Status of Renal Denervation Studies for Indications Beyond

Status of Renal Denervation Studies for Indications Beyond Hypertension, CHF, CKD, Sleep Apnea Dr Justin E Davies Consultant Cardiologist Hammersmith Hospital Imperial College London CRT 2016

Adverse Effects of Systolic heart failure Cardiac output Sympathetic over activation
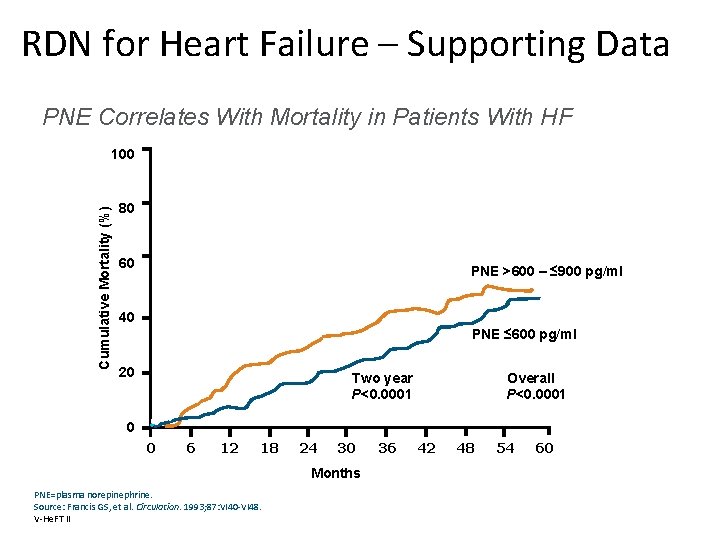
RDN for Heart Failure – Supporting Data PNE Correlates With Mortality in Patients With HF Cumulative Mortality (%) 100 80 PNE >900 pg/ml 60 PNE >600 – ≤ 900 pg/ml 40 PNE ≤ 600 pg/ml 20 Two year P<0. 0001 Overall P<0. 0001 0 0 6 12 18 24 30 Months PNE=plasma norepinephrine. Source: Francis GS, et al. Circulation. 1993; 87: VI 40 -VI 48. V-He. FT II 36 42 48 54 60
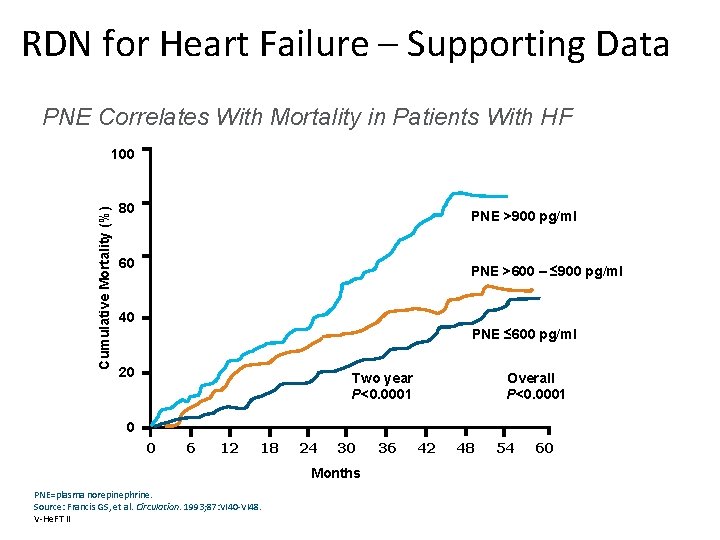
RDN for Heart Failure – Supporting Data PNE Correlates With Mortality in Patients With HF Cumulative Mortality (%) 100 80 PNE >900 pg/ml 60 PNE >600 – ≤ 900 pg/ml 40 PNE ≤ 600 pg/ml 20 Two year P<0. 0001 Overall P<0. 0001 0 0 6 12 18 24 30 Months PNE=plasma norepinephrine. Source: Francis GS, et al. Circulation. 1993; 87: VI 40 -VI 48. V-He. FT II 36 42 48 54 60
