Standards for Drug Labels Medical information standards in

Standards for Drug Labels Medical information standards in support of SPL 03 October 2020

Standards within standards within… 2

What sort of standards exist? • “Quality” standards • criteria against which accomplishments are judged. • standards forensics competition • “Reference” standards • to assure that what I hear you say is what you meant to tell me. • to align communications 3

Judgment standards • Tolerance standards • “This tablet contains 40 ± 2 mg of esomeprazole” • AAHA Certification standards • “All AAHA certified hospitals will have an EKG machine” • The 10 Commandments • “Though shall not steal” 4

Reference standards • USP Drug Standards • Known concentration and purity • Information Standards • General: XML, HTML, etc. • Medical: HL 7, SNOMED, NCI Thesaurus… • Rosetta Stone • demotic ↔ hieroglyphic ↔ Greek 5

Standards enhance communication • Common “language” between users and systems • Community consensus • Collaboration • Are we interoperable? 6

SPL is built on standards Message • XML • HL 7 • CDA Architecture 7 Terminology • LOINC • SNOMED-CT • NCI Thesaurus • UCUM • NDC • Etc.

XML standardizes the mechanics of the SPL Hierarchical Validate structure against schema <dose. Form> <id>55325</id> <name>solution</name> Values embedded Tags declare </dose. Form> in or nested within attributes tags <dose. Form id=“ 55325” name=“solution”/> 8 Can include any digital artifact
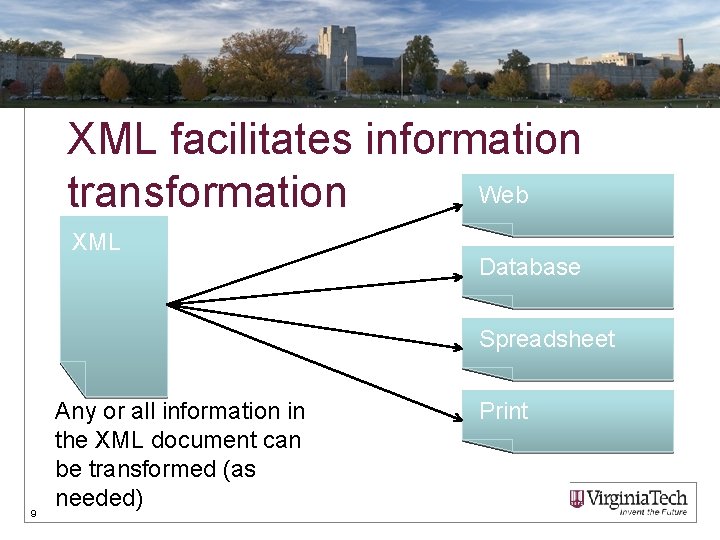
XML facilitates information Web transformation XML Database Spreadsheet 9 Any or all information in the XML document can be transformed (as needed) Print

XML Schema describes structure of XML document • Specify what is required within the XML document they support • Process specific documents against a schema • Document is “valid” (schema-valid) or “invalid” for its type 10

Attributes (qualities) and values Columns = attributes Rows = values DOSE FORM ROUTE Solution Intravenous Suspension Intramuscular Tablet Oral Document / message standards tend to deal with attributes Vocabulary / terminology standards tend to deal with values 11

Attributes (qualities) and values (in XML) <dose. Form id=“ 55325” name=“solution” /> 2 <dose. Form> <id>55325</id> <name>solution</name> </dose. Form> 12 1

HL 7 • • Health Level 7 international Open standard Member only access Dominated by corporate and government membership • Funded through membership fees • www. hl 7. org 13

HL 7 (why 7? ) International Organization for standards (ISO) Organisation internationale de normalisation 14

HL 7 • Establishes semantics • Specifies the structure of messages • Consensus-based (balloted) development of CDA architecture • Determines the SPL schema 15

HL 7 • Version 2. x establishes attribute lists, a consistent syntax, and a “pipe-delimited” format • Version 3. x establishes an underlying information model, maintains a consistent syntax and uses XML • document/message schema, attribute definitions and components, uses 16

Clinical Document Architecture (CDA) • An XML-based markup standard • specify the encoding, structure and semantics of clinical documents for exchange. • HL 7 version 3 standard • based on the HL 7 Reference Information Model (RIM) • HL 7 Version 3 Data Types. CDA documents are persistent in nature. 17

Clinical Document Architecture (CDA) • Mandatory textual part (for human interpretation of the document contents) • Optional structured parts (for software processing). • Structured parts rely on coding systems to represent concepts. 18

Multiple Terminologies • • 19 Content varies Mechanics vary Management varies “Politics” and path to new modification or addition vary

