STATE OF THE INDUSTRY 2017 Pharmaceutical Biotech Lisa

STATE OF THE INDUSTRY 2017 Pharmaceutical & Biotech Lisa Bannerman, Business Intelligence © 2015 Envigo 1 envigo. com

Pharmaceutical macro environment + 2016 marked huge uncertainty for the pharma world + US presidential election (drug pricing, health insurance, tax reform) + Weak economic growth + Brexit + Although this has continued into 2017, the outlook for pharma remains broadly positive + Sales forecast of 6. 5% CAGR to hit over $1 tr in 2022 + Global R&D growing at ~3% + Successful launch of several blockbusters, with more in the pipeline + Growth in higher priced, speciality drugs for orphan indications + First CAR-T drug approved + The biotech bubble has deflated but hasn’t burst + Funds remain well stocked, although investors are perhaps taking fewer risks + The IPO market has dimmed in light of global market uncertainty 2

Pharmaceutical macro environment + Big pharma remains a key backer of innovation + Continue to in-license molecules, especially biologics from smaller firms + Oncology treatments continue to dominate both current and projected drug pipelines + Other major growth areas include immune therapies, biologics, rare-disease drugs and neurological treatments + However, there are potential industry headwinds to look for + Rising cost of R&D – average of $4 bn per NME over last 10 years + Maintaining drug approval levels + The need to restock pipelines ahead of the next patent cliff in 2022 + Huge downward pressure on drug pricing 3

Sales trends 4
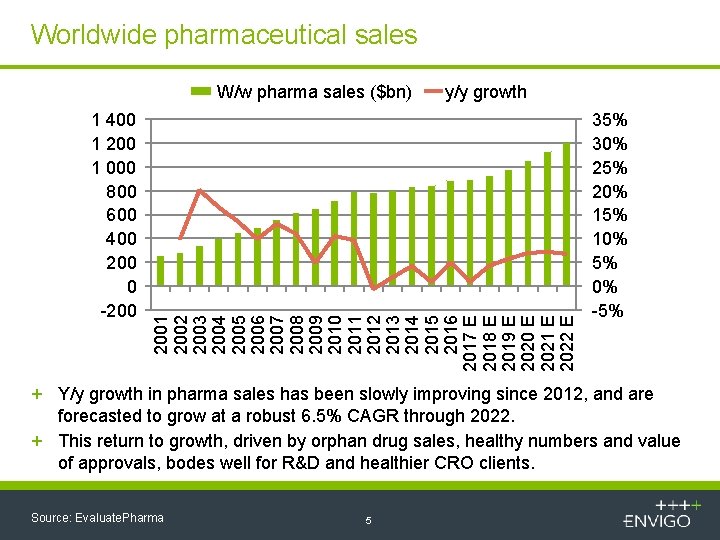
Worldwide pharmaceutical sales 1 400 1 200 1 000 800 600 400 200 0 -200 y/y growth 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 E 2018 E 2019 E 2020 E 2021 E 2022 E W/w pharma sales ($bn) 35% 30% 25% 20% 15% 10% 5% 0% -5% + Y/y growth in pharma sales has been slowly improving since 2012, and are forecasted to grow at a robust 6. 5% CAGR through 2022. + This return to growth, driven by orphan drug sales, healthy numbers and value of approvals, bodes well for R&D and healthier CRO clients. Source: Evaluate. Pharma 5
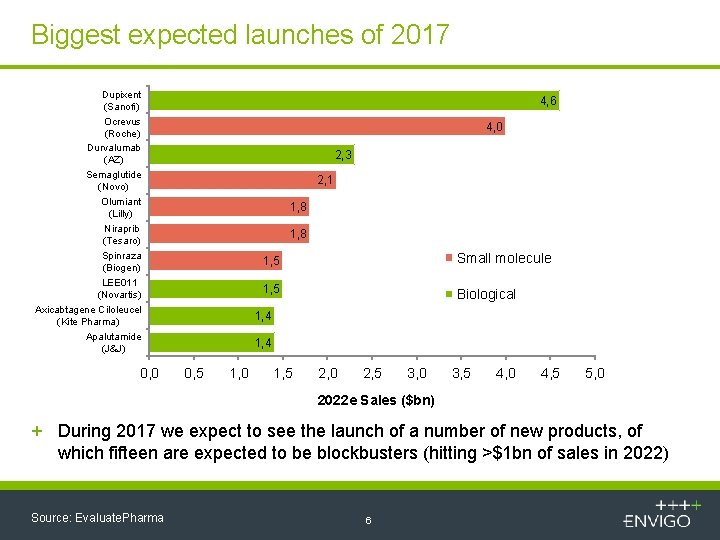
Biggest expected launches of 2017 Dupixent (Sanofi) Ocrevus (Roche) Durvalumab (AZ) Semaglutide (Novo) Olumiant (Lilly) Niraprib (Tesaro) Spinraza (Biogen) LEE 011 (Novartis) Axicabtagene Ciloleucel (Kite Pharma) Apalutamide (J&J) 0, 0 4, 6 4, 0 2, 3 2, 1 1, 8 1, 5 Small molecule 1, 5 Biological 1, 4 0, 5 1, 0 1, 5 2, 0 2, 5 3, 0 3, 5 4, 0 4, 5 5, 0 2022 e Sales ($bn) + During 2017 we expect to see the launch of a number of new products, of which fifteen are expected to be blockbusters (hitting >$1 bn of sales in 2022) Source: Evaluate. Pharma 6
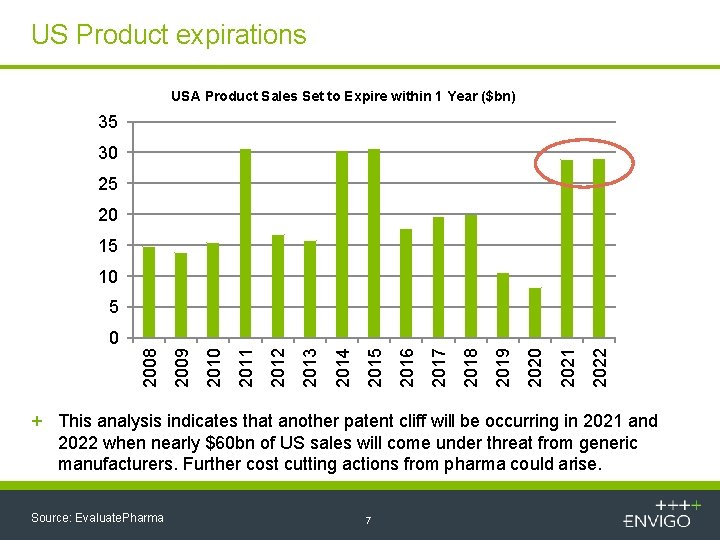
US Product expirations USA Product Sales Set to Expire within 1 Year ($bn) 35 30 25 20 15 10 5 2022 2021 2020 2019 2018 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 0 + This analysis indicates that another patent cliff will be occurring in 2021 and 2022 when nearly $60 bn of US sales will come under threat from generic manufacturers. Further cost cutting actions from pharma could arise. Source: Evaluate. Pharma 7
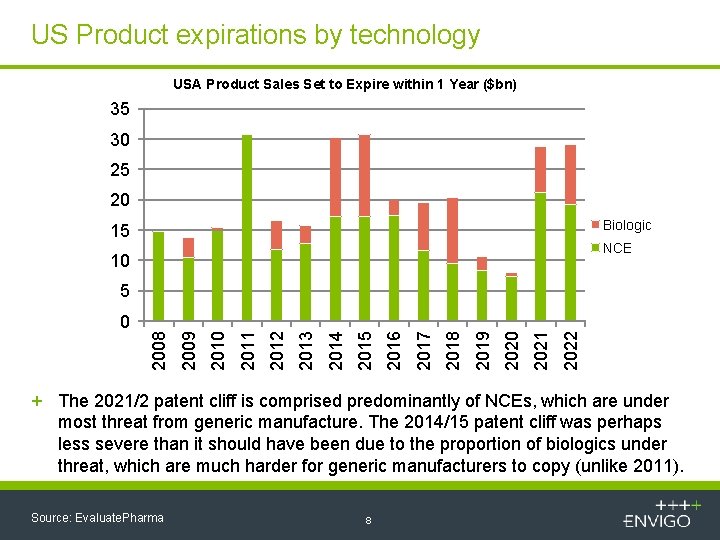
US Product expirations by technology USA Product Sales Set to Expire within 1 Year ($bn) 35 30 25 20 Biologic 15 NCE 10 5 2022 2021 2020 2019 2018 2017 2016 2015 2014 2013 2012 2011 2010 2009 2008 0 + The 2021/2 patent cliff is comprised predominantly of NCEs, which are under most threat from generic manufacture. The 2014/15 patent cliff was perhaps less severe than it should have been due to the proportion of biologics under threat, which are much harder for generic manufacturers to copy (unlike 2011). Source: Evaluate. Pharma 8
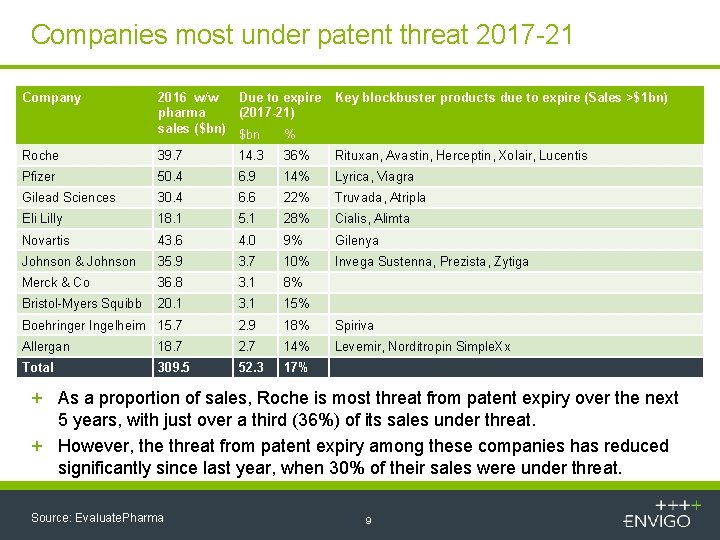