Adverse Effects of Systolic heart failure Arterial Vasoconstriction Cardiac output Sympathetic over activation Increased heart rate Fluid retention
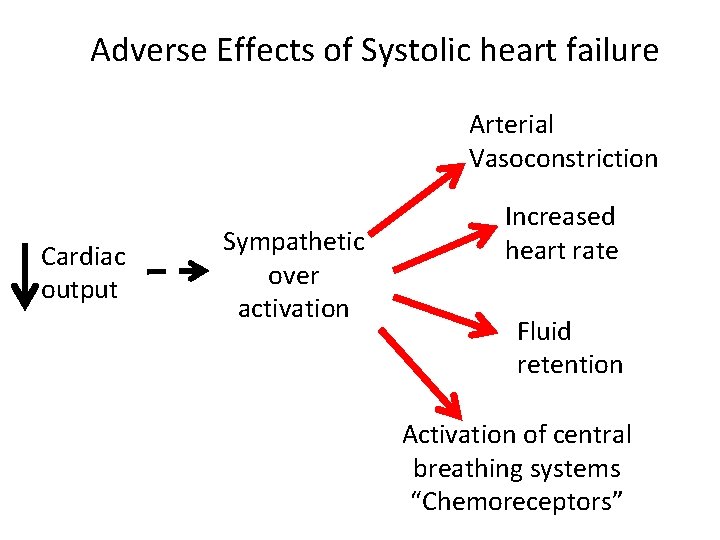
Adverse Effects of Systolic heart failure Arterial Vasoconstriction Cardiac output Sympathetic over activation Increased heart rate Fluid retention Activation of central breathing systems “Chemoreceptors”
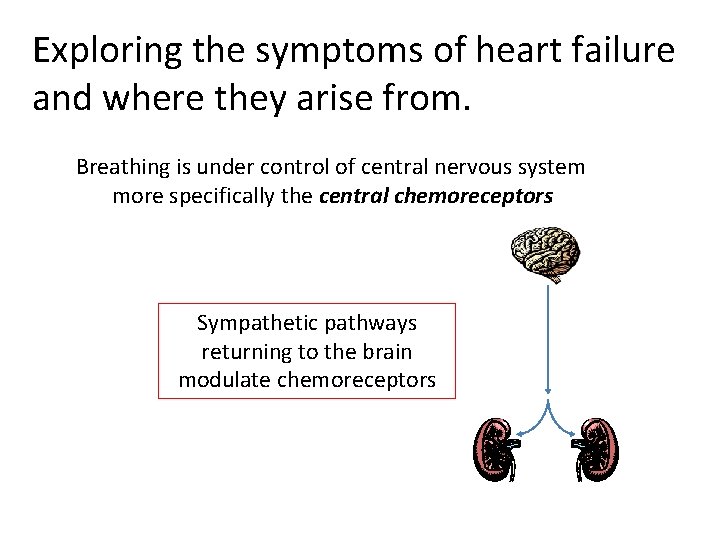
Exploring the symptoms of heart failure and where they arise from. Breathing is under control of central nervous system more specifically the central chemoreceptors Sympathetic pathways returning to the brain modulate chemoreceptors
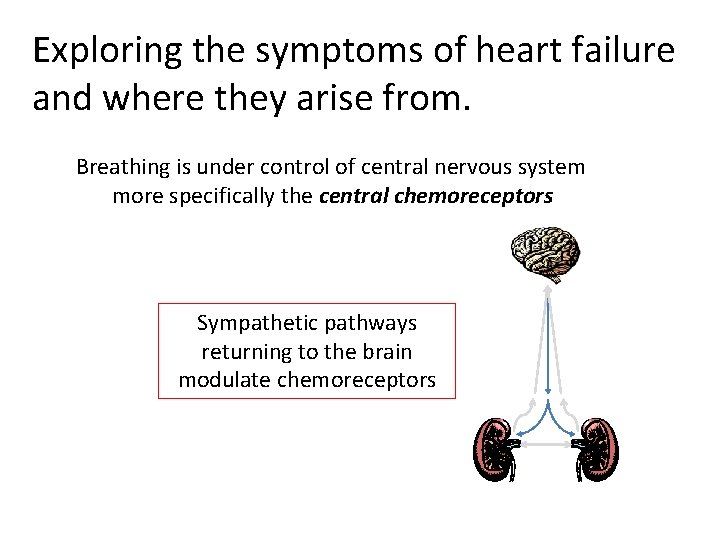
Exploring the symptoms of heart failure and where they arise from. Breathing is under control of central nervous system more specifically the central chemoreceptors Sympathetic pathways returning to the brain modulate chemoreceptors
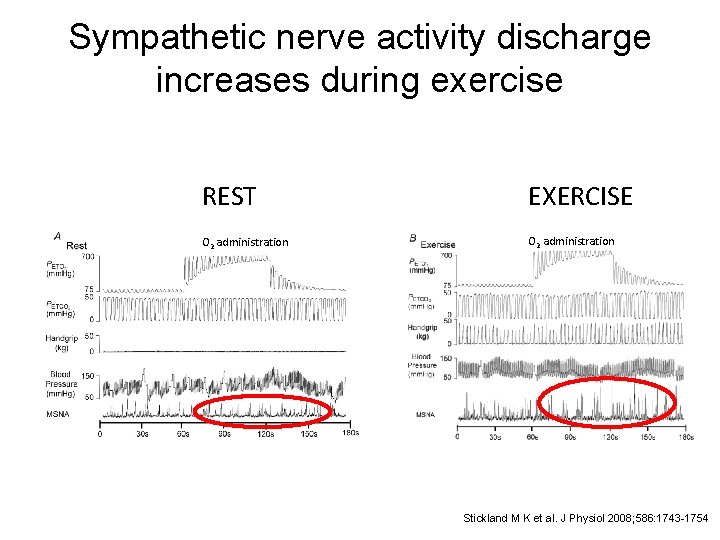
Sympathetic nerve activity discharge increases during exercise REST EXERCISE O 2 administration Stickland M K et al. J Physiol 2008; 586: 1743 -1754
