Statistics for Analytical Chemistry Reading lots to revise
































































- Slides: 72

Statistics for Analytical Chemistry

Reading –lots to revise and learn § § Chapter 3 Chapter 4 Chapter 5 -1 and 5 -2 Chapter 5 -3 will be necessary background for the AA lab § Chapter 5 -4 we will use later

Data Analysis § Most data quantitative - derived from measurements § Never really know error § With more measurements you get a better idea what it might be § Don’t spend a lot of time on an answer -where only 20% accuracy is required -or where sampling error is big although you don’t want to make the error worse

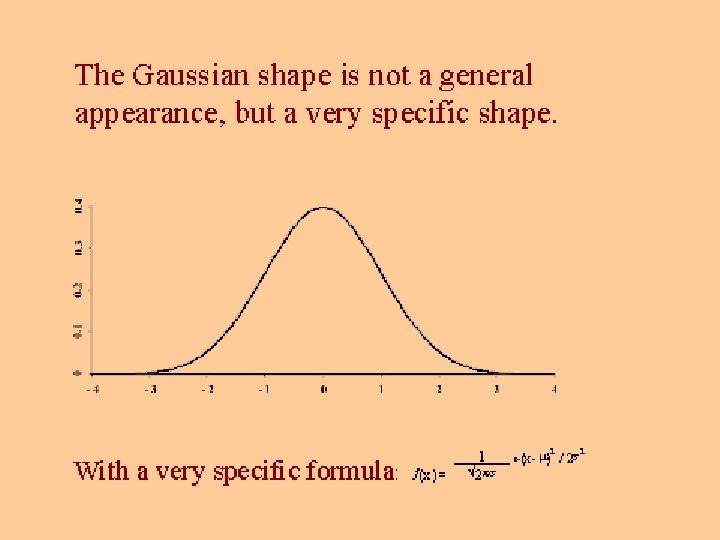
Significant Figure Convention § Final answer should only contain figures that are certain, plus the first uncertain number § eg 45. 2% § error less than 1% or we would only write 45% § error larger than 0. 05% or would write 45. 23%

Remember § § Leading zeros are not significant Trailing zeros are significant 0. 06037 - 4 significant figures 0. 060370 - 5 significant figures § 1200 ? ? § 12 x 102 - 2 significant figures

Rounding Off § Round a 5 to nearest even number § 4. 55 to 4. 6 § Carry an extra figure all through calculations § BUT NOT 6 EXTRA § Just round off at the end

Adding § Absolute uncertainty of answer must not exceed that of most uncertain number § Simple rule: Decimal places in answer = decimal places in number with fewest places 12. 2 00. 365 01. 04 13. 605 goes to 13. 6

When errors are known § R r =(A a) + (B b) + (C c) § where r 2 = a 2 + b 2 + c 2 § Example: Calculate the error in the MW of Fe. S from the following atomic weights: § Fe: 55. 847 0. 004 S: 32. 064 0. 003 § r = (0. 0042 + 0. 0032)1/2 § MW = 87. 911 0. 005

Multiplication and Division § Simplest rule: Sig figs in answer = smallest number of sig figs in any value used § This can lead to problems - particularly if the first digit of the number is 9. § 1. 07400 x 0. 993 = 1. 07 § 1. 07400 x 1. 002 = 1. 076 § Error is ~ 1/1000 therefore 4 significant figs in answer

Multiplication and Division § The relative uncertainty of the answer must fall between 0. 2 and 2. 0 times the largest relative uncertainty in the data used in the calculation. § Unless otherwise specified, the absolute uncertainty in an experimental measurement is taken to be +/- the last digit

Multiplication and Division § With known errors - add squares of relative uncertainties § r/R = [(a/A)2 + (b/B)2 +(c/C)2]1/2

Logs § Only figures in the mantissa (after the decimal point) are significant figures § Use as many places in mantissa as there are significant figures in the corresponding number § p. H = 2. 45 has 2 sig figs

Definitions § Arithmetic mean, (average) § Median -middle value § for N=even number, use average of central pair

Accuracy § Deviation from true answer § Difficult to know § Best way is to use Reference standards § National Bureau of Standards § Traceable Standards