Companies most under patent threat 2017 -21 Company 2016 w/w Due to expire pharma (2017 -21) sales ($bn) $bn % Key blockbuster products due to expire (Sales >$1 bn) Roche 39. 7 14. 3 36% Rituxan, Avastin, Herceptin, Xolair, Lucentis Pfizer 50. 4 6. 9 14% Lyrica, Viagra Gilead Sciences 30. 4 6. 6 22% Truvada, Atripla Eli Lilly 18. 1 5. 1 28% Cialis, Alimta Novartis 43. 6 4. 0 9% Gilenya Johnson & Johnson 35. 9 3. 7 10% Invega Sustenna, Prezista, Zytiga Merck & Co 36. 8 3. 1 8% Bristol-Myers Squibb 20. 1 3. 1 15% Boehringer Ingelheim 15. 7 2. 9 18% Spiriva Allergan 18. 7 2. 7 14% Levemir, Norditropin Simple. Xx Total 309. 5 52. 3 17% + As a proportion of sales, Roche is most threat from patent expiry over the next 5 years, with just over a third (36%) of its sales under threat. + However, the threat from patent expiry among these companies has reduced significantly since last year, when 30% of their sales were under threat. Source: Evaluate. Pharma 9

R&D trends 10

Growth in biopharmaceutical R&D + R&D spend is a major driver of growth in the CRO industry + Traditionally, companies have spent ~25% of Rx sales on R&D, although this has fallen to 20% in recent years. + Pressures on pharma sales lead to pressures on R&D + R&D growth fell dramatically in 2009 as pharma companies attempted to reduce their cost bases + The patent cliff (2010 -12) saw a huge proportion of industry sales under threat from generic competition + A huge wave of consolidation in the industry, as companies attempted to bolster their pipelines + Focus on fewer therapeutic areas + Focus on later stage-pipelines to generate near-term revenues + R&D is forecast to grow at ~3% over the next few years 11
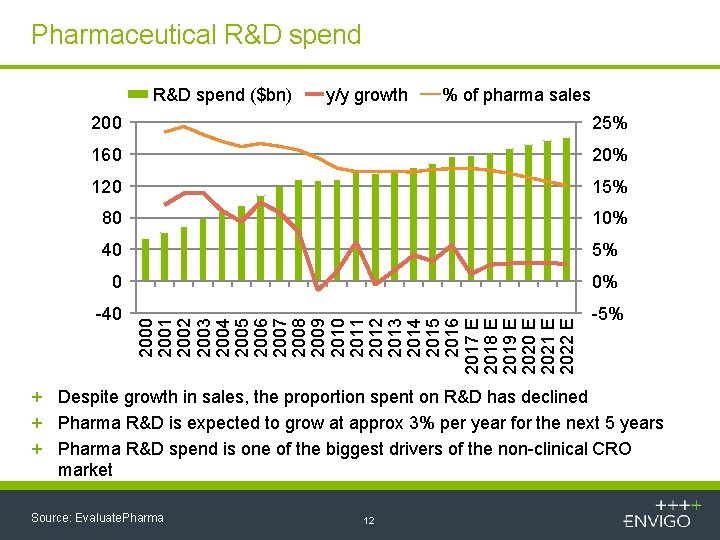
Pharmaceutical R&D spend ($bn) y/y growth % of pharma sales 25% 160 20% 120 15% 80 10% 40 5% 0 0% -40 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 E 2018 E 2019 E 2020 E 2021 E 2022 E 200 -5% + Despite growth in sales, the proportion spent on R&D has declined + Pharma R&D is expected to grow at approx 3% per year for the next 5 years + Pharma R&D spend is one of the biggest drivers of the non-clinical CRO market Source: Evaluate. Pharma 12
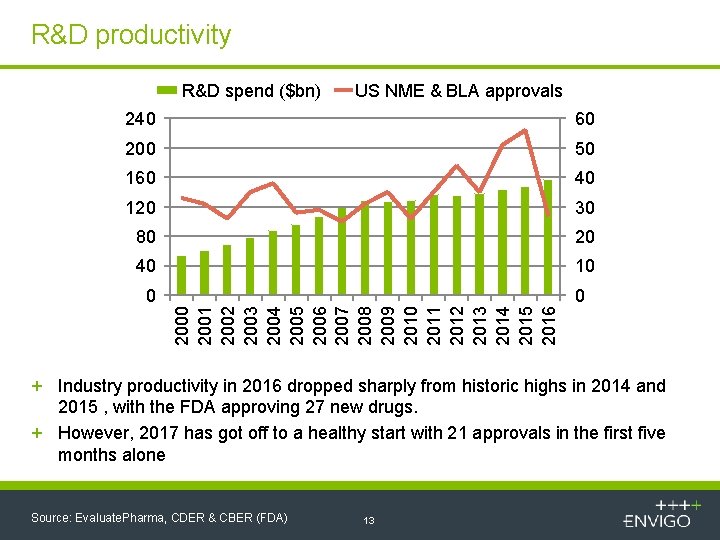
R&D productivity R&D spend ($bn) US NME & BLA approvals 60 200 50 160 40 120 30 80 20 40 10 0 0 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 240 + Industry productivity in 2016 dropped sharply from historic highs in 2014 and 2015 , with the FDA approving 27 new drugs. + However, 2017 has got off to a healthy start with 21 approvals in the first five months alone Source: Evaluate. Pharma, CDER & CBER (FDA) 13
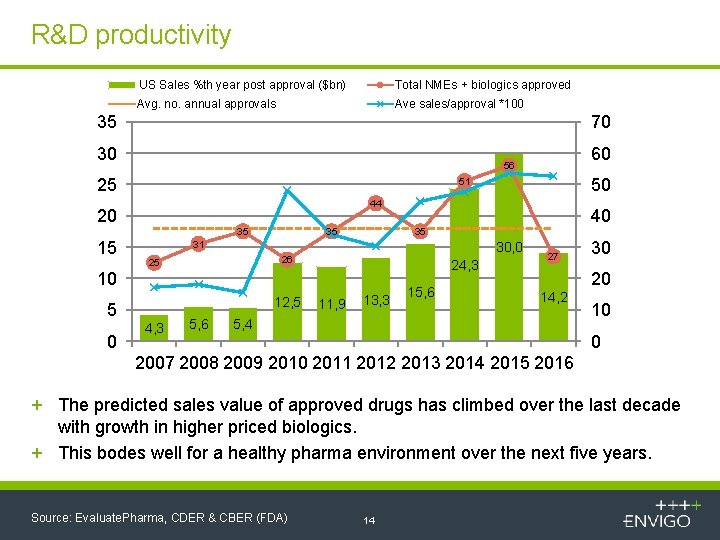
R&D productivity US Sales %th year post approval ($bn) Total NMEs + biologics approved Avg. no. annual approvals Ave sales/approval *100 35 70 30 25 44 35 35 0 40 35 31 30, 0 26 25 24, 3 10 5 50 51 20 15 60 56 12, 5 4, 3 5, 6 11, 9 13, 3 15, 6 27 30 20 14, 2 5, 4 10 0 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 + The predicted sales value of approved drugs has climbed over the last decade with growth in higher priced biologics. + This bodes well for a healthy pharma environment over the next five years. Source: Evaluate. Pharma, CDER & CBER (FDA) 14
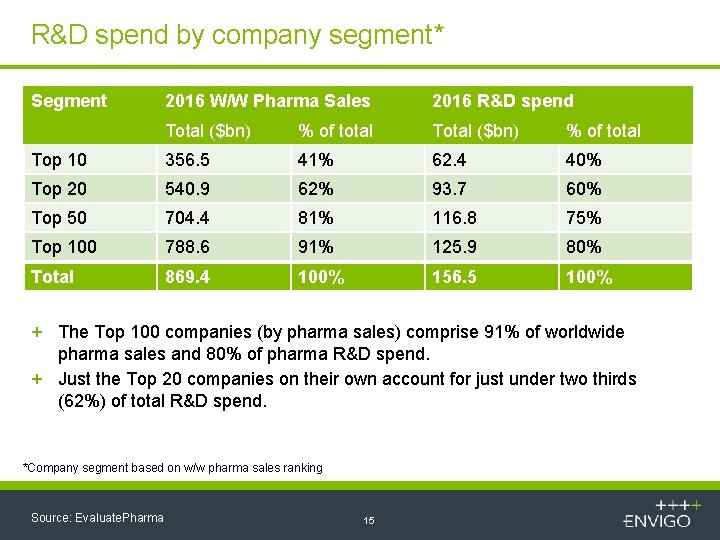
R&D spend by company segment* Segment 2016 W/W Pharma Sales 2016 R&D spend Total ($bn) % of total Top 10 356. 5 41% 62. 4 40% Top 20 540. 9 62% 93. 7 60% Top 50 704. 4 81% 116. 8 75% Top 100 788. 6 91% 125. 9 80% Total 869. 4 100% 156. 5 100% + The Top 100 companies (by pharma sales) comprise 91% of worldwide pharma sales and 80% of pharma R&D spend. + Just the Top 20 companies on their own account for just under two thirds (62%) of total R&D spend. *Company segment based on w/w pharma sales ranking Source: Evaluate. Pharma 15

Top R&D spenders Company R&D spend ($bn) 2016 2015 Change Roche 8. 7 8. 5 3% Novartis 7. 9 8. 5 -7% Pfizer 7. 8 7. 7 2% Johnson & Johnson 7. 0 6. 8 2% Merck & Co 6. 8 6. 6 3% Sanofi 5. 7 5. 6 1% Astra. Zeneca 5. 6 0% Eli Lilly 4. 9 4. 5 10% Glaxo. Smith. Kline 4. 7 -1% Bristol-Myers Squibb 4. 4 4. 0 9% Top 10 total 63. 7 62. 5 +2% % of total pharma R&D 41% 42% Source: Evaluate. Pharma 16 + The top R&D spenders have remained fairly consistent over the last few years. + However, their share of total R&D spend has gradually fallen over this time. + This shows the declining importance of the top players as the engines of R&D. + However, the Top 10 still account for 41% of total pharma R&D.
