State Veterinary Drug Quality Control Laboratory Accreditation Laboratory

State Veterinary Drug Quality Control Laboratory /Accreditation Laboratory/ Uuganbayar, Dorjbat

Brief introduction of SVDQCL • SVDQCL stands for State Veterinary Drug Quality Control Laboratory and it was first established in 1973 • At that time it was controlling the quality of only vaccination which was produced by the domestic producer Bio-combinat.

Brief introduction of SVDQCL • In 1990 mongolia shifted to a market economy system; regurding this drug producting and importing have beenrapidly developing until today. Therefore, the operations and scope of SVDQCL is also broadening. • SVDQCLis a government owned organization that has the right of doing tests on drugs, biopreparation, and medical raw materials, and certify ther on beholf of the state. • There is a Certification Unit in SVDQCL and that does conformation assessment.

Goal To introduce quality system in production of medicine and drugs produced in Mongolia and imported into the country, to manage professional service in implementation of strategy in the sector with high quality biopreparations.

Activities • To test and certify drugs preparation, bio- preparations registered by the drug registration list of Mongolia and suggested ones; • To manage monitoring service for enterprises and grant certificates to tested products; • To introduce quality system into animal drug production and its importation; • To set national standard related to certification of animal drugs and make it closer to international level; • To expand foreign relations and technique and technology;

Our Team • We have 35 staff totally working together (DVMS, Chemists, biotechnologists and pharmacists) • 65 percent of laboratory staff has higher education • Ph. doctor-1, doctoral candidate-2, master -6, postgraduate-5

SVDQCL Certification • The Testing laboratory of SVDQCL met the standard of MNS/ISO 17025 and got certified by the National Center for Standardization and meteorology in 2004, 2007 and 2009 • The Certification Unit of SVDQCL met the standard of MNS/ISO/IEC Guide 65 -2004 and got certified by the national center for Standardization and meteorology in 2004, 2007 and 2009
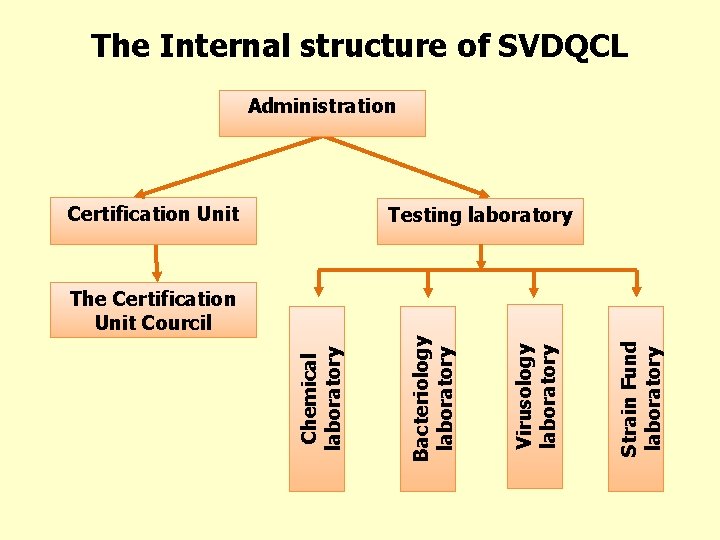
The Internal structure of SVDQCL Administration Certification Unit Testing laboratory Strain Fund laboratory Virusology laboratory Bacteriology laboratory Chemical laboratory The Certification Unit Courcil
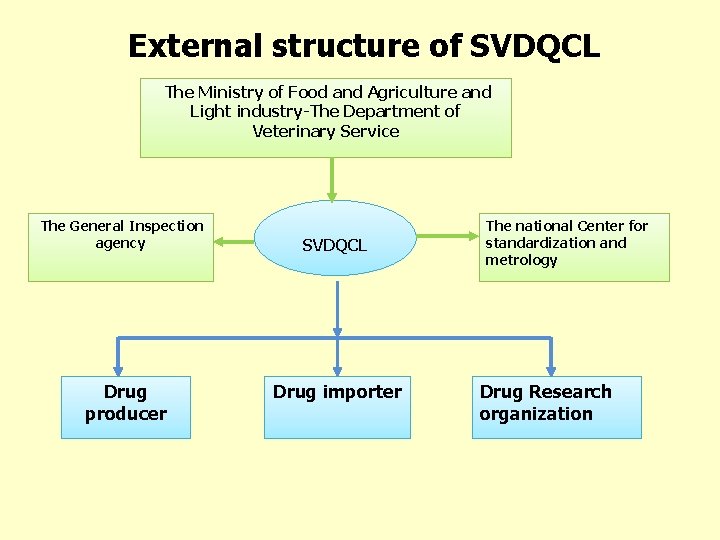
External structure of SVDQCL The Ministry of Food and Agriculture and Light industry-The Department of Veterinary Service The General Inspection agency Drug producer SVDQCL Drug importer The national Center for standardization and metrology Drug Research organization