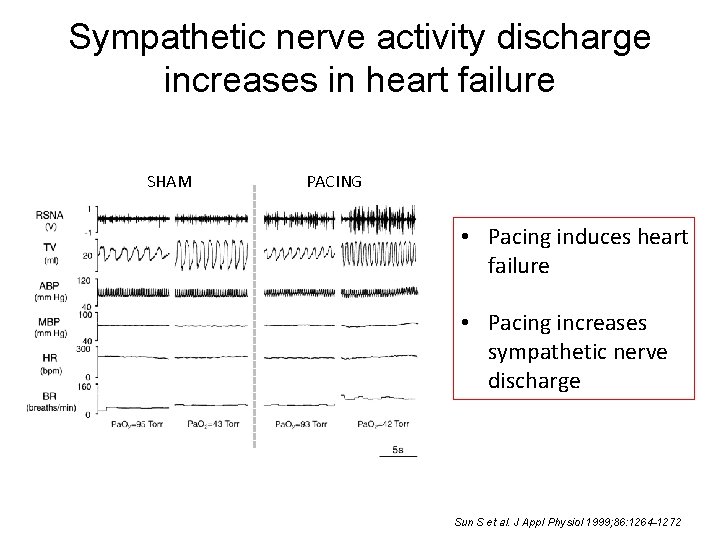
Sympathetic nerve activity discharge increases in heart failure SHAM PACING • Pacing induces heart failure • Pacing increases sympathetic nerve discharge Sun S et al. J Appl Physiol 1999; 86: 1264 -1272
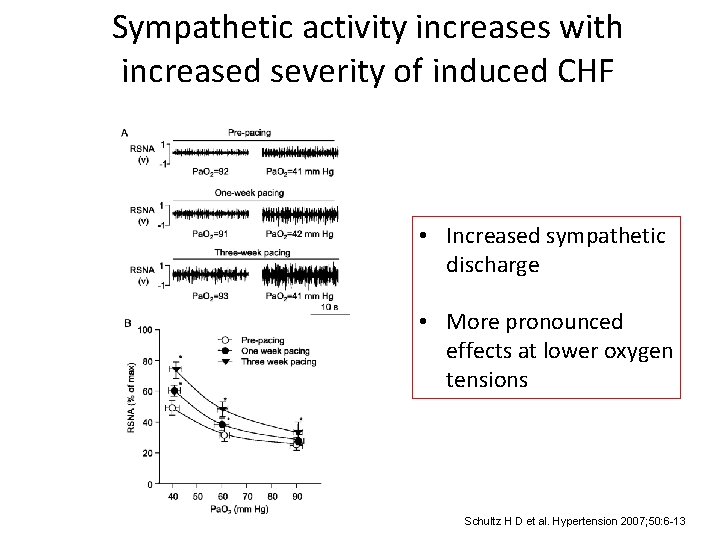
Sympathetic activity increases with increased severity of induced CHF • Increased sympathetic discharge • More pronounced effects at lower oxygen tensions Schultz H D et al. Hypertension 2007; 50: 6 -13
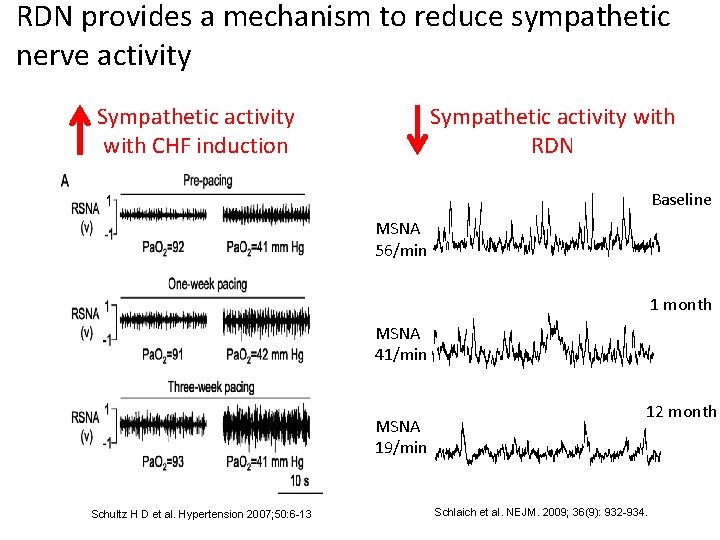
RDN provides a mechanism to reduce sympathetic nerve activity Sympathetic activity with CHF induction Sympathetic activity with RDN Baseline MSNA 56/min 1 month MSNA 41/min MSNA 19/min Schultz H D et al. Hypertension 2007; 50: 6 -13 12 month Schlaich et al. NEJM. 2009; 36(9): 932 -934.
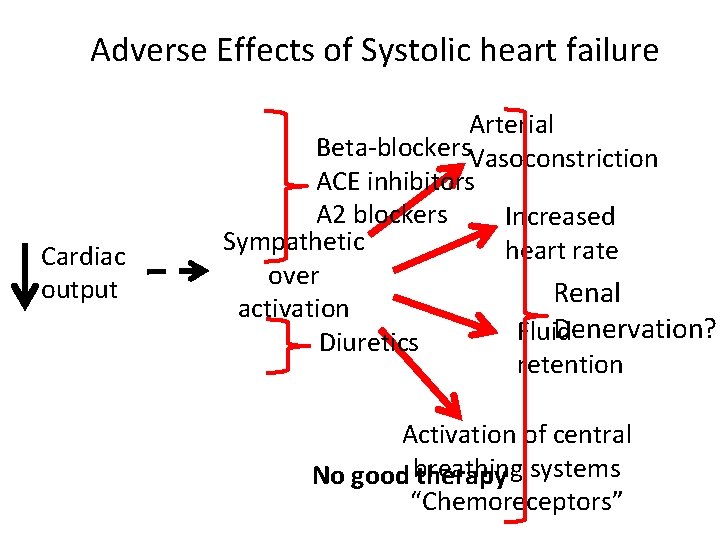
Adverse Effects of Systolic heart failure Cardiac output Arterial Beta-blockers. Vasoconstriction ACE inhibitors A 2 blockers Increased Sympathetic heart rate over Renal activation Denervation? Fluid Diuretics retention Activation of central No good breathing systems therapy “Chemoreceptors”