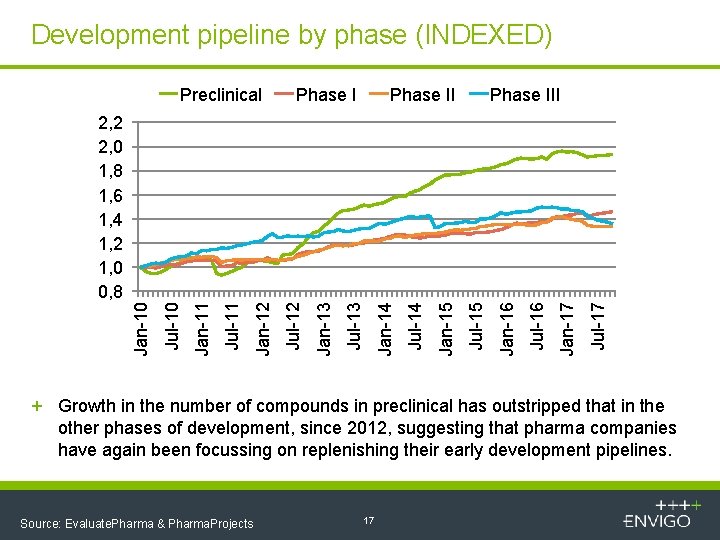
Development pipeline by phase (INDEXED) Preclinical Phase III Jul-17 Jan-17 Jul-16 Jan-16 Jul-15 Jan-15 Jul-14 Jan-14 Jul-13 Jan-13 Jul-12 Jan-12 Jul-11 Jan-11 Jul-10 Jan-10 2, 2 2, 0 1, 8 1, 6 1, 4 1, 2 1, 0 0, 8 + Growth in the number of compounds in preclinical has outstripped that in the other phases of development, since 2012, suggesting that pharma companies have again been focussing on replenishing their early development pipelines. Source: Evaluate. Pharma & Pharma. Projects 17
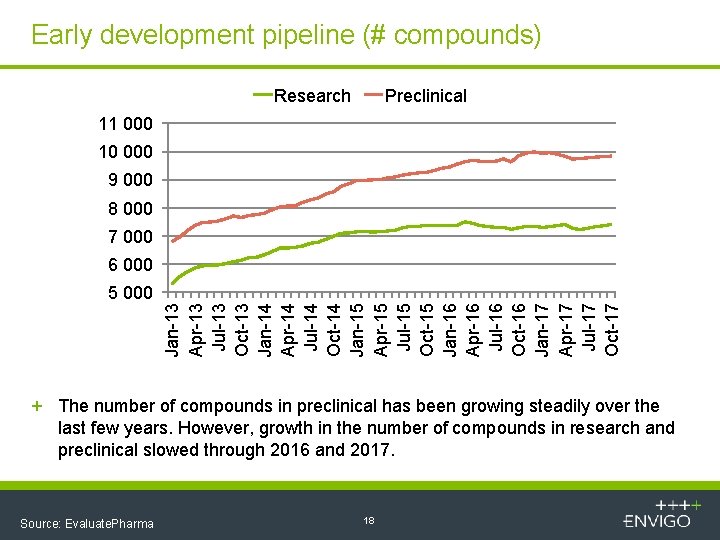
Early development pipeline (# compounds) Research Preclinical 11 000 10 000 9 000 8 000 7 000 6 000 Jan-13 Apr-13 Jul-13 Oct-13 Jan-14 Apr-14 Jul-14 Oct-14 Jan-15 Apr-15 Jul-15 Oct-15 Jan-16 Apr-16 Jul-16 Oct-16 Jan-17 Apr-17 Jul-17 Oct-17 5 000 + The number of compounds in preclinical has been growing steadily over the last few years. However, growth in the number of compounds in research and preclinical slowed through 2016 and 2017. Source: Evaluate. Pharma 18
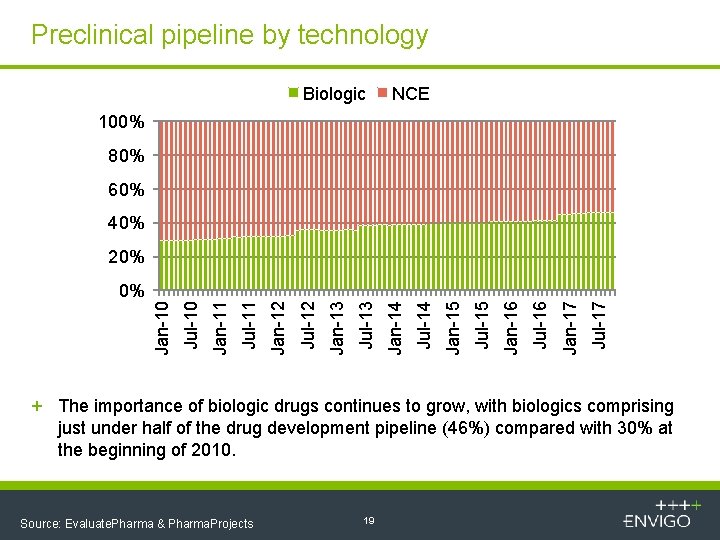
Preclinical pipeline by technology Biologic NCE 100% 80% 60% 40% 20% Jul-17 Jan-17 Jul-16 Jan-16 Jul-15 Jan-15 Jul-14 Jan-14 Jul-13 Jan-13 Jul-12 Jan-12 Jul-11 Jan-11 Jul-10 Jan-10 0% + The importance of biologic drugs continues to grow, with biologics comprising just under half of the drug development pipeline (46%) compared with 30% at the beginning of 2010. Source: Evaluate. Pharma & Pharma. Projects 19
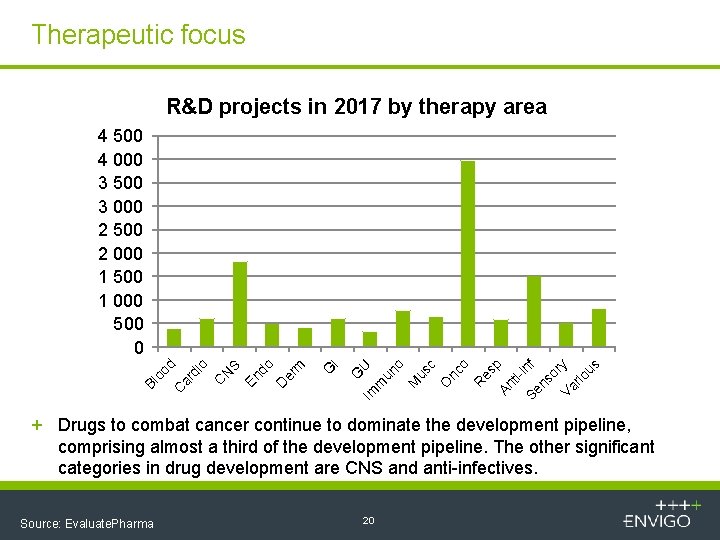
Therapeutic focus R&D projects in 2017 by therapy area nc o R es p An ti. Se inf ns or Va y rio us c O us M o un U m I G G Im Bl oo d C ar di o C N S En do D er m 4 500 4 000 3 500 3 000 2 500 2 000 1 500 1 000 500 0 + Drugs to combat cancer continue to dominate the development pipeline, comprising almost a third of the development pipeline. The other significant categories in drug development are CNS and anti-infectives. Source: Evaluate. Pharma 20

Biotech funding environment 21

Biotech macro environment + The biotech ‘bubble’ has deflated + Volume and value of venture financing deals down from record highs in 2014/15 + However, venture funding levels have merely normalized to pre-boom levels and are still showing healthy growth + Still substantial funds to be tapped + Focus on later-stage deals and companies with adequate capitalization + Q 4 16 saw three rounds break the $200 m mark + Biotechs have plenty of cash in hand to see them through the next few years + The IPO market has been hit hard by wider political and market uncertainty 22
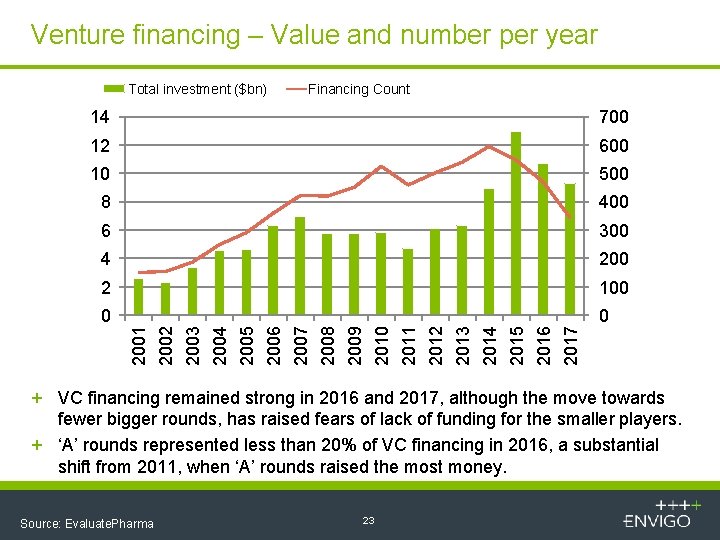
Venture financing – Value and number per year Total investment ($bn) Financing Count 700 12 600 10 500 8 400 6 300 4 200 2 100 0 0 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 14 + VC financing remained strong in 2016 and 2017, although the move towards fewer bigger rounds, has raised fears of lack of funding for the smaller players. + ‘A’ rounds represented less than 20% of VC financing in 2016, a substantial shift from 2011, when ‘A’ rounds raised the most money. Source: Evaluate. Pharma 23
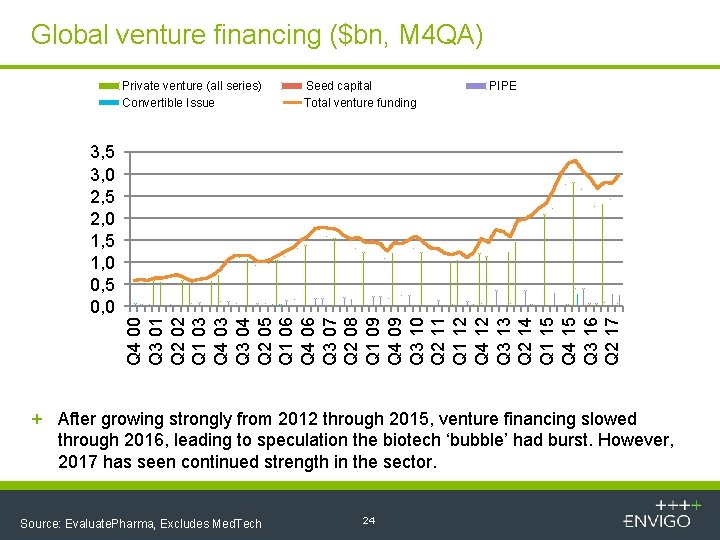