Local authority and regulations related to the veterinary medicine management and quality control • MNS ISO/IEC 7025: 2007 • MNS ISO/TR 10013: 2001 • MNS ISO 8402: 97 • MNS ISO 9000: 2001 • MNS ISO 9001: 2001 • MNS ISO/IEC Guide 65: 2003 • • • Pharmacopeia of: USA China Russia British National standard SOP
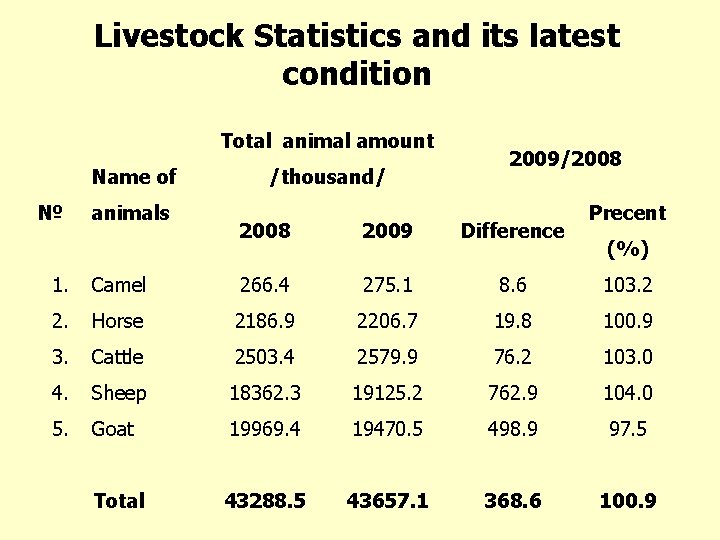
Livestock Statistics and its latest condition Total animal amount Name of № animals /thousand/ 2009/2008 2009 Difference Precent (%) 1. Camel 266. 4 275. 1 8. 6 103. 2 2. Horse 2186. 9 2206. 7 19. 8 100. 9 3. Cattle 2503. 4 2579. 9 76. 2 103. 0 4. Sheep 18362. 3 19125. 2 762. 9 104. 0 5. Goat 19969. 4 19470. 5 498. 9 97. 5 Total 43288. 5 43657. 1 368. 6 100. 9

Introduction of animal health products industry • The veterinary manufacture was first founded in MGL in 1923. • In 1973, Bio-combinate was established by the investment of The Republic of Hungary to produce vaccines, serum and diagnostic tests for the prevention of contagious diseases. • At the present the Bio-combinate has become one of the biggest manufacturers that produces 26 different types of vaccines, 6 serum and 24 diagnostic tests.

Introduction of animal health products industry • There more drug factories have been established that produce antiparasitic drugs because parasitic diseases are quite common in our country. • There have been more and more manufacturers established that produce commonly used drugs.

Number of manufacturers, importers 7 Drug manufacturers and 38 Drug importers in Mongolia.

Chart of top companies especially domestic importers 1. Mongolia V. E. T. NET NGO 2. Monvetmed LLC 3. Global Business Link LLC 4. Sorlog sureg LLC 5. UNN LLC

Number of registered products Over 370 drugs have been registered in the veterinary drug registration of Mongolia. Those are: Antibiotics-80 Vaccines-54 Parasitic drugs-56 Others-180 The state veterinary drug registration does not register botanical and traditional drugs but our laboratory tests these drugs.
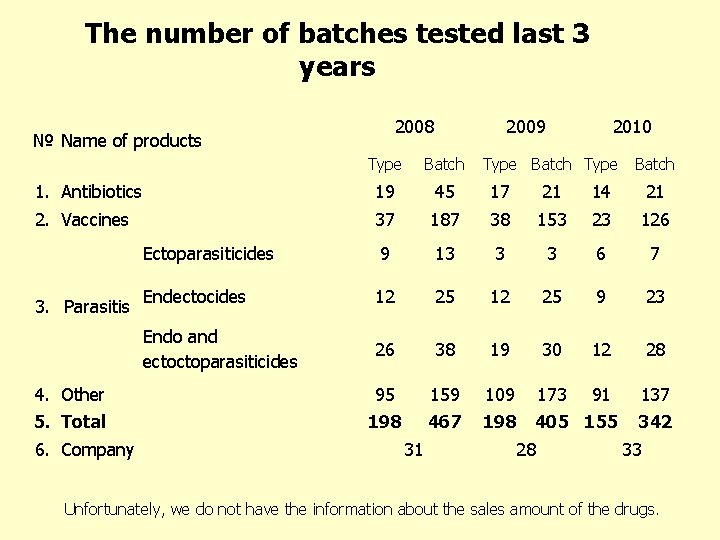
The number of batches tested last 3 years 2008 № Name of products 2009 2010 Type Batch 1. Antibiotics 19 45 17 21 14 21 2. Vaccines 37 187 38 153 23 126 Ectoparasiticides 9 13 3 3 6 7 Endectocides 12 25 9 23 Endo and ectoctoparasiticides 26 38 19 30 12 28 4. Other 95 159 109 173 91 137 5. Total 198 467 198 405 155 3. Parasitis 6. Company 31 Type Batch 28 342 33 Unfortunately, we do not have the information about the sales amount of the drugs.