Precision § Describes reproducibility of results § What is used to calculate the confidence limit § Can use deviation from mean § or relative deviation § 0. 1/5 x 1000 = 20 ppt (parts per thousand) § 0. 1/5 x 100% = 2%

Precision of Analytical Methods § § § Absolute standard deviation s or sd Relative standard deviation (RSD) Standard deviation of the mean sm Sm = s/N½ Coefficient of variation (CV) s/ x x 100% Variance s 2

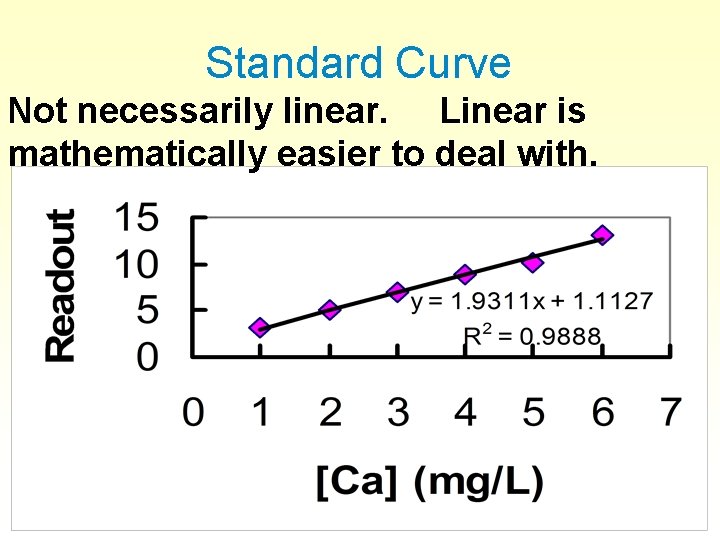
Standard Curve Not necessarily linear. Linear is mathematically easier to deal with.

Correlation coefficients § § § Show good a fit you have. R or R 2 For perfect correlation, R = 1, R 2 = 1

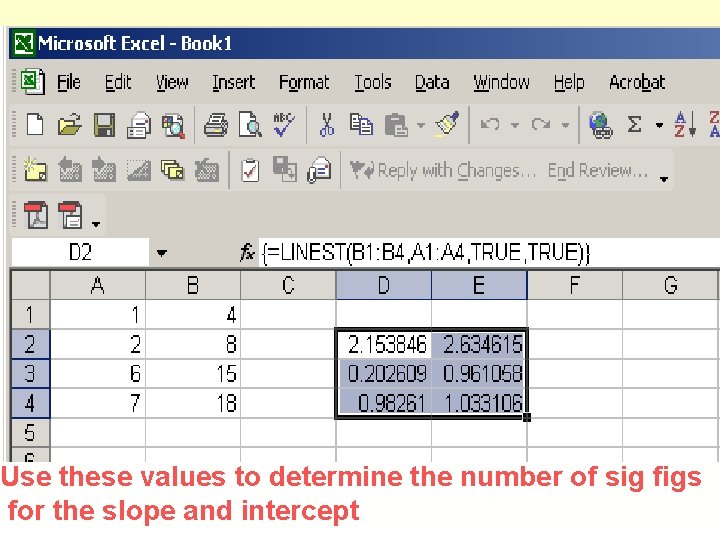
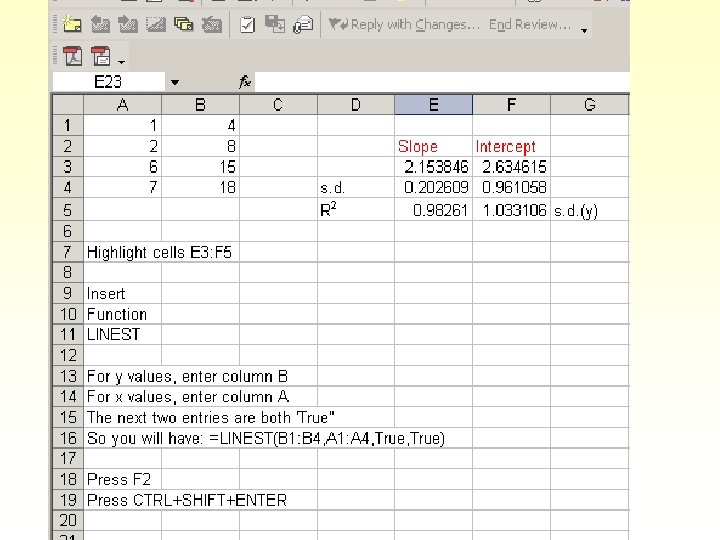
LINEST § Calculates slope and intercept § Calculates the uncertainty in the slope and the intercept § Calculates R 2 § Calculates s. d. of the population of y values § See page pp 68 -72, Harris.