Global venture financing ($bn, M 4 QA) Private venture (all series) Convertible Issue Seed capital Total venture funding PIPE Q 4 00 Q 3 01 Q 2 02 Q 1 03 Q 4 03 Q 3 04 Q 2 05 Q 1 06 Q 4 06 Q 3 07 Q 2 08 Q 1 09 Q 4 09 Q 3 10 Q 2 11 Q 1 12 Q 4 12 Q 3 13 Q 2 14 Q 1 15 Q 4 15 Q 3 16 Q 2 17 3, 5 3, 0 2, 5 2, 0 1, 5 1, 0 0, 5 0, 0 + After growing strongly from 2012 through 2015, venture financing slowed through 2016, leading to speculation the biotech ‘bubble’ had burst. However, 2017 has seen continued strength in the sector. Source: Evaluate. Pharma, Excludes Med. Tech 24
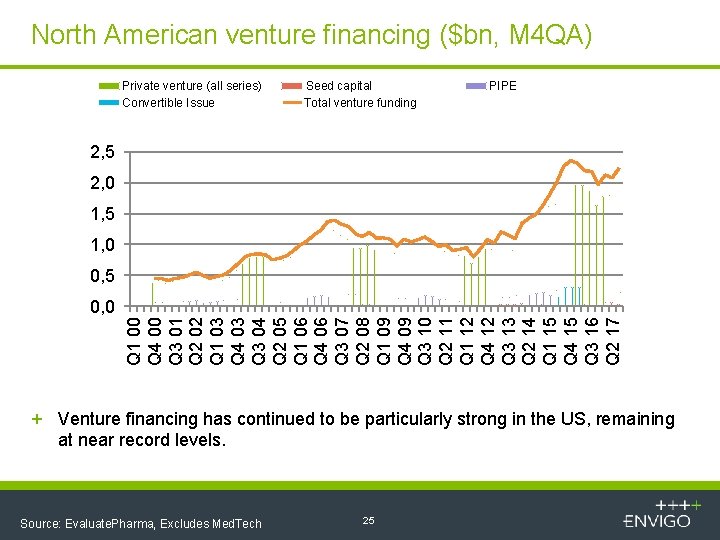
North American venture financing ($bn, M 4 QA) Private venture (all series) Convertible Issue Seed capital Total venture funding PIPE 2, 5 2, 0 1, 5 1, 0 0, 5 Q 1 00 Q 4 00 Q 3 01 Q 2 02 Q 1 03 Q 4 03 Q 3 04 Q 2 05 Q 1 06 Q 4 06 Q 3 07 Q 2 08 Q 1 09 Q 4 09 Q 3 10 Q 2 11 Q 1 12 Q 4 12 Q 3 13 Q 2 14 Q 1 15 Q 4 15 Q 3 16 Q 2 17 0, 0 + Venture financing has continued to be particularly strong in the US, remaining at near record levels. Source: Evaluate. Pharma, Excludes Med. Tech 25
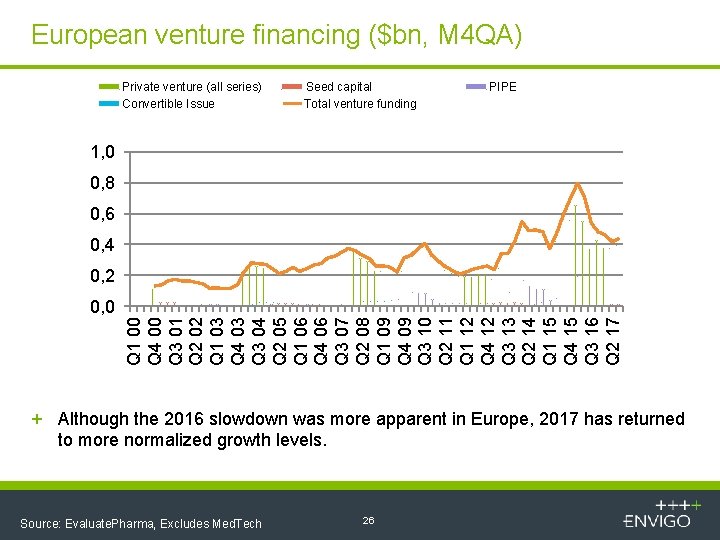
European venture financing ($bn, M 4 QA) Private venture (all series) Convertible Issue Seed capital Total venture funding PIPE 1, 0 0, 8 0, 6 0, 4 0, 2 Q 1 00 Q 4 00 Q 3 01 Q 2 02 Q 1 03 Q 4 03 Q 3 04 Q 2 05 Q 1 06 Q 4 06 Q 3 07 Q 2 08 Q 1 09 Q 4 09 Q 3 10 Q 2 11 Q 1 12 Q 4 12 Q 3 13 Q 2 14 Q 1 15 Q 4 15 Q 3 16 Q 2 17 0, 0 + Although the 2016 slowdown was more apparent in Europe, 2017 has returned to more normalized growth levels. Source: Evaluate. Pharma, Excludes Med. Tech 26
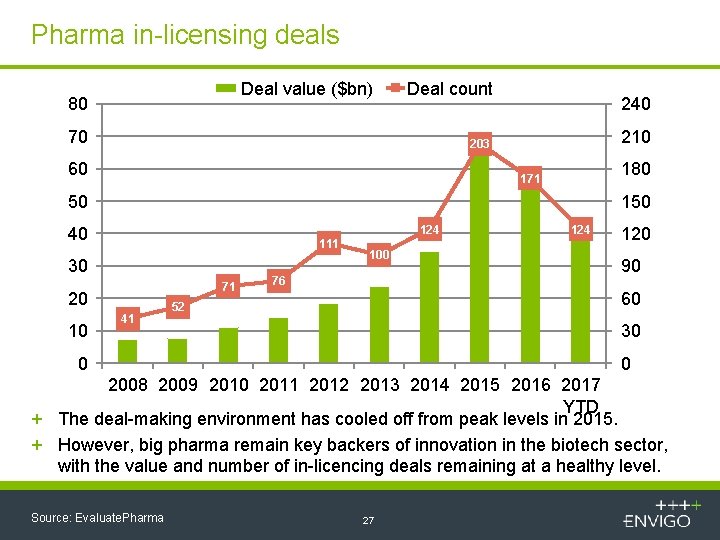
Pharma in-licensing deals Deal value ($bn) 80 Deal count 70 240 210 203 60 180 171 50 124 40 111 30 71 20 10 41 100 76 124 120 90 60 52 30 0 0 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 YTD + The deal-making environment has cooled off from peak levels in 2015. + However, big pharma remain key backers of innovation in the biotech sector, with the value and number of in-licencing deals remaining at a healthy level. Source: Evaluate. Pharma 27
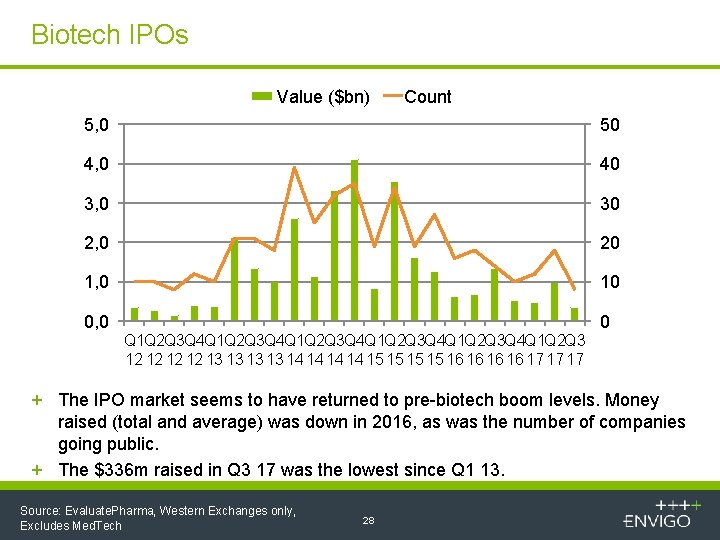
Biotech IPOs Value ($bn) Count 5, 0 50 4, 0 40 3, 0 30 2, 0 20 1, 0 10 0, 0 0 Q 1 Q 2 Q 3 Q 4 Q 1 Q 2 Q 3 Q 4 Q 1 Q 2 Q 3 12 12 13 13 14 14 15 15 16 16 17 17 17 + The IPO market seems to have returned to pre-biotech boom levels. Money raised (total and average) was down in 2016, as was the number of companies going public. + The $336 m raised in Q 3 17 was the lowest since Q 1 13. Source: Evaluate. Pharma, Western Exchanges only, Excludes Med. Tech 28
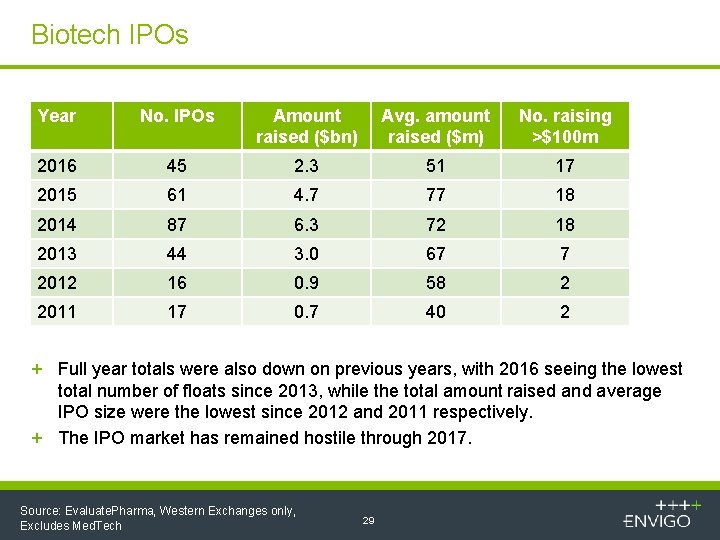
Biotech IPOs Year No. IPOs Amount raised ($bn) Avg. amount raised ($m) No. raising >$100 m 2016 45 2. 3 51 17 2015 61 4. 7 77 18 2014 87 6. 3 72 18 2013 44 3. 0 67 7 2012 16 0. 9 58 2 2011 17 0. 7 40 2 + Full year totals were also down on previous years, with 2016 seeing the lowest total number of floats since 2013, while the total amount raised and average IPO size were the lowest since 2012 and 2011 respectively. + The IPO market has remained hostile through 2017. Source: Evaluate. Pharma, Western Exchanges only, Excludes Med. Tech 29

Industry consolidation 30
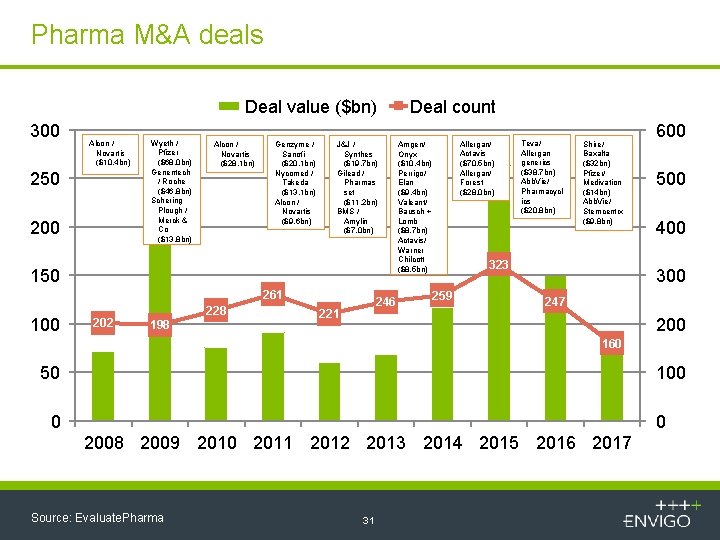
