STATES OF MATTER Self Study CHANGES IN STATE











![3. Dipole-Dipole [polar] - end attracted /// to + end of another 3. Dipole-Dipole [polar] - end attracted /// to + end of another](https://slidetodoc.com/presentation_image_h2/16d359c310a5ee3a1a2f8641eb3b6f5f/image-17.jpg)
![4. Dispersion [nonpolar] weakest interaction Induced + Induced - -a dipole is induced -lowest 4. Dispersion [nonpolar] weakest interaction Induced + Induced - -a dipole is induced -lowest](https://slidetodoc.com/presentation_image_h2/16d359c310a5ee3a1a2f8641eb3b6f5f/image-18.jpg)






















- Slides: 51

STATES OF MATTER Self Study

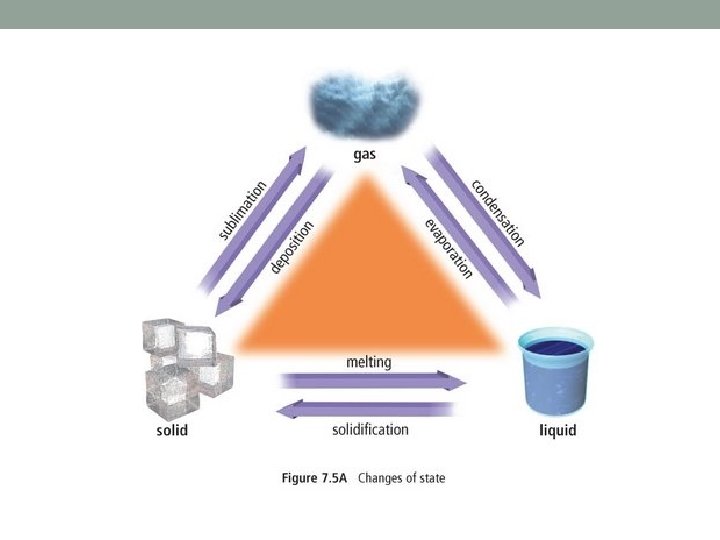
CHANGES IN STATE Four states of matter video clip: click here


PHASE DIAGRAM Book video clip: click here

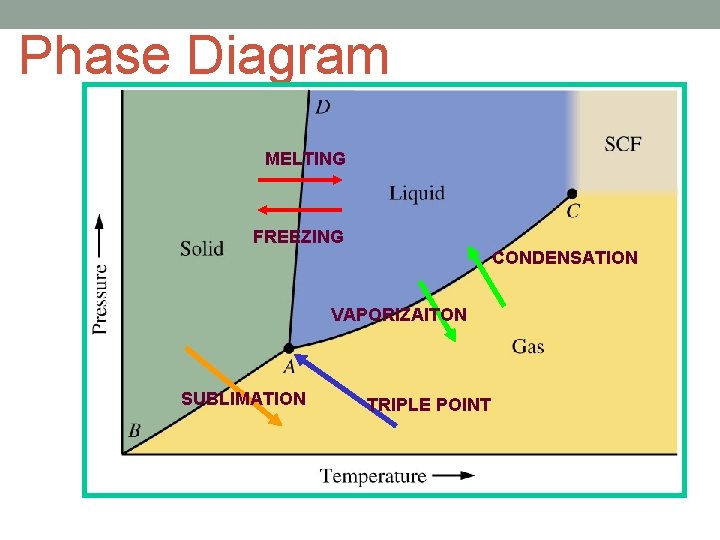
Phase Diagram MELTING FREEZING CONDENSATION VAPORIZAITON SUBLIMATION TRIPLE POINT

WHAT WORD DESCRIBES MOTION OR MOVEMENT ENERGY?

KINETIC ENERGY All matter’s particles are in constant motion

State of Matter is determined by • Particle arrangement • Energy of particles • Distance b/w particles

KINETIC THEORY: SOLIDS Atoms are vibrating on a fixed point in a solid

TYPES OF SOLIDS Book video clip: click here

Amorphous Solid Has no organized internal structure Rubber Plastic Glass Asphalt Cotton Candy

COMPOUNDS WITH THE HIGHEST MELTING POINTS ARE….

