States Boiling Point Melting Point and Solubility Solid
States, Boiling Point, Melting Point, and Solubility Solid Liquid Gas

Questions � What are the 3 states of matter? � Write a definition for each state?

Defining States of Matter �States of matter are NOT defined by what they are made of. �Example: solids can be elements (gold), compounds (Salt = Na. Cl), or mixtures (butter) Element (Au) Compound (Na. Cl) Mixture (Milk, Salt, etc)

Defining States of Matter �States of matter are defined by whether they hold SHAPE and VOLUME Element (Au) Compound (Na. Cl) Mixture (Milk, Salt, etc) ALL KEEP THE SAME SHAPE AND VOLUME = Solids

Defining States of Matter �Solids – have a definite SHAPE and VOLUME. Element (Au) Compound (Na. Cl) Mixture (Milk, Salt, etc) ALL KEEP THE SAME SHAPE AND VOLUME
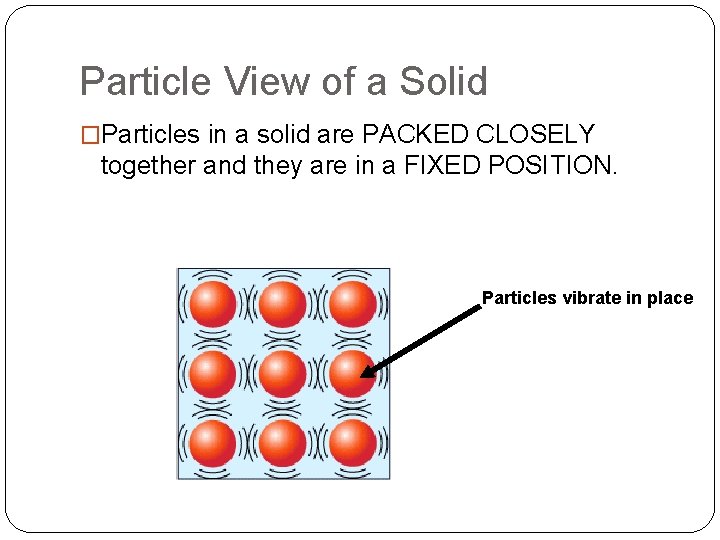
Particle View of a Solid �Particles in a solid are PACKED CLOSELY together and they are in a FIXED POSITION. Particles vibrate in place

Liquids �Liquids – has definite VOLUME but no defined SHAPE 100 ml

Particle View of a Liquid �Packed CLOSELY (like a solid), but move FREELY around each other (must stay in contact).

Gases �Gases - do NOT have definite SHAPE or VOLUME. Bromine gas fills up the entire volume of the container
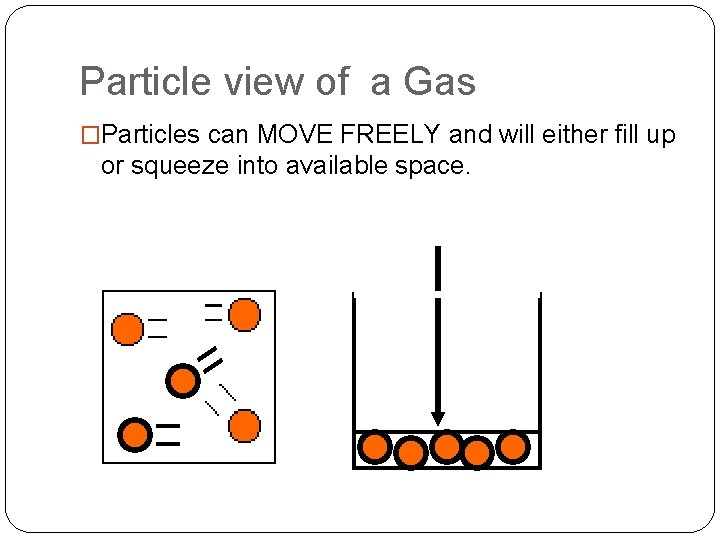
Particle view of a Gas �Particles can MOVE FREELY and will either fill up or squeeze into available space.