Pharma M&A deals Deal value ($bn) 300 Alcon / Novartis ($10. 4 bn) 250 200 Wyeth / Pfizer ($68. 0 bn) Genentech / Roche ($46. 8 bn) Schering Plough / Merck & Co ($13. 8 bn) Alcon / Novartis ($28. 1 bn) Genzyme / Sanofi ($20. 1 bn) Nycomed / Takeda ($13. 1 bn) Alcon / Novartis ($9. 6 bn) J&J / Synthes ($19. 7 bn) Gilead / Pharmas set ($11. 2 bn) BMS / Amylin ($7. 0 bn) 150 261 100 202 228 198 246 221 Deal count Allergan/ Actavis ($70. 5 bn) Allergan/ Forest ($28. 0 bn) Amgen/ Onyx ($10. 4 bn) Perrigo/ Elan ($9. 4 bn) Valeant/ Bausch + Lomb ($8. 7 bn) Actavis/ Warner Chilcott ($8. 5 bn) Teva/ Allergan generics ($38. 7 bn) Abb. Vie/ Pharmacycl ics ($20. 8 bn) Shire/ Baxalta ($32 bn) Pfizer/ Medivation ($14 bn) Abb. Vie/ Stemcentrx ($9. 8 bn) 323 259 600 500 400 300 247 200 160 50 100 0 0 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 Source: Evaluate. Pharma 31
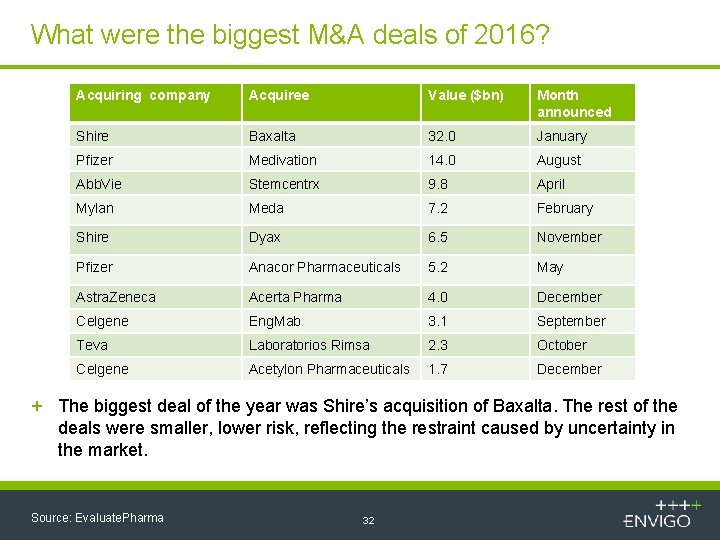
What were the biggest M&A deals of 2016? Acquiring company Acquiree Value ($bn) Month announced Shire Baxalta 32. 0 January Pfizer Medivation 14. 0 August Abb. Vie Stemcentrx 9. 8 April Mylan Meda 7. 2 February Shire Dyax 6. 5 November Pfizer Anacor Pharmaceuticals 5. 2 May Astra. Zeneca Acerta Pharma 4. 0 December Celgene Eng. Mab 3. 1 September Teva Laboratorios Rimsa 2. 3 October Celgene Acetylon Pharmaceuticals 1. 7 December + The biggest deal of the year was Shire’s acquisition of Baxalta. The rest of the deals were smaller, lower risk, reflecting the restraint caused by uncertainty in the market. Source: Evaluate. Pharma 32
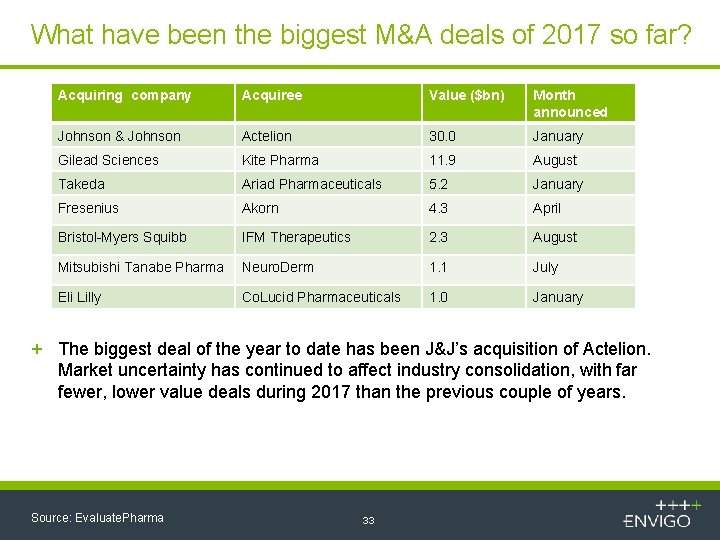
What have been the biggest M&A deals of 2017 so far? Acquiring company Acquiree Value ($bn) Month announced Johnson & Johnson Actelion 30. 0 January Gilead Sciences Kite Pharma 11. 9 August Takeda Ariad Pharmaceuticals 5. 2 January Fresenius Akorn 4. 3 April Bristol-Myers Squibb IFM Therapeutics 2. 3 August Mitsubishi Tanabe Pharma Neuro. Derm 1. 1 July Eli Lilly Co. Lucid Pharmaceuticals 1. 0 January + The biggest deal of the year to date has been J&J’s acquisition of Actelion. Market uncertainty has continued to affect industry consolidation, with far fewer, lower value deals during 2017 than the previous couple of years. Source: Evaluate. Pharma 33
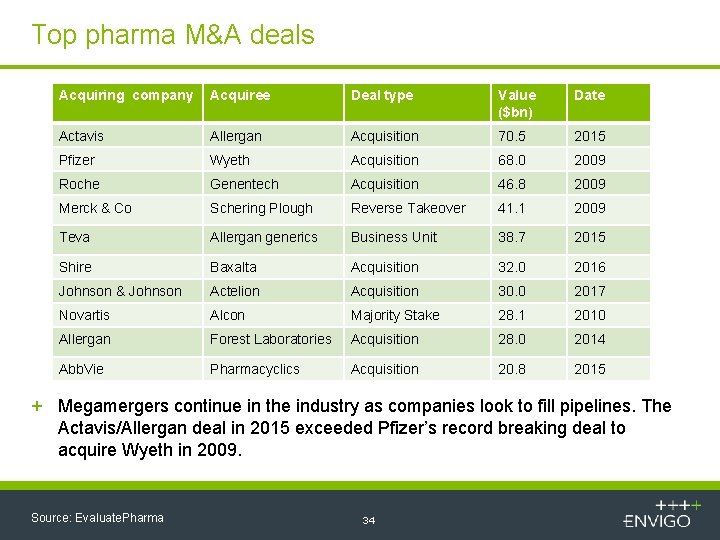
Top pharma M&A deals Acquiring company Acquiree Deal type Value ($bn) Date Actavis Allergan Acquisition 70. 5 2015 Pfizer Wyeth Acquisition 68. 0 2009 Roche Genentech Acquisition 46. 8 2009 Merck & Co Schering Plough Reverse Takeover 41. 1 2009 Teva Allergan generics Business Unit 38. 7 2015 Shire Baxalta Acquisition 32. 0 2016 Johnson & Johnson Actelion Acquisition 30. 0 2017 Novartis Alcon Majority Stake 28. 1 2010 Allergan Forest Laboratories Acquisition 28. 0 2014 Abb. Vie Pharmacyclics Acquisition 20. 8 2015 + Megamergers continue in the industry as companies look to fill pipelines. The Actavis/Allergan deal in 2015 exceeded Pfizer’s record breaking deal to acquire Wyeth in 2009. Source: Evaluate. Pharma 34

Market size & outsourcing trends 35
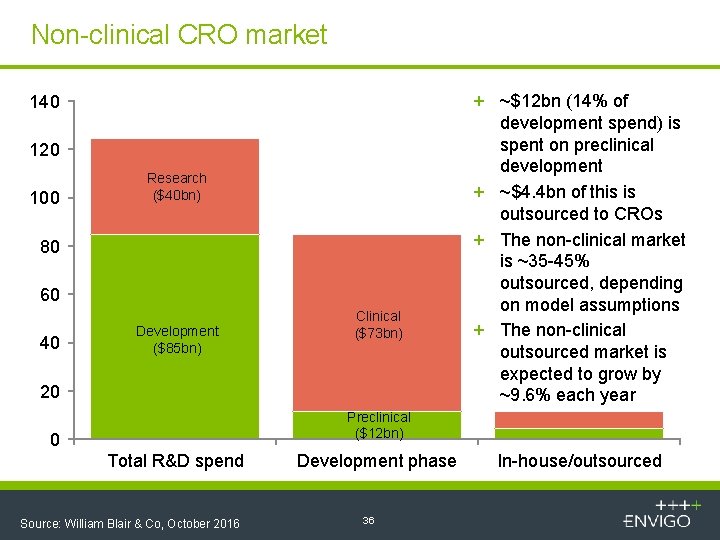
Non-clinical CRO market 140 120 100 Research ($40 bn) 80 60 40 Development ($85 bn) Clinical ($73 bn) 20 + ~$12 bn (14% of development spend) is spent on preclinical development + ~$4. 4 bn of this is outsourced to CROs + The non-clinical market is ~35 -45% outsourced, depending on model assumptions + The non-clinical outsourced market is expected to grow by ~9. 6% each year Preclinical ($12 bn) 0 Total R&D spend Source: William Blair & Co, October 2016 Development phase 36 In-house/outsourced
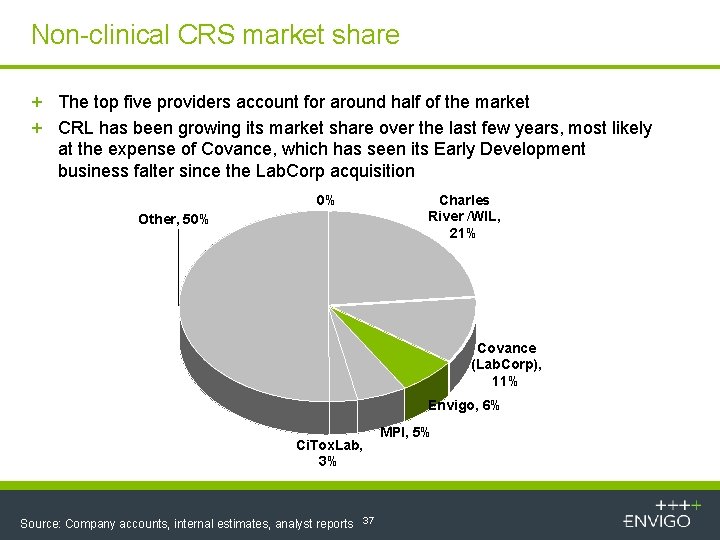