Melting Points Strongest ( highest): Lowest ( weakest) Ionic Bonding Hydrogen Bonding Dipole-Dipole Dispersion

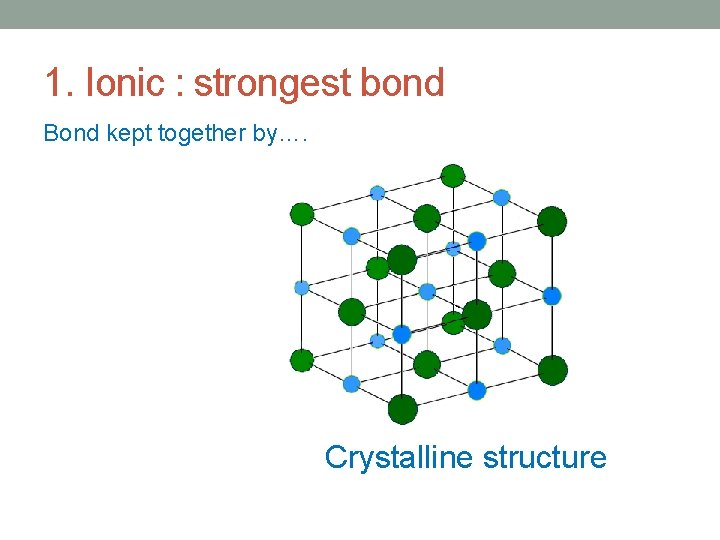
1. Ionic : strongest bond Bond kept together by…. Crystalline structure

Covalent Compounds have three interactions( intermolecular forces) that keep them together….

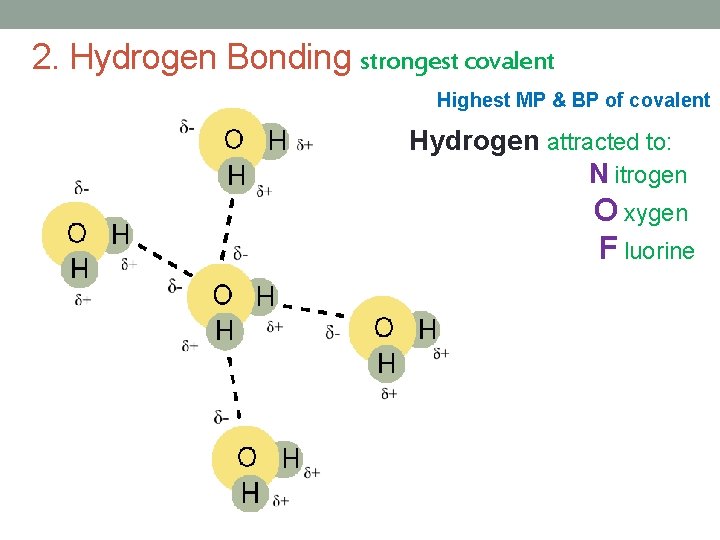
2. Hydrogen Bonding strongest covalent Highest MP & BP of covalent Hydrogen attracted to: N itrogen O xygen F luorine
![3 DipoleDipole polar end attracted to end of another 3. Dipole-Dipole [polar] - end attracted /// to + end of another](https://slidetodoc.com/presentation_image_h2/16d359c310a5ee3a1a2f8641eb3b6f5f/image-17.jpg)
3. Dipole-Dipole [polar] - end attracted /// to + end of another
![4 Dispersion nonpolar weakest interaction Induced Induced a dipole is induced lowest 4. Dispersion [nonpolar] weakest interaction Induced + Induced - -a dipole is induced -lowest](https://slidetodoc.com/presentation_image_h2/16d359c310a5ee3a1a2f8641eb3b6f5f/image-18.jpg)
4. Dispersion [nonpolar] weakest interaction Induced + Induced - -a dipole is induced -lowest MP & BP

KINETIC THEORY: LIQUIDS Particles are not bound together in fixed positions, but there is still some attractive forces keeping them close during movement.

Fluidity Particles flow, slide over each other

Viscosity

KINETIC THEORY OF GASES Book clip: clip here

Gas Particles Assumption #1: • Have a small volume • Far apart from each other • Independent : No attractive or repulsive forces

Gas Particles Assumption #2: • Move: • Rapid • Independent • Random • Straight paths • Only direction when rebounding

Gas Particles Assumption #3 When particles collide, there collisions are elastic: • no loss of energy • it is transferred between the particles

