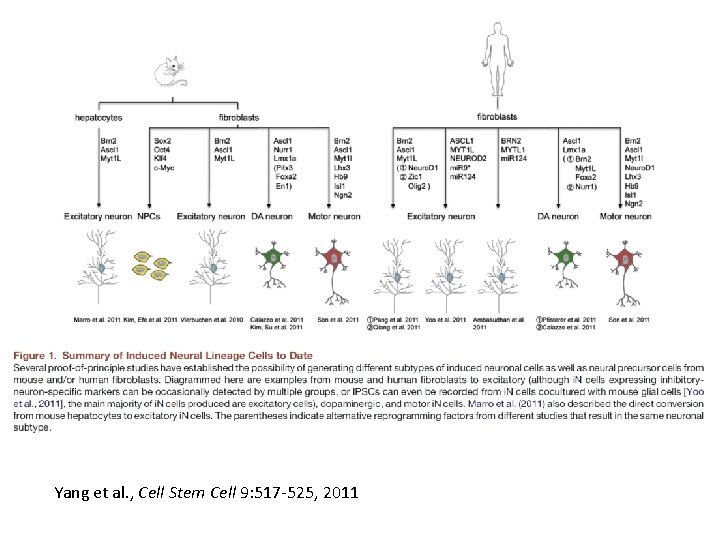
Yang et al. , Cell Stem Cell 9: 517 -525, 2011

Conversion process • Asc 1 b. HLH transcription factor binds to many of the same genomic loci when expressed in fibroblasts, myoblasts or neural progenitors. • These sites are marked by specific histone modifications (H 3 K 4 me 1, H 3 K 27 acetyl, H 3 K 9 me 3) • these sites are not accessible in keratinocytes or osteoblasts, which resist transdifferentiation into neurons. • Brn 2 Pou-Homeodomain transcription factor is recruited by Asc 1 to a subset of locations

Evaluation • SCNT vs i. PSCs from isogenic cells: Ma H, Morey R, O'Neil RC, et al. , (2014) Abnormalities in human pluripotent cells due to reprogramming mechanisms. Nature 511: 177 -83. Johannesson B, Sagi I, Gore A, et al. , (2014) Comparable frequencies of coding mutations and loss of imprinting in human pluripotent cells derived by nuclear transfer and defined factors. Cell Stem Cell 15: 634 -642. • Origin-dependence after i. PSC differentiation: Hargus G, Ehrlich M, Araúzo-Bravo MJ, et al. , (2014) Origin-dependent neural cell identities in differentiated human i. PSCs in vitro and after transplantation into the mouse brain. Cell Reports 8: 1697 -703. • An optimization strategy? Morris SA, Cahan P, Li H, et al. , (2014) Dissecting engineered cell types and enhancing cell fate conversion via Cell. Net. Cell 158: 889 -902.
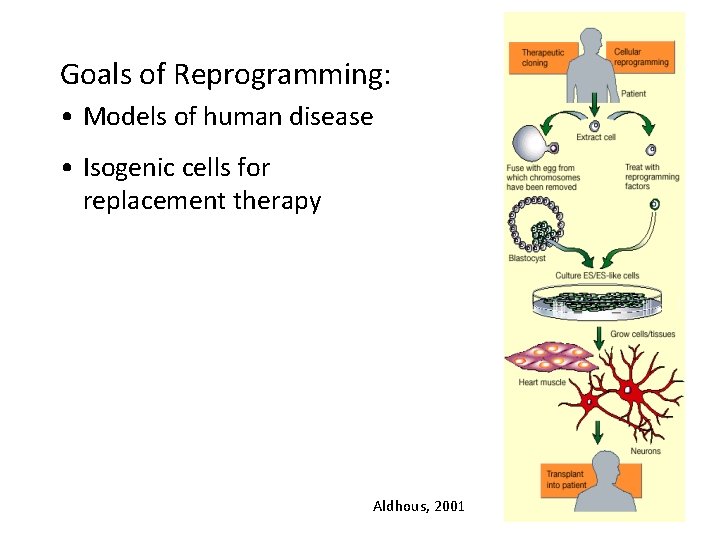
Goals of Reprogramming: • Models of human disease • Isogenic cells for replacement therapy Aldhous, 2001
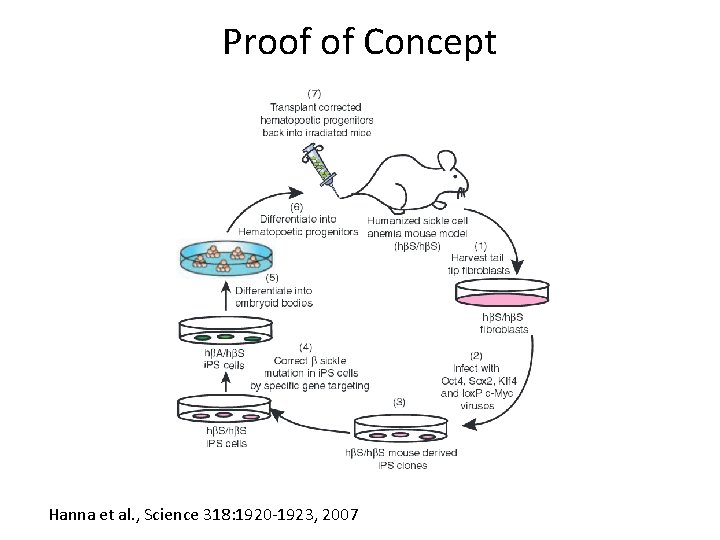
Proof of Concept Hanna et al. , Science 318: 1920 -1923, 2007
- Slides: 44