Non-clinical CRS market share + The top five providers account for around half of the market + CRL has been growing its market share over the last few years, most likely at the expense of Covance, which has seen its Early Development business falter since the Lab. Corp acquisition 0% Other, 50% Charles River /WIL, 21% Covance (Lab. Corp), 11% Envigo, 6% Ci. Tox. Lab, 3% Source: Company accounts, internal estimates, analyst reports 37 MPI, 5%
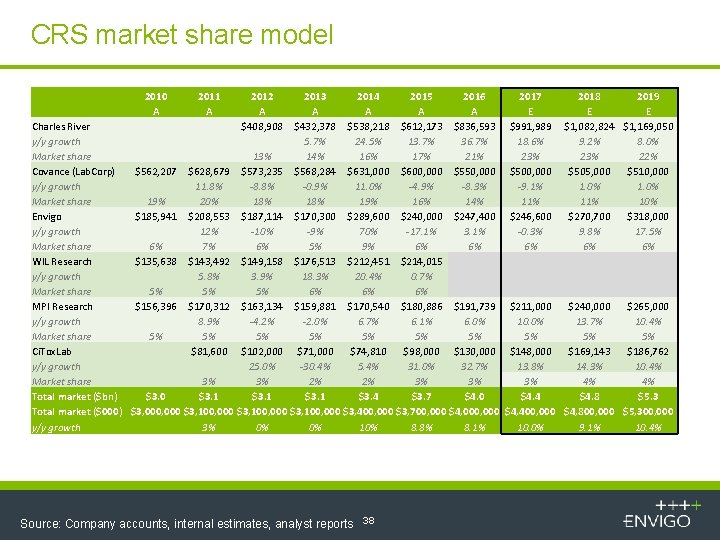
CRS market share model 2010 A 2011 A 2012 A $408, 908 2013 2014 2015 2016 A A Charles River $432, 378 $538, 218 $612, 173 $836, 593 y/y growth 5. 7% 24. 5% 13. 7% 36. 7% Market share 13% 14% 16% 17% 21% Covance (Lab. Corp) $562, 207 $628, 679 $573, 235 $568, 284 $631, 000 $600, 000 $550, 000 y/y growth 11. 8% -8. 8% -0. 9% 11. 0% -4. 9% -8. 3% Market share 19% 20% 18% 19% 16% 14% Envigo $185, 941 $208, 553 $187, 114 $170, 300 $289, 600 $240, 000 $247, 400 y/y growth 12% -10% -9% 70% -17. 1% 3. 1% Market share 6% 7% 6% 5% 9% 6% 6% WIL Research $135, 638 $143, 492 $149, 158 $176, 513 $212, 451 $214, 015 y/y growth 5. 8% 3. 9% 18. 3% 20. 4% 0. 7% Market share 5% 5% 5% 6% 6% 6% MPI Research $156, 396 $170, 312 $163, 134 $159, 881 $170, 540 $180, 886 $191, 739 y/y growth 8. 9% -4. 2% -2. 0% 6. 7% 6. 1% 6. 0% Market share 5% 5% Ci. Tox. Lab $81, 600 $102, 000 $71, 000 $74, 810 $98, 000 $130, 000 y/y growth 25. 0% -30. 4% 5. 4% 31. 0% 32. 7% Market share 3% 3% 2% 2% 3% 3% Total market ($bn) $3. 0 $3. 1 $3. 4 $3. 7 $4. 0 Total market ($000) $3, 000 $3, 100, 000 $3, 400, 000 $3, 700, 000 $4, 000 y/y growth 3% 0% 0% 10% 8. 8% 8. 1% Source: Company accounts, internal estimates, analyst reports 38 2017 E $991, 989 18. 6% 23% $500, 000 -9. 1% 11% $246, 600 -0. 3% 6% 2018 2019 E E $1, 082, 824 $1, 169, 050 9. 2% 8. 0% 23% 22% $505, 000 $510, 000 1. 0% 11% 10% $270, 700 $318, 000 9. 8% 17. 5% 6% 6% $211, 000 $240, 000 $265, 000 10. 0% 13. 7% 10. 4% 5% 5% 5% $148, 000 $169, 143 $186, 762 13. 8% 14. 3% 10. 4% 3% 4% 4% $4. 4 $4. 8 $5. 3 $4, 400, 000 $4, 800, 000 $5, 300, 000 10. 0% 9. 1% 10. 4%
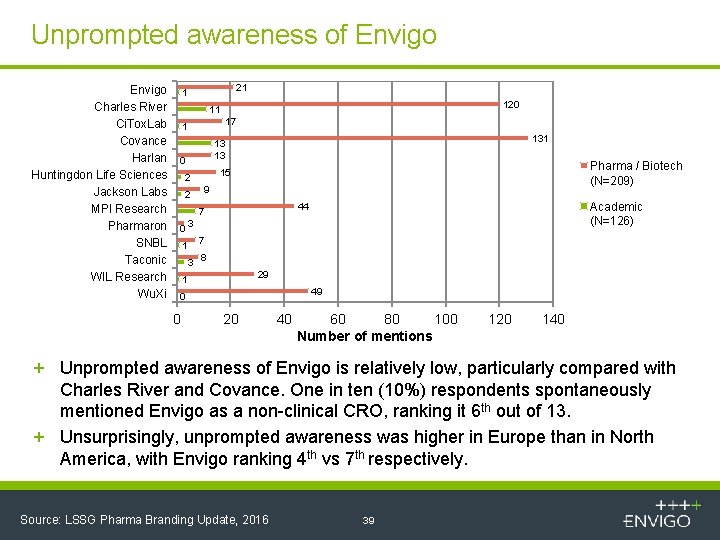
Unprompted awareness of Envigo Charles River Ci. Tox. Lab Covance Harlan Huntingdon Life Sciences Jackson Labs MPI Research Pharmaron SNBL Taconic WIL Research Wu. Xi 21 1 120 11 17 1 13 13 0 2 Pharma / Biotech (N=209) 15 2 9 Academic (N=126) 44 7 03 1 7 3 8 29 1 49 0 0 20 40 60 80 100 Number of mentions 120 140 + Unprompted awareness of Envigo is relatively low, particularly compared with Charles River and Covance. One in ten (10%) respondents spontaneously mentioned Envigo as a non-clinical CRO, ranking it 6 th out of 13. + Unsurprisingly, unprompted awareness was higher in Europe than in North America, with Envigo ranking 4 th vs 7 th respectively. Source: LSSG Pharma Branding Update, 2016 39
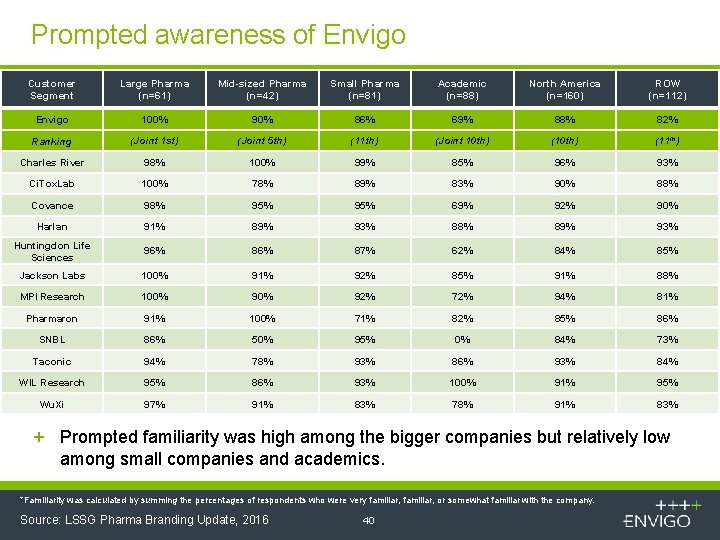
Prompted awareness of Envigo Customer Segment Large Pharma (n=61) Mid-sized Pharma (n=42) Small Pharma (n=81) Academic (n=88) North America (n=160) ROW (n=112) Envigo 100% 90% 86% 69% 88% 82% Ranking (Joint 1 st) (Joint 5 th) (11 th) (Joint 10 th) (11 th) Charles River 98% 100% 99% 85% 96% 93% Ci. Tox. Lab 100% 78% 89% 83% 90% 88% Covance 98% 95% 69% 92% 90% Harlan 91% 89% 93% 88% 89% 93% Huntingdon Life Sciences 96% 87% 62% 84% 85% Jackson Labs 100% 91% 92% 85% 91% 88% MPI Research 100% 92% 72% 94% 81% Pharmaron 91% 100% 71% 82% 85% 86% SNBL 86% 50% 95% 0% 84% 73% Taconic 94% 78% 93% 86% 93% 84% WIL Research 95% 86% 93% 100% 91% 95% Wu. Xi 97% 91% 83% 78% 91% 83% + Prompted familiarity was high among the bigger companies but relatively low among small companies and academics. *Familiarity was calculated by summing the percentages of respondents who were very familiar, or somewhat familiar with the company. Source: LSSG Pharma Branding Update, 2016 40