REACH-Pilot 1 Design • 7 patient safety study • Chronic mod-severe heart failure • Maximal tolerated medical therapy • In-patient follow-up 5 day admission • Bilateral renal denervation Aims • Safety of renal denervation • Follow-up for 6 months Results • No procedural complications • No acute/medium term complication • Improvement in 6 min walk REACH-Pilot, Davies JE, Francis DP et al. Int J Cardiol. 2013 Jan 20; 162(3): 189 -92
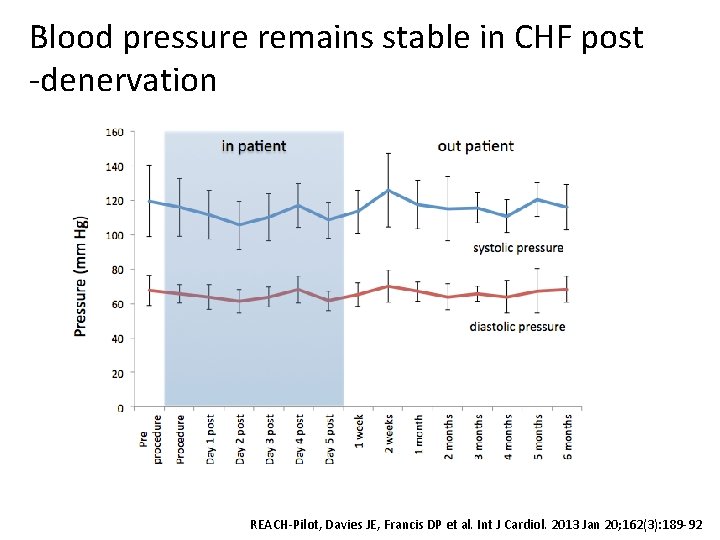
Blood pressure remains stable in CHF post -denervation REACH-Pilot, Davies JE, Francis DP et al. Int J Cardiol. 2013 Jan 20; 162(3): 189 -92
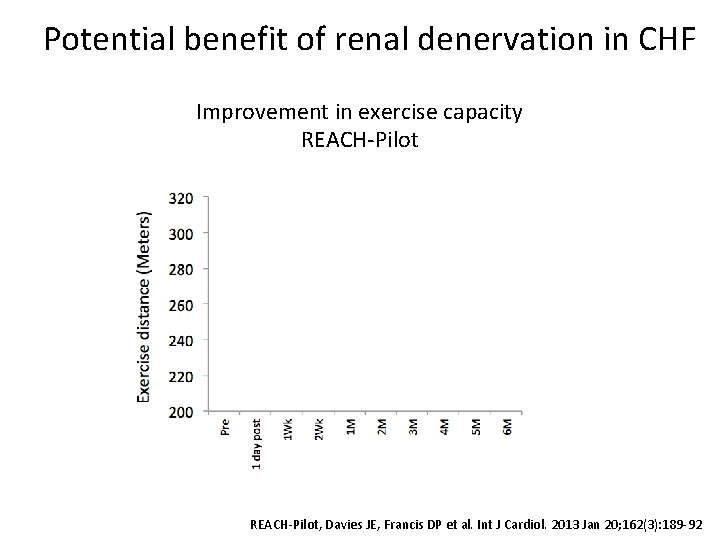
Potential benefit of renal denervation in CHF Improvement in exercise capacity REACH-Pilot Δ 27. 1 m p=0. 03 REACH-Pilot, Davies JE, Francis DP et al. Int J Cardiol. 2013 Jan 20; 162(3): 189 -92
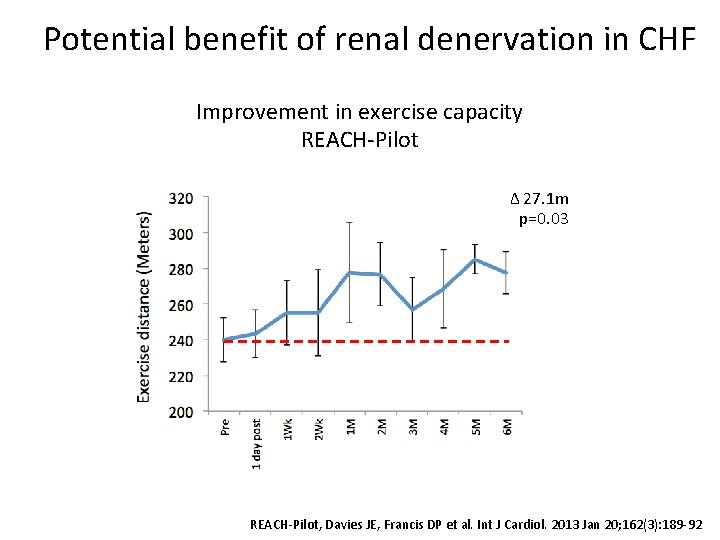
Potential benefit of renal denervation in CHF Improvement in exercise capacity REACH-Pilot Δ 27. 1 m p=0. 03 REACH-Pilot, Davies JE, Francis DP et al. Int J Cardiol. 2013 Jan 20; 162(3): 189 -92

