States of Matter Matter exists as Solid 1














- Slides: 18

States of Matter

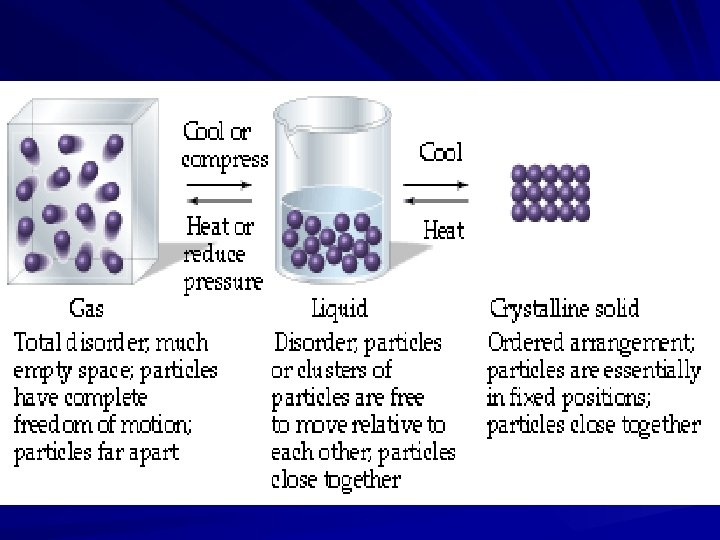
Matter exists as: *_____Solid_______: 1. Dense, incompressible, highly organized structure, usually crystalline. 2. Definite shape, definite volume. ** __amorphous__ _____solid___: A solid that lacks a rigid internal structure.

*___Liquid_______: 1. Fluid, slightly compressible, held together by +/- forces between particles. 2. Indefinite shape, definite volume. ** ____Glass_____ Transparent, inorganic products of fusion that have cooled to a rigid state, without crystallizing.

*_____Gas______: 1. Fluid, easily compressed, mostly large empty spaces with particles scattered throughout. 2. No positive or negative forces exist between particles. 3. Indefinite shape, indefinite volume. *____Plasma_________: 1. Stream of electrons and positive ions. 2. Exist only at extremely high temperatures (> 5000°C).


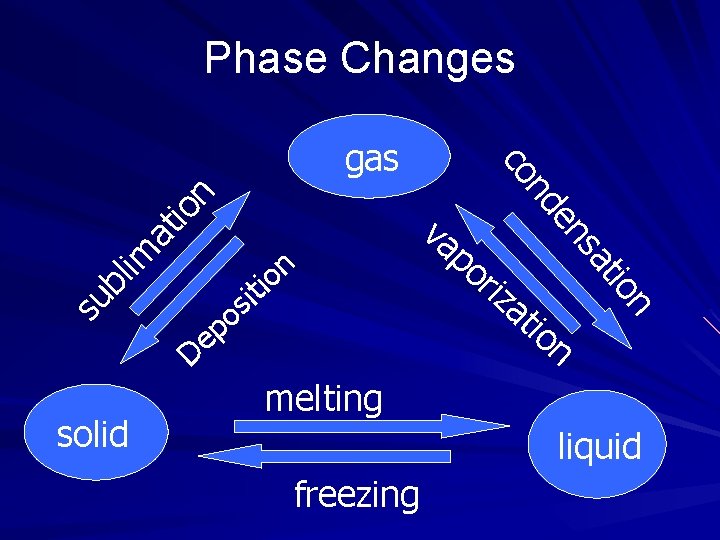
Phase changes occur due to a change in energy and/or temperature. Phase changes are PHYSICAL changes. As heat is absorbed or released in a phase change, the change in energy, not temperature, caused the change to occur.

Phase Changes n io at lim su b ep D solid at io n tio i s o riz sa en n o ti va po nd co gas n melting liquid freezing

Solid to liquid Melting point and freezing point of a substance occur at the same temperature. Covalent Compounds usually have low melting points. Ionic Compounds usually have high melting points.