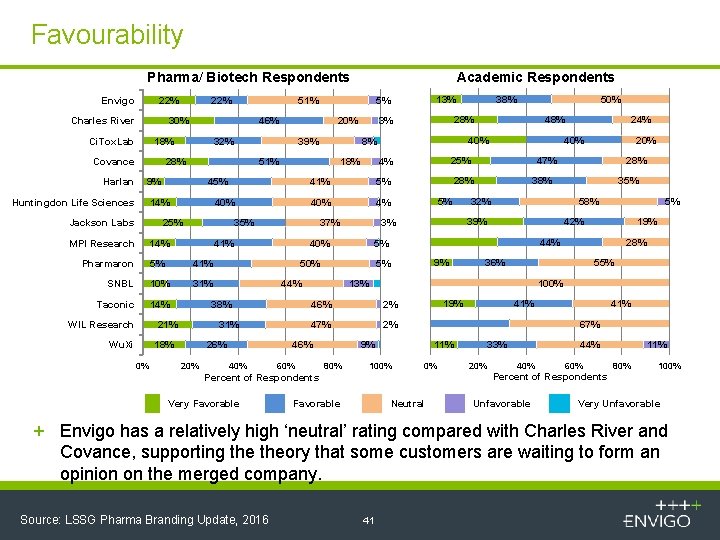
Favourability Pharma/ Biotech Respondents Envigo 22% Charles River 51% 30% Ci. Tox. Lab 39% 28% 45% 14% Jackson Labs 40% 25% MPI Research 41% 5% 41% SNBL 10% 31% Taconic 14% WIL Research 4% 26% 20% 58% 5% 42% 19% 9% 28% 36% 55% 100% 2% 47% 2% 19% 11% 100% 41% 67% 9% 80% 35% 44% 0% 33% 20% 44% 40% 60% 11% 80% 100% Percent of Respondents Very Favorable 32% 5% 46% 60% 28% 39% 3% 20% 47% 13% 46% 40% 5% 24% 40% 28% 5% 44% 31% 18% 0% 40% 50% 38% 21% Wu. Xi 5% 40% 48% 25% 4% 37% 50% 41% 35% 14% Pharmaron 18% 38% 28% 3% 8% 51% 9% Huntingdon Life Sciences 20% 32% 13% 5% 46% 18% Covance Harlan 22% Academic Respondents Favorable Neutral Unfavorable Very Unfavorable + Envigo has a relatively high ‘neutral’ rating compared with Charles River and Covance, supporting theory that some customers are waiting to form an opinion on the merged company. Source: LSSG Pharma Branding Update, 2016 41

Envigo favourability “New branding; waiting to see if company quality remains. ” -North America, Mid-Size Pharma/ Biotech “Uncertain about Harlan integration. ” -North America, Small Pharma/ Biotech “Problems are rare. And if there is a problem, they do everything to solve it. ” -Western Europe, Academic “Great marketing, lack of available research tools. ” -North America, Small Pharma/ Biotech “Good quality, good expertise, competitive. ” “Many animal models, expertise, fast and good interaction, and pro-active services. ” -Western Europe, Small Pharma/ Biotech “Work with extensively- close second to Covance Munster. ” -Western Europe, Large Pharma/ Biotech -North America, Large Pharma/ Biotech “Still not sure about the new integration and company name. ” -North America, Small Pharma/ Biotech “High level of GLP, timing respected, big catalogues of NC studies. ” -Western Europe, Large Pharma/ Biotech “Worldwide company with interesting animal models. ” -Western Europe, Academic N=54 + Respondents working with Envigo view the company favorably, however many are waiting to see how the new branding/integration affects the quality of service. Source: LSSG Pharma Branding Update, 2016 42
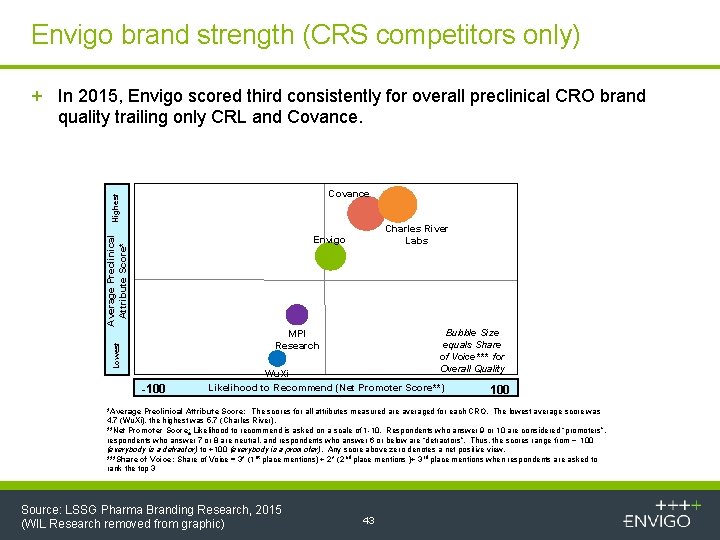
Envigo brand strength (CRS competitors only) + In 2015, Envigo scored third consistently for overall preclinical CRO brand quality trailing only CRL and Covance. Average Preclinical Attribute Score* Highest Covance Charles River Labs Envigo Bubble Size equals Share of Voice*** for Overall Quality Lowest MPI Research -100 Wu. Xi Likelihood to Recommend (Net Promoter Score**) 100 *Average Preclinical Attribute Score: The scores for all attributes measured are averaged for each CRO. The lowest average score was 4. 7 (Wu. Xi), the highest was 5. 7 (Charles River). **Net Promoter Score: Likelihood to recommend is asked on a scale of 1 -10. Respondents who answer 9 or 10 are considered “promoters”, respondents who answer 7 or 8 are neutral, and respondents who answer 6 or below are “detractors”. Thus, the scores range from − 100 (everybody is a detractor) to +100 (everybody is a promoter). Any score above zero denotes a net positive view. ***Share of Voice: Share of Voice = 3* (1 st place mentions) + 2* (2 nd place mentions )+ 3 rd place mentions when respondents are asked to rank the top 3 Source: LSSG Pharma Branding Research, 2015 (WIL Research removed from graphic) 43