Summary of drug changes Loop diuretics were reduced or stopped in 4/7 patients No patient had an increase in diuretic Beta-blockers decrease in 2 and increased in 1. A 2 blocker decreased in 2 and increased in 1

Conclusion from REACH • No acute, early or medium term adverse events • Potentially exciting trends • Small numbers

Caution Non-Blinded study Open label study Non-Randomised study Needs exploration in more highly powered randomised clincial studies and in more severe CHF
N=20 10 in RDN arm 10 in medical therapy arm EP ablation catheters

No Change in HR or BP No Change in BNP / EF SAFETY Improvement in symptoms

Caution Non-Blinded study Open label study Non-Randomised study Needs exploration in more highly powered randomised clincial studies and in more severe CHF

The SYMPLICITY HF Feasibility Study: Twelve Month Outcomes of Renal Denervation in Patients with Chronic Heart Failure and Renal Impairment Henry Krum, MD 1, Sidney A. Cohen, MD 2, Ingrid Hopper, MD 1, Bengt Rundqvist, MD 3, Uta Hoppe, MD 4, Tom Marwick, MD 5, Clemens Steinwender, MD 6, Sharad Shetty, MD 7, Christopher Hayward, MD 8 and Eduardo Gronda, MD 9 1 Monash University and the Alfred Hospital, Melbourne, Australia; 2 Medtronic, Santa Rosa, CA, and the Hospital of the University of Pennsylvania, [city], PA; 3 Sahlgrenska Universitetssjukhuset, Gothenburg, Sweden; 4 Landeskrankenhaus Salzburg-Universitätsklinikum, Salzburg, Austria; 5 Menzies Research Institute Tasmania, Hobart, Australia; 6 Allgemeines Krankenhaus der Stadt Linz, Austria; 7 Royal Perth Hospital, Perth, Australia; 8 Saint Vincent's Hospital, Sydney, Australia; 9 IRCCS Multi. Medica, Milan, Italy.

Study Design The SYMPLICITY HF Study is a prospective, multi-center, nonrandomized, single arm, open-label research study • Patient Population: – 39 Patients with chronic systolic heart failure (LVEF <40%, NYHA Class II-III) and renal impairment (e. GFR < 60 ml/min/1. 73 m 2), who were on a stable medical therapy were enrolled • Intervention: – Renal nerve denervation using the Symplicity Flex catheter – Experienced RDN interventionalists from 8 sites in Europe and Australia