Use these values to determine the number of sig figs for the slope and intercept


Dealing with Random Errors

Indeterminate Error § Repeating a coarse measurement gives the same result § eg weighing 50 g object to nearest g - only error would be determinate - such as there being a fault in the balance § If same object was weighed to several decimal places -get random errors

How many eggs in a dozen? § How wide is your desk? § Will everyone get the same answer? § What does this depend on?

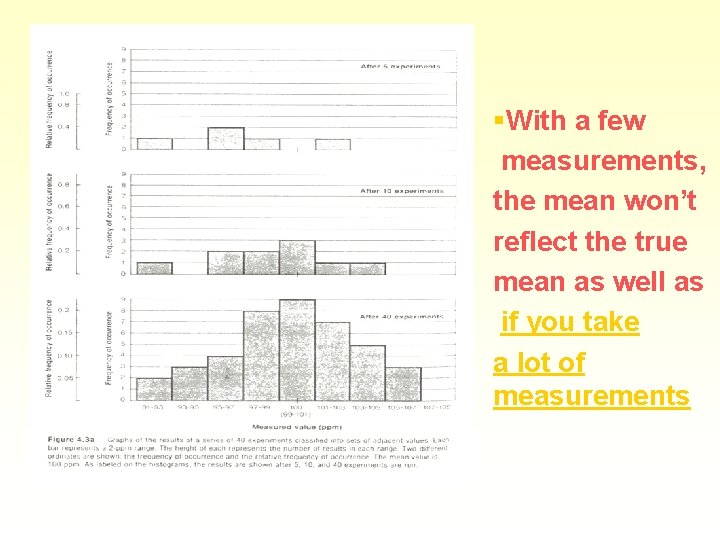
§With a few measurements, the mean won’t reflect the true mean as well as if you take a lot of measurements

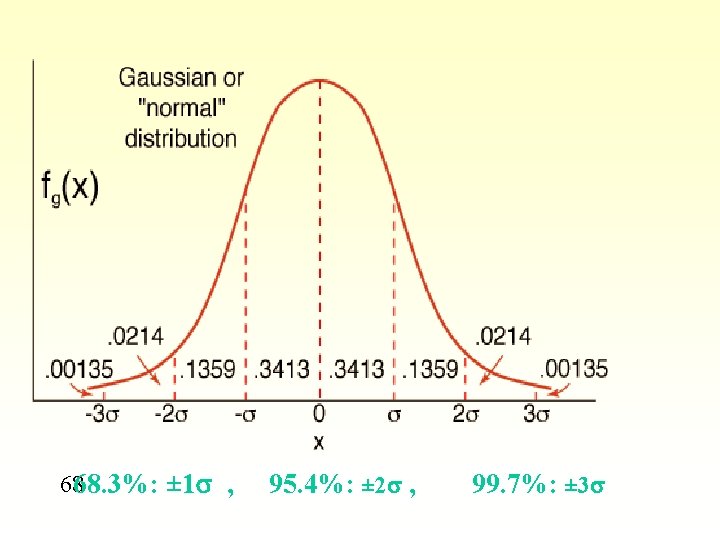
Random errors § With many measurements, more will be close to the mean § Various little errors add in different ways § Some cancel - sometimes will all be one way § A plot of frequency versus value gives a bell curve or Gaussian curve or normal error curve § Errors in a chemical analysis will fit this curve



Equation for Gaussian Curve


If z is abscissa (x axis) § Same curve is always obtained as z expresses the deviation from the mean in units of standard deviation