Liquid to a gas • Vapor: The gas phase of a substance that normally exists as a liquid or solid. • Evaporation: Change of a liquid to a gas/vapor at a temperature below its boiling point. • Boiling : Change of a liquid to a gas/vapor at a temperature when the vapor pressure of a liquid equals the external pressure

• Normal boiling point: The temperature at which a liquid boils at standard pressure • Elevation and atmospheric pressure are inversely proportional. • Elevation and boiling point are inversely proportional.

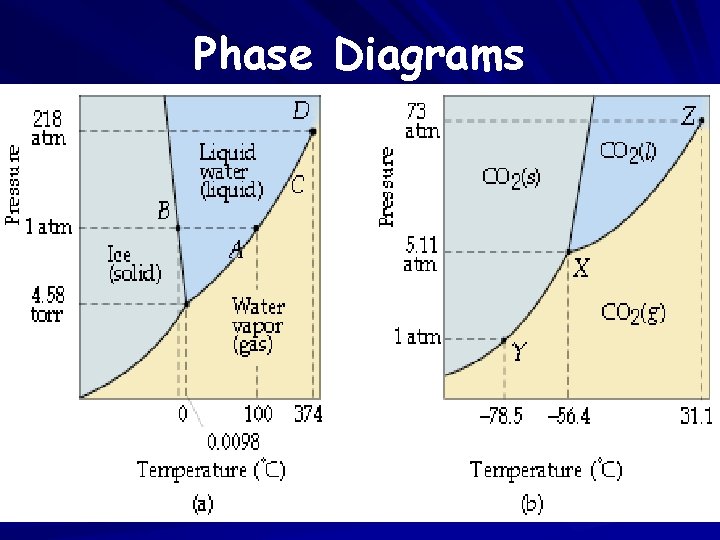
Phase Diagrams

Terms to Know… Triple point Normal MP and Normal BP Critical Point

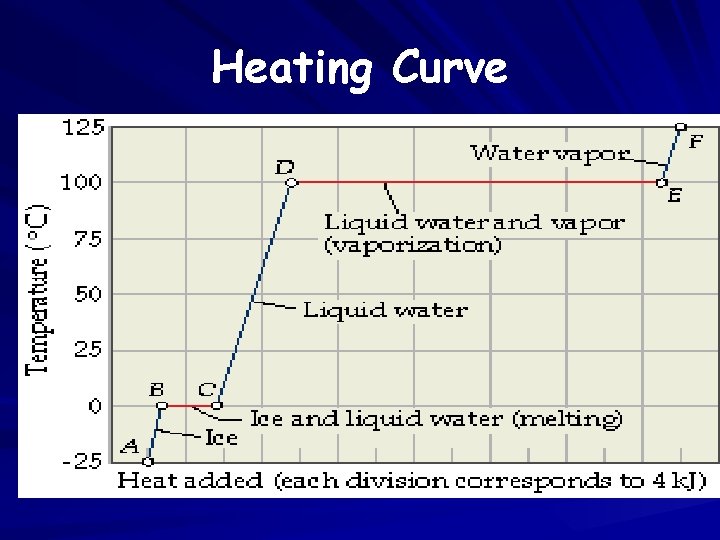
Heating Curve

Phase Diagram * At any point on a line, the two phases bordering the line are in equilibrium. * At any other point (not on a line), only one phase exists. * A phase change can be caused by changing one or both variables (temperature or pressure). *_Critical Temperature: The highest temperature at which a substance can exist as a liquid (regardless of pressure). *Triple Point: Point at which all three phases co-exist in equilibrium.

Intramolecular Forces within a particle – Ionic – attraction between anions and cations – Covalent – attraction between a nucleus and shared electrons – Metallic – attraction between metal cations and mobile electrons

Intermolecular Forces Force of Attraction between particles – Dipole-dipole: attraction between + and – ends of two polar molecules. – Hydrogen bonding: attraction between hydrogen atoms and atoms with high electronegativities in other molecules. H to N or O or F

London Dispersion forces: Very slight attraction between all atoms due to “polar moments” caused by the movements of electrons. – Used to explain why gases can be liquefied.

Arrange the following gases in order of increasing boiling point: A. H 2 B. NH 3 C. HCl D. HF E. Na. Cl