Study Design Safety Evidence of Renal Denervation by change from baseline to 6 and 12 months in: a) Renal noradrenaline spillover (up to 10 subjects Alfred Hospital, Australia) b) MSNA (up to 10 subjects Alfred Hospital, Australia) Evidence of a Physiologic Effect by change from baseline to 12 months in: a) left ventricular mass, volumes and function, as documented by echocardiography b) e. GFR c) total body volume status d) 24 hour urine Na, Cr and protein e) plasma BNP/NT and pro-BNP f) neurohormones g) 6 MHW h) QT intervals on ECG i) Glycemic status (fasting C peptide, Hb. A 1 c, blood glucose, GTT) j) Electrophysiologic parameters
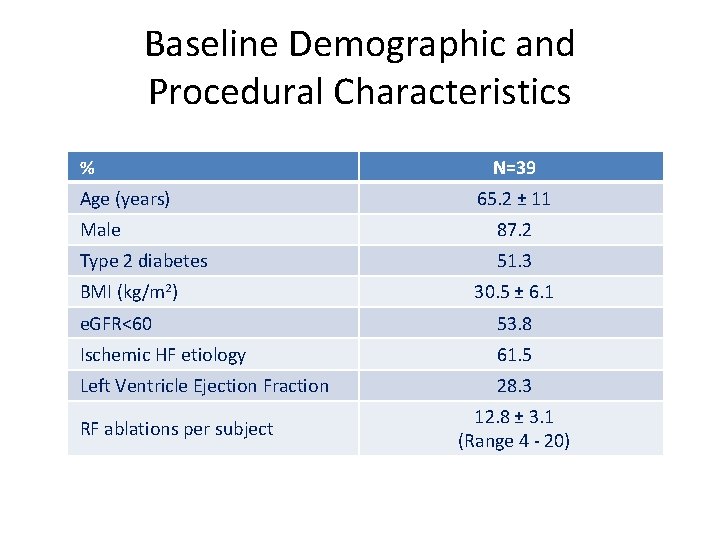
Baseline Demographic and Procedural Characteristics % Age (years) N=39 65. 2 ± 11 Male 87. 2 Type 2 diabetes 51. 3 BMI (kg/m 2) 30. 5 ± 6. 1 e. GFR<60 53. 8 Ischemic HF etiology 61. 5 Left Ventricle Ejection Fraction 28. 3 RF ablations per subject 12. 8 ± 3. 1 (Range 4 - 20)
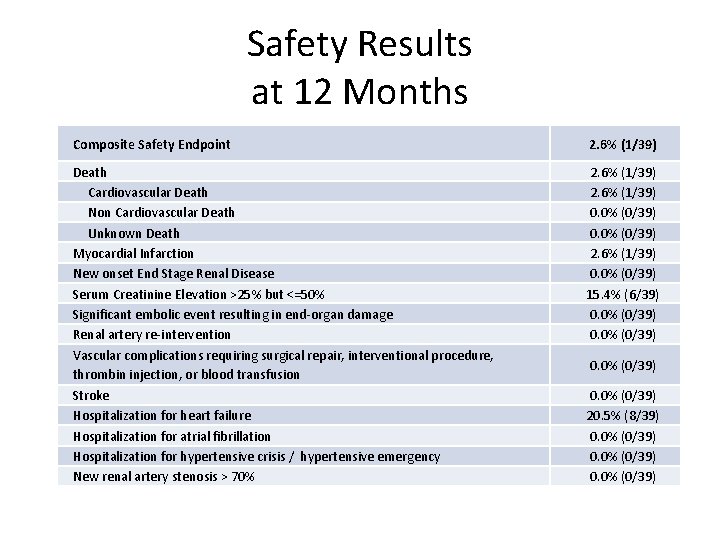
Safety Results at 12 Months Composite Safety Endpoint 2. 6% (1/39) Death Cardiovascular Death Non Cardiovascular Death Unknown Death Myocardial Infarction New onset End Stage Renal Disease Serum Creatinine Elevation >25% but <=50% Significant embolic event resulting in end-organ damage Renal artery re-intervention Vascular complications requiring surgical repair, interventional procedure, thrombin injection, or blood transfusion Stroke Hospitalization for heart failure Hospitalization for atrial fibrillation Hospitalization for hypertensive crisis / hypertensive emergency New renal artery stenosis > 70% 2. 6% (1/39) 0. 0% (0/39) 15. 4% (6/39) 0. 0% (0/39) 20. 5% (8/39) 0. 0% (0/39)
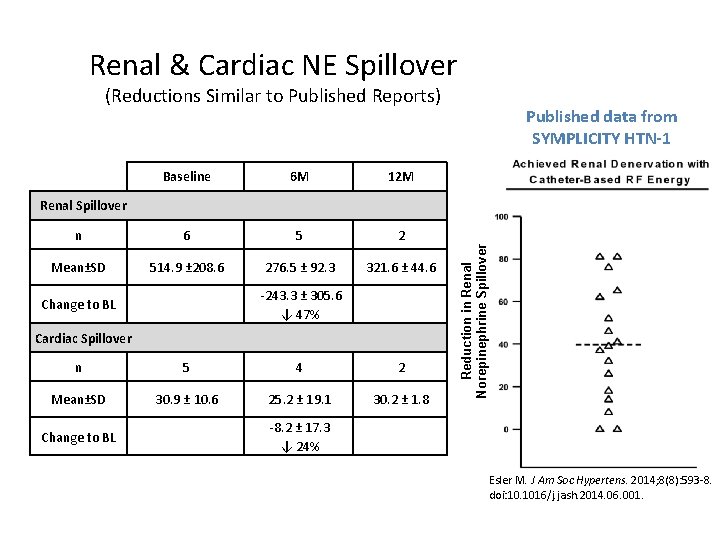
Renal & Cardiac NE Spillover (Reductions Similar to Published Reports) Baseline 6 M 12 M n 6 5 2 Mean±SD 514. 9 ± 208. 6 276. 5 ± 92. 3 321. 6 ± 44. 6 Published data from SYMPLICITY HTN-1 -243. 3 ± 305. 6 ↓ 47% Change to BL Cardiac Spillover n 5 4 2 Mean±SD 30. 9 ± 10. 6 25. 2 ± 19. 1 30. 2 ± 1. 8 Change to BL Reduction in Renal Norepinephrine Spillover Renal Spillover -8. 2 ± 17. 3 ↓ 24% Esler M. J Am Soc Hypertens. 2014; 8(8): 593 -8. doi: 10. 1016/j. jash. 2014. 06. 001.