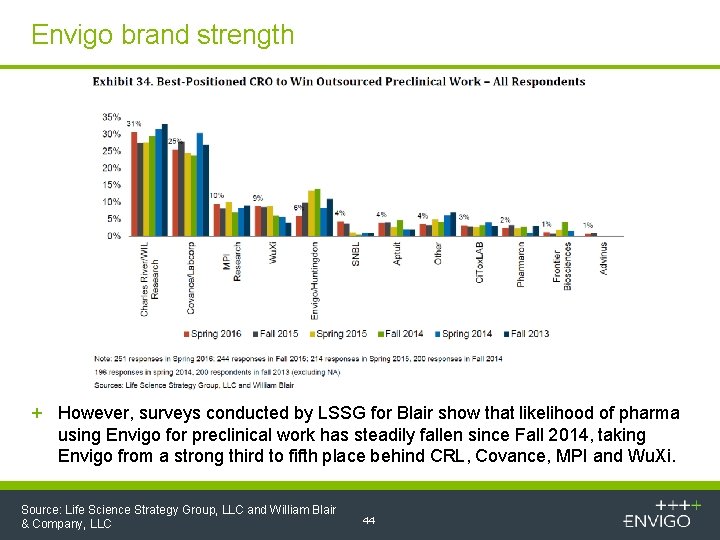
Envigo brand strength + However, surveys conducted by LSSG for Blair show that likelihood of pharma using Envigo for preclinical work has steadily fallen since Fall 2014, taking Envigo from a strong third to fifth place behind CRL, Covance, MPI and Wu. Xi. Source: Life Science Strategy Group, LLC and William Blair & Company, LLC 44
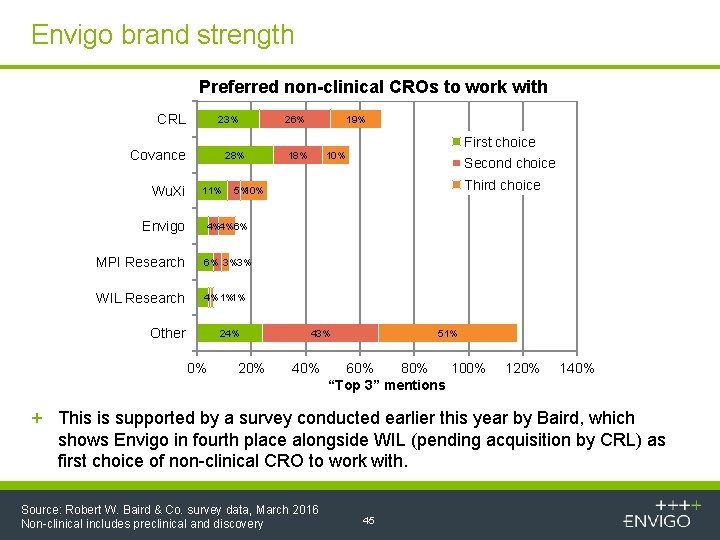
Envigo brand strength Preferred non-clinical CROs to work with CRL 23% Covance Wu. Xi Envigo 28% 11% 26% 19% 18% First choice 10% Second choice Third choice 5%10% 4%4%6% MPI Research 6% 3%3% WIL Research 4% 1%1% Other 24% 0% 20% 43% 40% 51% 60% 80% 100% “Top 3” mentions 120% 140% + This is supported by a survey conducted earlier this year by Baird, which shows Envigo in fourth place alongside WIL (pending acquisition by CRL) as first choice of non-clinical CRO to work with. Source: Robert W. Baird & Co. survey data, March 2016 Non-clinical includes preclinical and discovery 45
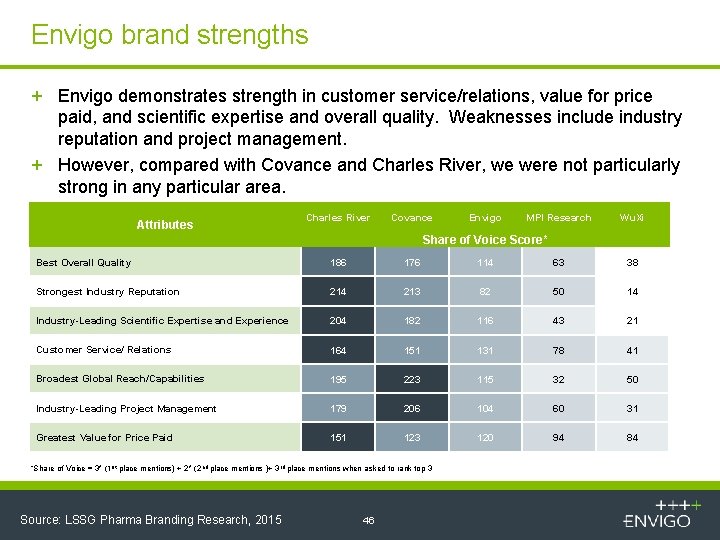
Envigo brand strengths + Envigo demonstrates strength in customer service/relations, value for price paid, and scientific expertise and overall quality. Weaknesses include industry reputation and project management. + However, compared with Covance and Charles River, we were not particularly strong in any particular area. Attributes Charles River Covance Envigo MPI Research Wu. Xi Share of Voice Score* Best Overall Quality 186 176 114 63 38 Strongest Industry Reputation 214 213 82 50 14 Industry-Leading Scientific Expertise and Experience 204 182 116 43 21 Customer Service/ Relations 164 151 131 78 41 Broadest Global Reach/Capabilities 195 223 115 32 50 Industry-Leading Project Management 179 206 104 60 31 Greatest Value for Price Paid 151 123 120 94 84 “Share of Voice = 3* (1 st place mentions) + 2* (2 nd place mentions )+ 3 rd place mentions when asked to rank top 3 Source: LSSG Pharma Branding Research, 2015 46
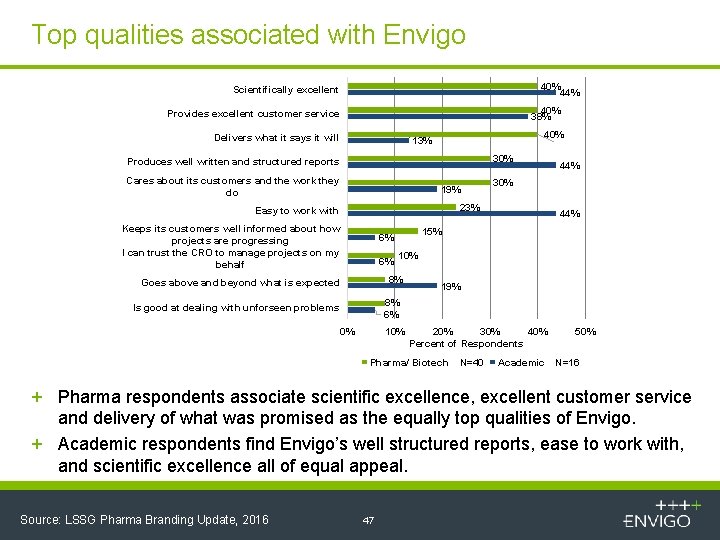
Top qualities associated with Envigo 40% 44% Scientifically excellent 40% 38% Provides excellent customer service Delivers what it says it will 40% 13% 30% Produces well written and structured reports Cares about its customers and the work they do 19% 30% 23% Easy to work with Keeps its customers well informed about how projects are progressing I can trust the CRO to manage projects on my behalf 10% 8% Goes above and beyond what is expected 44% 15% 6% 6% 44% 19% 8% 6% Is good at dealing with unforseen problems 0% 10% 20% 30% 40% Percent of Respondents Pharma/ Biotech N=40 Academic 50% N=16 + Pharma respondents associate scientific excellence, excellent customer service and delivery of what was promised as the equally top qualities of Envigo. + Academic respondents find Envigo’s well structured reports, ease to work with, and scientific excellence all of equal appeal. Source: LSSG Pharma Branding Update, 2016 47
- Slides: 47