Questions � 1) Which Statement is True? �A) Liquids have a definite shape and volume �B) States of matter are defined by the substances they are made up of �C) Gases have a definite shape and volume �D) Solids have a definite shape and volume

Task �Draw a diagram of �A) Gas particles �B) Liquid particles �C) Solid particles
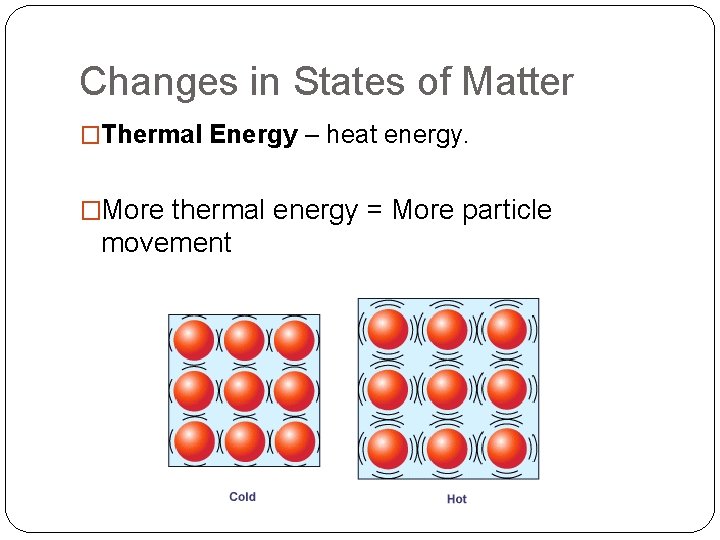
Changes in States of Matter �Thermal Energy – heat energy. �More thermal energy = More particle movement
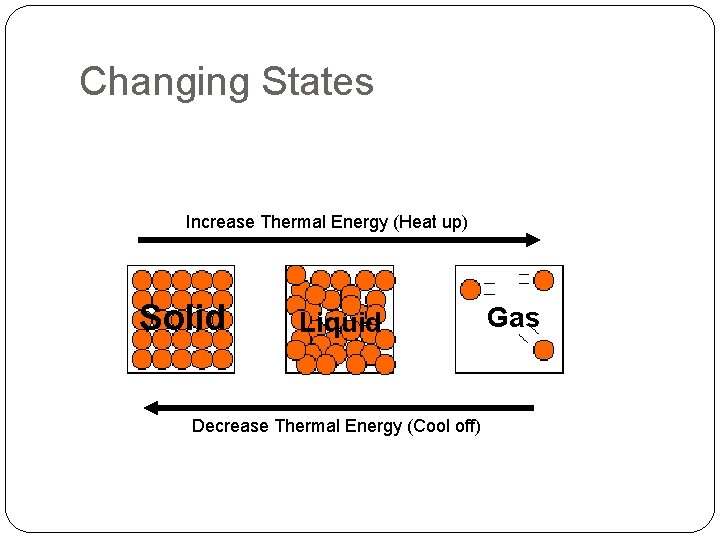
Changing States Increase Thermal Energy (Heat up) Solid Liquid Decrease Thermal Energy (Cool off) Gas

Melting point �Melting - change from solid to liquid �Melting point - SPECIFIC temperature when melting occurs. �Each pure substance has a SPECIFIC melting point. �Examples: �M. P. of Water = 0°C (32°F) �M. P. of Nitrogen = -209. 9 °C (-345. 81998 °F) �M. P. of Silver = 961. 93 °C (1763. 474 °F) �M. P. of Carbon = 3500. 0 °C (6332. 0 °F)
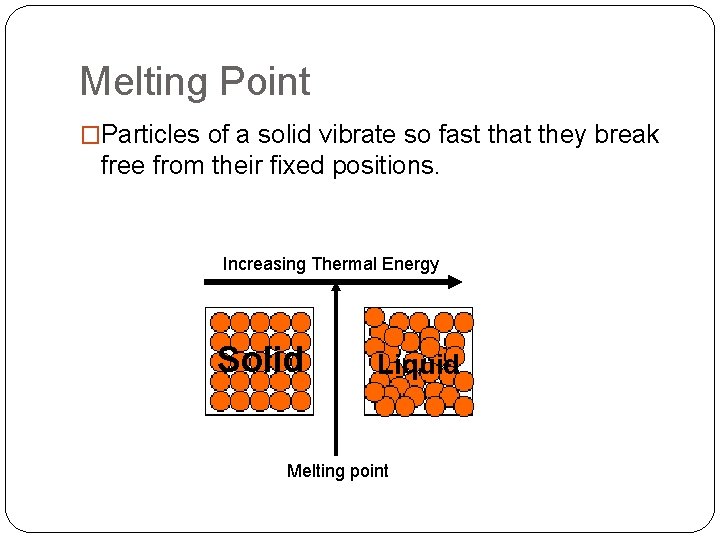
Melting Point �Particles of a solid vibrate so fast that they break free from their fixed positions. Increasing Thermal Energy Solid Liquid Melting point