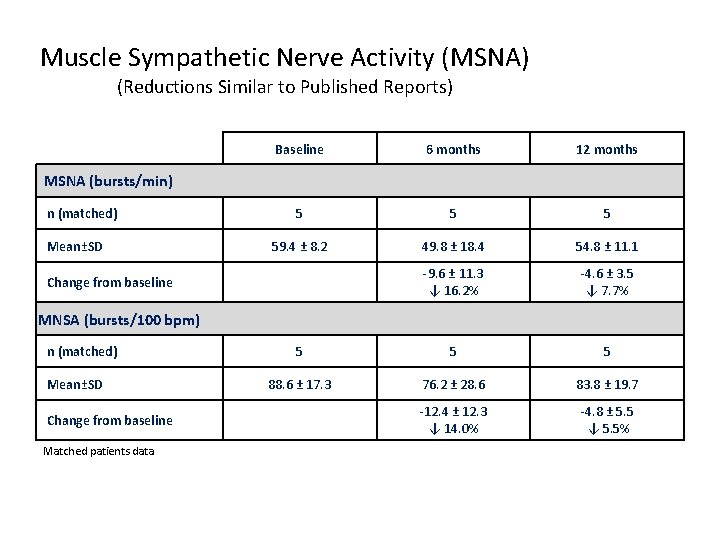
Muscle Sympathetic Nerve Activity (MSNA) (Reductions Similar to Published Reports) Baseline 6 months 12 months 5 59. 4 ± 8. 2 49. 8 ± 18. 4 54. 8 ± 11. 1 -9. 6 ± 11. 3 ↓ 16. 2% -4. 6 ± 3. 5 ↓ 7. 7% 5 5 5 88. 6 ± 17. 3 76. 2 ± 28. 6 83. 8 ± 19. 7 -12. 4 ± 12. 3 ↓ 14. 0% -4. 8 ± 5. 5 ↓ 5. 5% MSNA (bursts/min) n (matched) Mean±SD Change from baseline MNSA (bursts/100 bpm) n (matched) Mean±SD Change from baseline Matched patients data
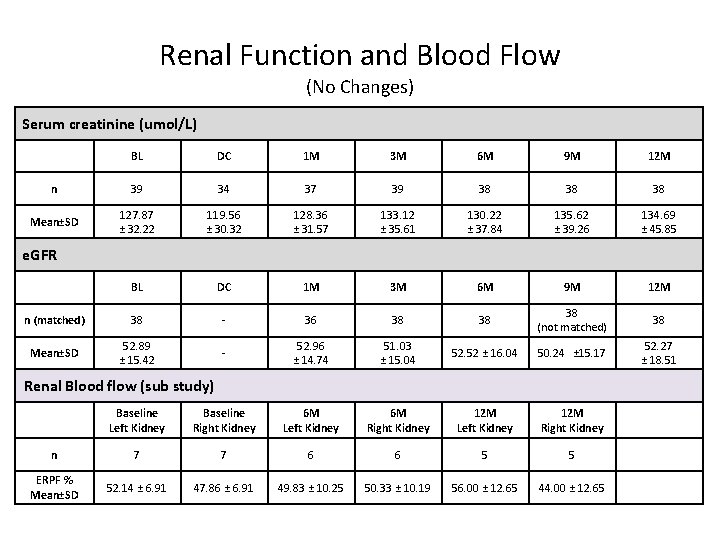
Renal Function and Blood Flow (No Changes) Serum creatinine (umol/L) BL DC 1 M 3 M 6 M 9 M 12 M n 39 34 37 39 38 38 38 Mean±SD 127. 87 ± 32. 22 119. 56 ± 30. 32 128. 36 ± 31. 57 133. 12 ± 35. 61 130. 22 ± 37. 84 135. 62 ± 39. 26 134. 69 ± 45. 85 BL DC 1 M 3 M 6 M 9 M 12 M n (matched) 38 - 36 38 38 38 (not matched) 38 Mean±SD 52. 89 ± 15. 42 - 52. 96 ± 14. 74 51. 03 ± 15. 04 52. 52 ± 16. 04 50. 24 ± 15. 17 52. 27 ± 18. 51 Baseline Left Kidney Baseline Right Kidney 6 M Left Kidney 6 M Right Kidney 12 M Left Kidney 12 M Right Kidney n 7 7 6 6 5 5 ERPF % Mean±SD 52. 14 ± 6. 91 47. 86 ± 6. 91 49. 83 ± 10. 25 50. 33 ± 10. 19 56. 00 ± 12. 65 44. 00 ± 12. 65 e. GFR Renal Blood flow (sub study)
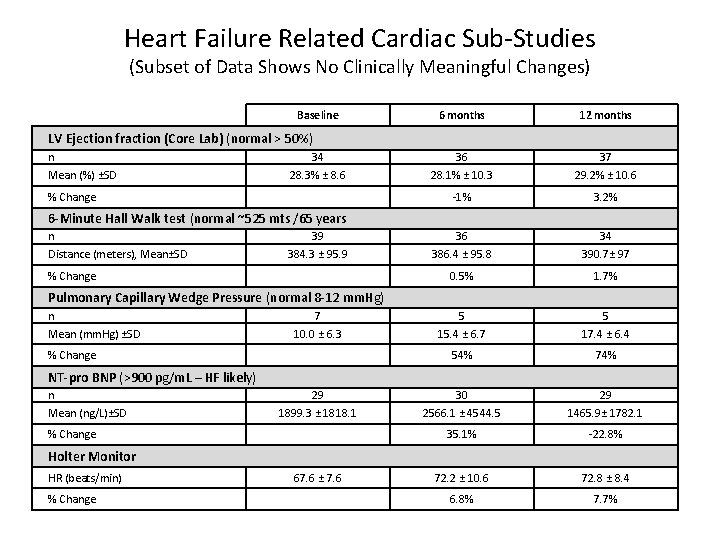
Heart Failure Related Cardiac Sub-Studies (Subset of Data Shows No Clinically Meaningful Changes) Baseline 6 months 12 months 36 28. 1% ± 10. 3 37 29. 2% ± 10. 6 -1% 3. 2% 36 386. 4 ± 95. 8 34 390. 7± 97 0. 5% 1. 7% 7 5 5 10. 0 ± 6. 3 15. 4 ± 6. 7 17. 4 ± 6. 4 54% 74% 30 2566. 1 ± 4544. 5 29 1465. 9± 1782. 1 35. 1% -22. 8% 72. 2 ± 10. 6 72. 8 ± 8. 4 6. 8% 7. 7% LV Ejection fraction (Core Lab) (normal > 50%) n Mean (%) ±SD 34 28. 3% ± 8. 6 % Change 6 -Minute Hall Walk test (normal ~525 mts /65 years n Distance (meters), Mean±SD 39 384. 3 ± 95. 9 % Change Pulmonary Capillary Wedge Pressure (normal 8 -12 mm. Hg) n Mean (mm. Hg) ±SD % Change NT-pro BNP (>900 pg/m. L – HF likely) n Mean (ng/L)±SD 29 1899. 3 ± 1818. 1 % Change Holter Monitor HR (beats/min) % Change 67. 6 ± 7. 6