Statistics § Statistics apply to an infinite number of results § Often we only do an analysis 2 or 3 times and want to use the results to estimate the mean and the precision


Standard deviation § § § 68. 3% of area is within ± 1 of mean 95. 5% of area is within ± 2 of mean 99. 7% of area is within ± 3 of mean § For any analysis, chances are 95. 5 in 100 that error is ± 2 § Can say answer is within ± 2 with 95. 5% confidence

For a large data set § Get a good estimate of the mean, § § § Know this formula -but use a calculator 2 = variance Useful because additive

Small set of data § § § Average ( x ) An extra uncertainty The standard deviation calculated will differ for each small set of data used It will be smaller than the value calculated over the larger set Could call that a negative bias

s § For use N in denominator § For s use N-1 in denominator (we have one less degree of freedom - don’t know ) § At end, round s to 2 sig figs or less if there are not enough sig figs in data

Confidence Interval § We are doing an analysis to find the true mean - it is unknown § What we measure is x but it may not be the same as § Set a confidence limit eg 4. 5 ± 0. 3 g § The mean of the measurements was 4. 5 g § The true mean is in the interval 4. 2 -4. 8 with some specified degree of confidence

Confidence limit § A measure of the reliability (Re) § The reliability of a mean ( x ) increases as more measurements are taken § Re = k(n)1/2 § Reliability increases with square root of number of measurements § Quickly reach a condition of limiting return

Reliability § Would you want a car that is 95% reliable? § How often would that break down?



Confidence Interval § § § For 100 % confidence - need a huge interval Often use 95 % The confidence level chosen can change with the reason for the analysis

Confidence Interval when s ~ § µ ± xi = 1. 96 for 95 % confidence § z = (xi - µ)/ =1. 96 § Appropriate z values are given as a table § This applies to a single measurement § The confidence limit decreases as (N)1/2 as more measurements are taken

Confidence Interval § In the lab this year I will make you go home before you can get enough data for s to = § Therefore we will have to do a different kind of calculation to estimate the precision.

Student’s t-test The Student's t-Test was formulated by W. Gossett in the early 1900's. His employer (brewery) had regulations concerning trade secrets that prevented him from publishing his discovery, but in light of the importance of the t distribution, Gossett was allowed to publish under the pseudonym "Student". The t-Test is typically used to compare the means of two populations

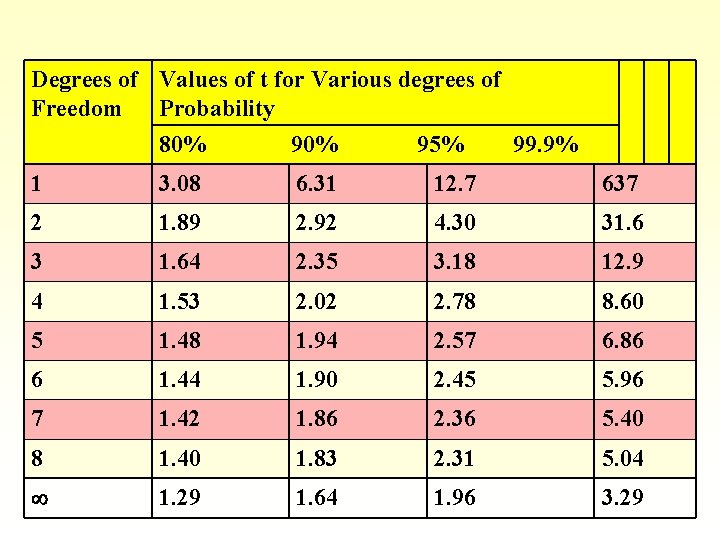
t-test § t depends on desired confidence limit § degrees of freedom (N-1)

Degrees of Values of t for Various degrees of Freedom Probability 80% 95% 99. 9% 1 3. 08 6. 31 12. 7 637 2 1. 89 2. 92 4. 30 31. 6 3 1. 64 2. 35 3. 18 12. 9 4 1. 53 2. 02 2. 78 8. 60 5 1. 48 1. 94 2. 57 6. 86 6 1. 44 1. 90 2. 45 5. 96 7 1. 42 1. 86 2. 36 5. 40 8 1. 40 1. 83 2. 31 5. 04 1. 29 1. 64 1. 96 3. 29