Sales amount and list of high ranking products by their types 1. The government of MGL funds 100% percent for manufacturing of the contagious disease prevention vaccines and provides them to numbers 70% of total livestock of MGL is vaccinated with those vaccines. Therefore, vaccine sale is the highest. 2. Anti-parasitic drugs 3. Antibiotics

Highlighted issues of Animal health product market • It is likely to have issues of fake drug production and importation • We need to train our human resource. • We also need to improve our equipments and technology

The approval process of animal health product Requirements: • The manufacturer should have GMP or ISO international standard certification. • The accredited lab result of the manufacturer. • Published material about pharmacological and clinical trials. • Registrations in not less than 3 countries. • Approved instruction that includes the drug ingredients, chemistry, usage, precautions, side effects, drug interactions, storage, warnings etc. • Drug sample for a test. • Methods and methodology to test

The approval process of animal health product • Analysis certificate • The manufacturer introduction. • Standard references indicated in testing methods. • Every manufacturer has its internal, control lab. We require the result of that lab. Additionally, the result of the accredited lab. • We try to pursue the methods, methodology, and standard used in countries such as USA although we have the pharmacopeia of the same country. Because the result can be various if we test with such pharmacopeia depending on the testing method, technology, substance, and reagent. For this reason we require the approved standard method of analysis which is pursued in that present country.

The approval process of animal health product Registration fee: The client is responsible for the cost of drug registration. The fee is constituted • Registration procedure • Test and certification The fee: • May be various depending on drug type and testing period. • We are changing the fee so far. That is why it is not possible to inform the realistic amount now.

The approval process of animal health product Duration for getting approval: The test is performed within approximately duration of 7 -30 days after the submission of medicine product registration request. The Veterinary Drug Committee should make its decision within 21 days after the test result come out. The state registration certificate should be given within 14 days after the decision of the veterinary committee. The state registration certificate is given for the period of 5 years.

The approval process of animal health product Renewal: Shall renew the national registration in the following condition: 1. If drug industry governance, name and address were changed 2. If drug standards were changed

The approval process of animal health product Registration period: Notify the expiration date of certain drugs prior to 3 months then it’ll be reregistered.

The approval process of feed and feed additive • There is no article in the law at this moment about how to register the feed and feed additives in the drug registration. • If anyone wants to import the feed and feed additives, the same documents requested for the drug registration is also needed, even those feed and feed additives are not required to be registered in the drug registration. And those feed and feed additives are supplied to the market after inspection of State Veterinary Drug Quality Control Laboratory.

Quality Management System Strategy: Approve drugs that provided quality, active and safe for environment, animal and human by testing as using appropriate methods according to standard of veterinary practice of Mongolia.

Purpose: 1. Must be provided by accommodation and environmental condition, human factors, equipment, reagent, animals and method analysis. 2. To have quality manual to approve testing process and principles 3. Basis of the testing process and result must be independence from other factors including administers and finances. 4. Testing result must be reliable and complete. 5. Laboratory must have International and National standards, Reference materials and Reference standards.
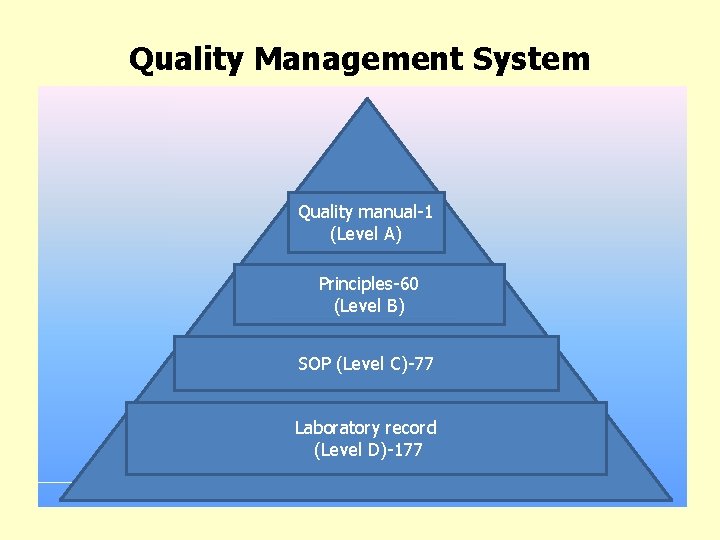
Quality Management System • 29 Quality manual-1 (Level А) Principles-60 (Level B) SOP (Level C)-77 Laboratory record (Level D)-177

Thank you very much Phone: (976)-(11)- 99061455 Email: u_dedee@yahoo. com
- Slides: 30