Caution Non-Blinded study Open label study Non-Randomised study Needs exploration in more highly powered randomised clincial studies and in more severe CHF

Ongoing studies in heart failure REACH Upto 100 patient Sham (3: 2 randomisation) Double blinded NYHA III, EF<40% Bilateral renal denervation in severe CHF >50 with Spyral Primary endpoint: Symptom improvement Reporting 2017

Summary • Mechanistic potential to expand denervation outside of resistant hypertension • Expand neuro-hormonal blockade offered by pharmacological therapies • Potential advantages to disruption of the overactivated afferent sympathetic system • Exciting field - many unanswered questions

Summary • Mechanistic potential to expand denervation outside of resistant hypertension • Expand neuro-hormonal blockade offered by pharmacological therapies • Potential advantages to disruption of the overactivated afferent sympathetic system • Exciting field - many unanswered questions

Summary • Mechanistic potential to expand denervation outside of resistant hypertension • Expand neuro-hormonal blockade offered by pharmacological therapies • Potential advantages to disruption of the overactivated afferent sympathetic system • Exciting field - many unanswered questions

Summary • Mechanistic potential to expand denervation outside of resistant hypertension • Expand neuro-hormonal blockade offered by pharmacological therapies • Potential advantages to disruption of the overactivated afferent sympathetic system • Exciting field - many unanswered questions

Summary • Mechanistic potential to expand denervation outside of resistant hypertension • Expand neuro-hormonal blockade offered by pharmacological therapies • Potential advantages to disruption of the overactivated afferent sympathetic system • Exciting field - many unanswered questions
- Slides: 39