For practical purposes § Assume = s if you have made 20 measurements § Sometimes can be evaluated for a particular technique rather than for each sample § Usually too time consuming to do 20 replicate measurements on each sample

CONFIDENCE

Example § Cal Culator obtained the following results for replicate determinations of calcium in limestone § 14. 35%, 14. 41%, 14. 40%, 14. 32%, 14. 37% § each is xi § Calculate the confidence interval

Answer § § § § Average = 14. 37 % S = 0. 037% Choose a 95 % confidence limit Degrees of freedom = N-1 = 5 -1 =4 From t-table, t = 2. 78 14. 37% ± ts/N½ 14. 37 % ± 2. 78 x 0. 037% / 5 ½ 14. 37 ± 0. 05 %

Significant figures § I say: Use two or less significant figures in a confidence limit. Then use the same number of decimal places in both (guided by the CL) § When less than two sig figs in the CL? § When using two would require you to have more decimal places than were in the actual data.

The bunny gave up

Pooled standard deviation

Comparison of Means § We analyze several samples and want to know if they are the same or different § For each sample we take several measurements and obtain a mean

Comparing two means

Comparing two means

Example § Two barrels of wine were analyzed for their alcohol content to determine whether or not they were from different sources: § 12. 61% (6 analyses), § 12. 53% (4 analyses) § Pooled standard deviation = 0. 07 %

§ § Degrees of freedom = 6+4 -2=8 t at 95% CL for 8 deg of freedom =2. 3 tcalc < ttable therefore difference is not significant at the 95% CL – the two samples are the same at the 95% CL

Rejection of data- Q Test § Qexp= questionable value-nearest numerical value § § range Look up Table of Qcritical If Qexp < Qcritical, keep the point If more observations are taken it is easier to determine if a point is an outlier

Calibration Sensitivity § The slope of the calibration curve at the concentration of interest § Doesn’t take precision into account

Analytical Sensitivity § Slope/s. d. = m/s. d. § Where s = standard deviation of the signal § Analytical sensitivity is independent of gain, but can vary with the concentration as s can depend on concentration

Limit of detection § The minimum concentration detectable at a known confidence level § Is the concentration corresponding to the lowest usable reading (LUR) § LUR = average blank + k s. d. blank § k determines the confidence level § We use k = 3 for a 95% C. L. § Do not confuse LOD and LUR

Harris page 103 § LUR corresponds to Signal detection limit § LOD corresponds to Concentration detection limit § When doing this in lab WE CHEAT § We should have 20 measurements of the blank and we never do because of time constraints. To publish a result or for a paying client, we would need 20.


§ Ideally, the average blank = b (the intercept) § However, if b > average blank, then recalculate LUR using LUR = b + k s. d. blank § Usually say LUR = b + 3 sd § LOD = 5. 2 mg/L (k = 3) § Note the 2 significant figures

Quality Assurance § § Begins with sampling Calibration Check Run standards every few samples. Reference standards are of known concentration. Do you get the right answer? § Include in Table of Results. § SOP’s are very important

SOP (Standard operating procedure) § Set of written instructions that document a routine or repetitive activity which is followed by employees in an organization. § The development and use of SOPs is an integral part of a successful quality system. § Provides information to perform a job properly and consistently in order to achieve pre-determined specifications and quality. § http: //people. stfx. ca/tsmithpa/Chem 361/

Numerical Criteria for Selecting Analytical Methods § § § Precision Bias Sensitivity Detection Limit Concentration Range Selectivity

Other characteristics to be considered § § § Speed Ease and convenience Skill required of operator Cost and availibility of equipment Per-sample-cost

Criterion Figure of Merit Precision Absolute sd, relative sd, coefficient of variation, variance Bias Absolute systematic error, relative systematic error Sensitivity Calibration sensitivity, analytical sensitivity Limit of detection Av. Blank + 3 sd blank Concentratio LOQ to LOL (limit of linearity) n range Selectivity Coefficient of selectivity