Vaporization �Vaporization – change from liquid to gas �Vaporization happens when particles in a liquid gain enough energy to form a gas. Increasing Thermal Energy Liquid Boiling point Gas

Two Kinds of Vaporization �Evaporation – vaporization that takes place only on the surface of the liquid �Boiling – when a liquid changes to a gas BELOW its surface as well as above.

Boiling Point �Boiling Point – temperature at which a liquid boils �Each pure substance has a SPECIFIC boiling point. �Examples: �B. P. of Water = 100°C (212°F) �B. P. of Nitrogen = -195. 79 °C (-320. 42 °F) �B. P. of Silver = 2162 °C (3924 °F) �B. P. of Carbon = 4027 °C (7281 °F)
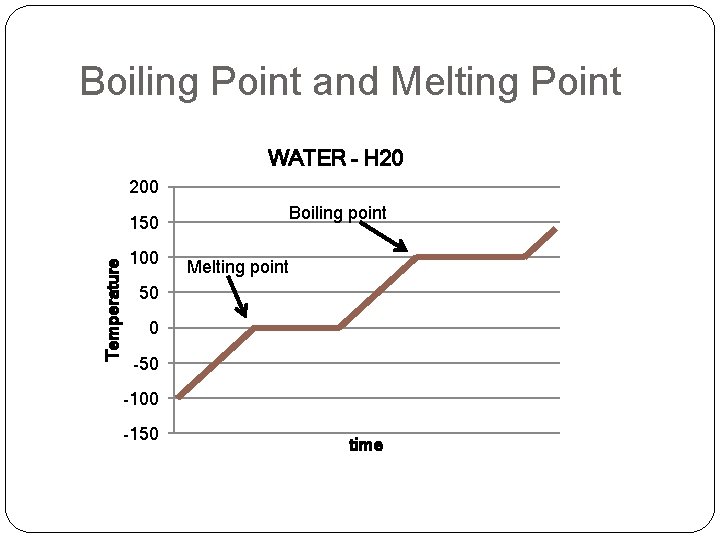
Boiling Point and Melting Point WATER – H 20 200 Boiling point Temperature 150 100 Melting point 50 0 -50 -100 -150 time

Solubility �Maximum amount of a substance that can be dissolved in a liquid (at a specific temperature). Salt (Na. Cl) Water (H 20) at 20°C

Solubility �Solute – substance being dissolved �Solvent – liquid substance that solute is dissolved into Salt (Na. Cl) Water (H 20) at 20°C

Solubility �Solute – ? ? ? �Solvent – ? ? ? Salt (Na. Cl) Salt Water (H 20) at 20°C
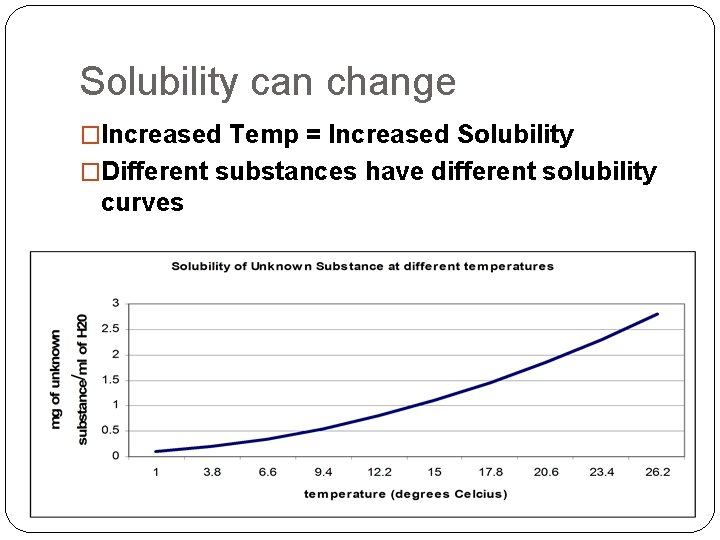
Solubility can change �Increased Temp = Increased Solubility �Different substances have different solubility curves
- Slides: 24