States of Matter Solids Solids are substances with

States of Matter

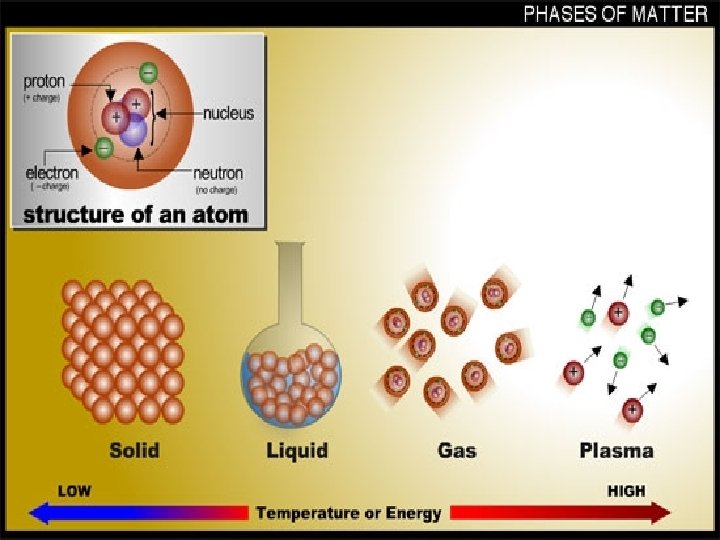
Solids • Solids are substances with densely packed particles, which may be ions, atoms, or molecules, depending upon the substance • The particles of a solid are arranged in a definite pattern, shape and volume • Most solids have a crystalline structure, in which particles are arranged in geometric patterns

Liquids • At any temp above absolute zero, atoms vibrate • These vibrations increase temp and are called thermal vibrations • At the melting point the vibrations become enough to break the forces holding the solid together • Liquids do not have their own shape, they take the shape of the container, but do have a specific volume

Gases • The particles in a liquid can vibrate enough to escape the liquid • This process that changes a liquid to gas is evaporation • The slow change of a state from a solid, ice crystals, to a gas, water vapor, without an intermediate is called sublimation • The particles in gas are separated by a large space and move around at extreme speeds • Gases have no definite shape or volume


Plasmas • When matter is heated to temps greater then 5000 degrees Celsius, the collisions between particles are so violent that electrons are knocked away from atoms • These conditions exist on stars – The gases of stars consist entirely of positive ions and free electrons • These hot, highly ionized, electrically conducting gases are called plasmas
- Slides: 7