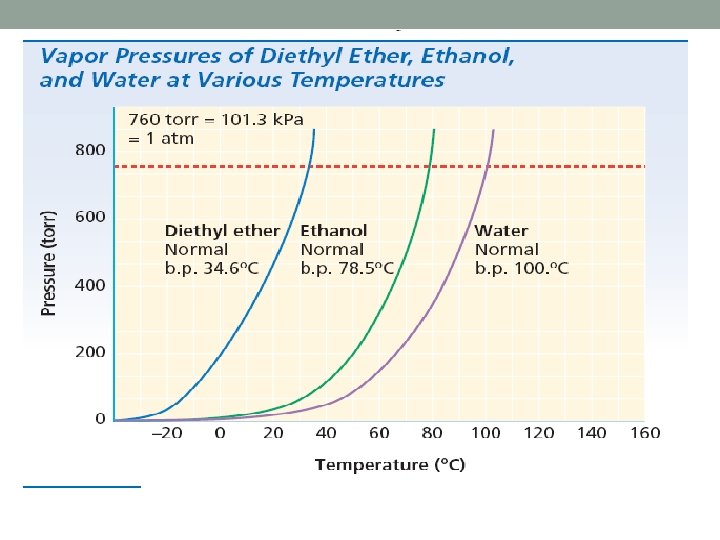
GAS PRESSURE Force exerted when gas particles collide: click here

Pressure Units • SI unit : Pascal (Pa) ▫ Sea level: 101. 3 k. Pa • Millimeters of Mercury: mm Hg ▫ blood pressure • Atmospheres: atm ▫ Atmospheric pressure • Torr • Pounds per Square Inch: PSI ▫ Tire pressure, scuba diving • Bar ▫ Atmospheric pressure

Conversions of Pressure 1 atm = 760 mm. Hg = 101. 3 k. Pa 1 torr = 1 mm Hg 1 atm = 14. 7 PSI 1 bar = 1 atm

EVAPORATION VS CONDENSATION Eureka video clip: click here

EVAPORATION Video Clip: Click here

EQUILIBRIUM Book clip: click here

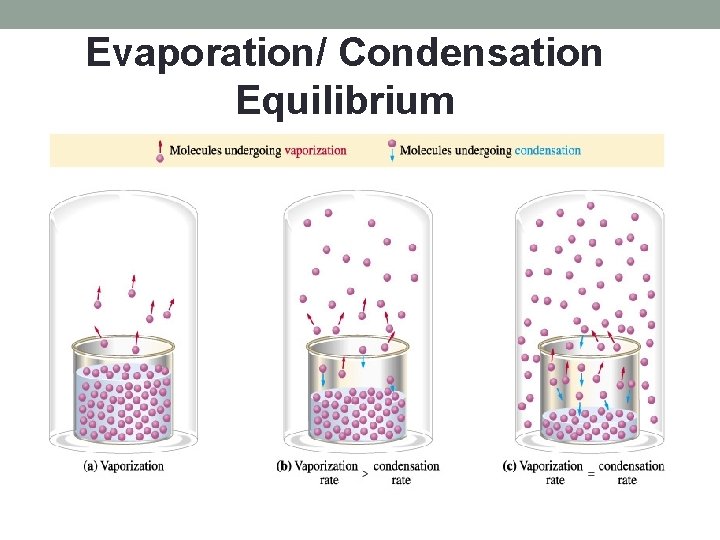
Evaporation/ Condensation Equilibrium

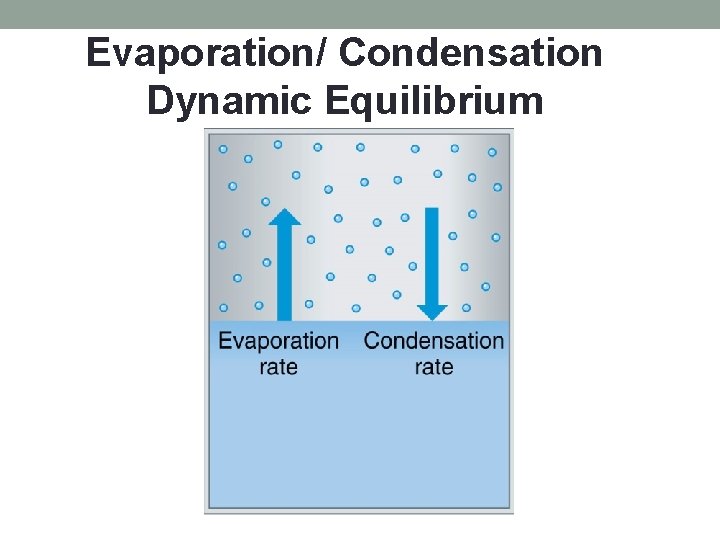
Evaporation/ Condensation Dynamic Equilibrium

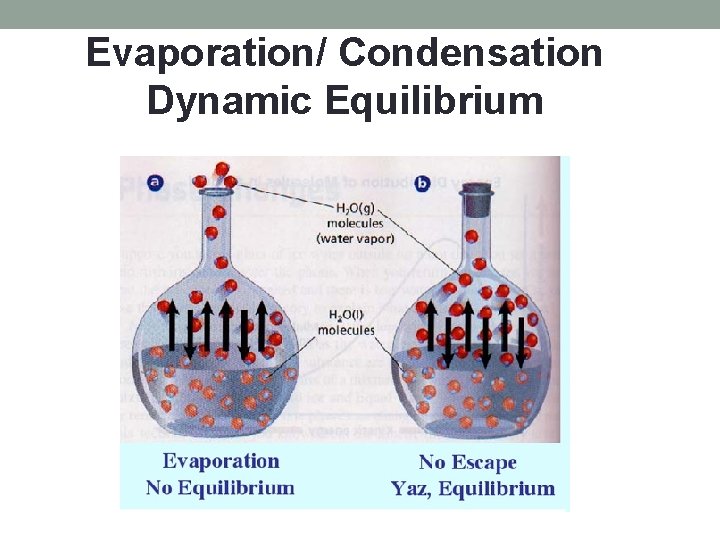
Evaporation/ Condensation Dynamic Equilibrium

BOILING Video clip: click here


TEMPERATURE Video clip: click here

MEASURING TEMPERATURE Eureka video clip: click here

TEMPERATURE Movement of particles

ABSOLUTE ZERO (0 K) No movement of particles

ABSOLUTE ZERO (0 K) Video Clip: Click here

KINETIC ENERGY TEMPERATURE Kinetic Energy↑ the Temp↑

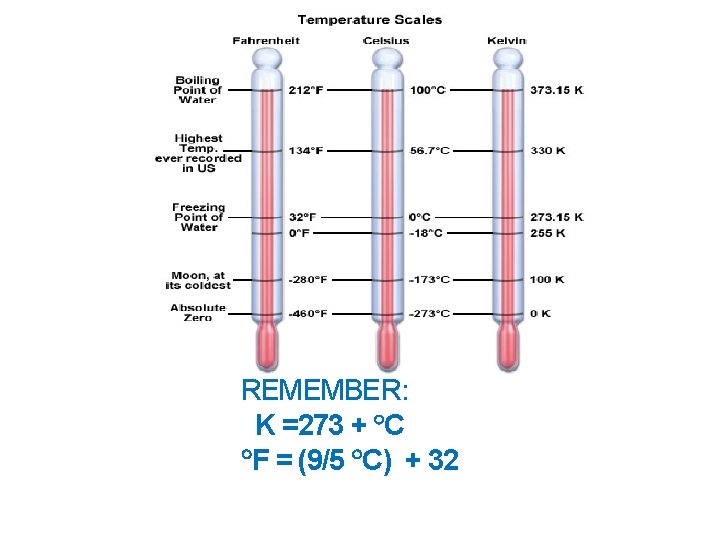
REMEMBER: K =273 + C F = (9/5 C) + 32

EXPLORING HEAT Video clip: click here

APPLICATION

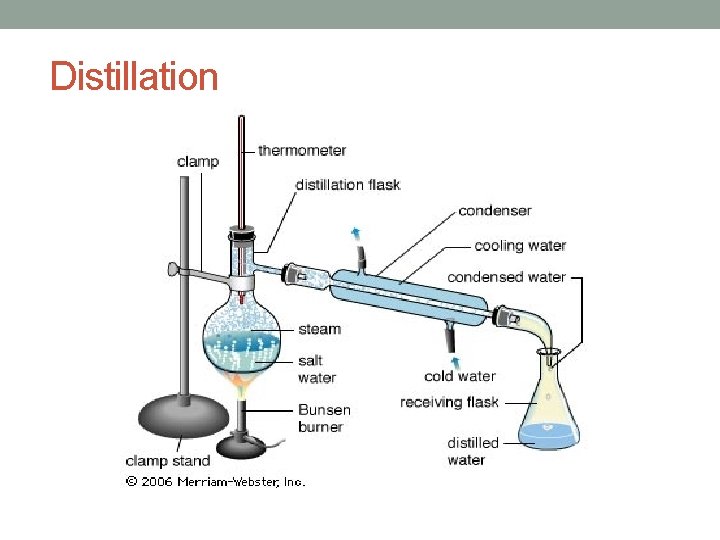
Distillation A homogenous mixture of liquids, a solution, can be separated using the knowledge that: • every substance has a unique boiling point • all gasses can condense

Distillation

PLASMA IN A BOTTLE Video Clip: click here

WEATHER Video clip: Click here

SCUBA Video Clip: click here

NITROGEN NARCOSIS Video Clip: click here