Vocabulary / terminology standards • Dun and Bradstreet D-U-N-S number • National Cancer Institute (NCI) Thesaurus • FDA Substance Registration System (SRS) – Unique Ingredient Identifier (UNII) • Logical Observation Identifiers Names and Codes (LOINC) • ISO 3166 -1 Alpha-3 Country Code (ISO 3166 -1) • Unified Code for Units of Measure (UCUM) • Health Level Seven • Veterans Administration / Kaiser Permanente Problem List Subset of SNOMED-CT (VA/KP Subset) 20

Vocabulary / terminology standards • Dept. of Veterans Affairs National Drug File Reference Terminology (NDF-RT) • National Drug Code (NDC) System • FDA Firm Establishment Identifier (FEI) • FDA Application Number • Globally Unique Identifier (GUID) • US Postal Service • Code Object Identifier (OID) • Local company terminology • ISBT-128 site and product codes for licensed minimally manipulated cell products 21

Questions: 22

D-U-N-S number • Dun and Bradstreet (http: //www. dnb. com) • A unique nine-digit number that tells the entire world you mean business • Global commercial database with 140 million business records • Obtain one by asking for a listing 23

NCI Thesaurus • A collection of terminologies that were developed to support research studies • • 24 National Cancer Institute Stable, unique alphanumeric code Synonyms, definitions, external link Available for browsing and download from their website

SRS – UNII • FDA Substance Registration System – Unique Ingredient Identifier • Ingredient, moiety, reference drug • Managed by SRS Board • Unique ten character alphanumeric identifier • Request additions through email 25

LOINC • Logical Observation Identifiers Names and Codes • • • 26 Lab tests, other clinical observations Managed by the Regenstrief Institute 3 to 7 character codes Download Access database or text file New search feature on website Released twice a year

ISO 3166 -1 • International Organization for Standardization (acronym is French) • Standard country names and code elements • Download Access database or text file list of codes • Updates when the UN updates its list • Changes in newsletter 27

UCUM • Unified Code for Units of Measure • Units of measure used in science, engineering and business • >250 unit symbols • Maintained by Regenstrief Institute • HTML and XML versions • Few requests needed 28

HL 7 • Health Level 7 • Structure and some vocabulary 29

VA/KP Problem list subset of SNOMED-CT • Veterans Administration / Kaiser Permanente subset of the Systematized Nomenclature of Medicine – Clinical Terms • ~17, 000 SNOMED concepts • Download from NCI Vocabulary Services 30

NDF-RT • Department of Veterans Affairs National Drug File Reference Terminology (NDFRT) • Ingredients, chemical structure, dose form, physiologic effect, mechanism of action, pharmacokinetics, and related diseases. • Updated monthly 31

NDC • • • 32 National Drug Code System Managed by FDA Universal product identifier for drugs Unique 10 -digit, three-segment number Can request an NDC code through SPL

FEI • • 33 FDA Firm Establishment Identifier Assigned to a facility Managed by FDA Up to 10 digits

(FDA) Application Number • NDA/ANDA or NADA/ANADA • Product approved for marketing by FDA 34

GUID • Globally Unique Identifier • Unique number generated by software • 32 character hexadecimal string • Example: 21 EC 2020 -3 AEA-1069 -A 2 DD 08002 B 30309 D 35

United States Postal Service • Official USPS state abbreviations • States plus 9 possessions/associated states and 6 military states • Full name and two character code • Codes available on USPS website 36

Code System Object Identifiers • OID - globally unique string representing an ISO Object Identifier • Identifies the coding (terminology) system from which a code is derived • Consists of numbers and dots (e. g. , 2. 16. 840. 1. 113883. 3. 1) • Lacks single universal management structure 37
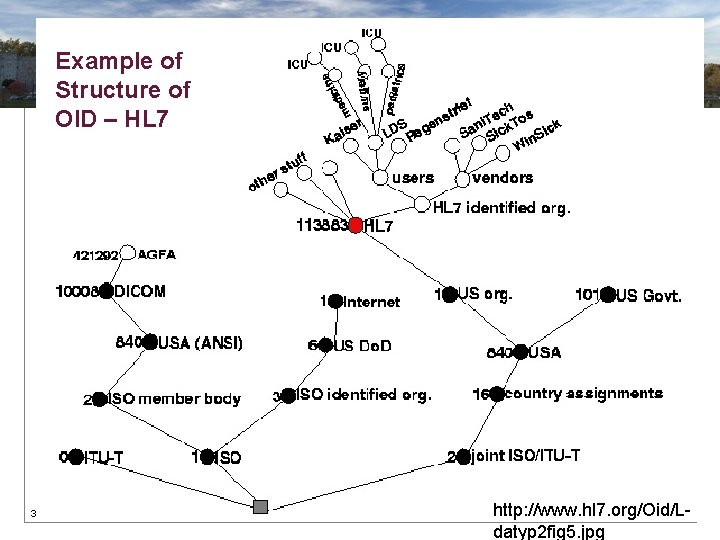
Example of Structure of OID – HL 7 38 http: //www. hl 7. org/Oid/Ldatyp 2 fig 5. jpg

39

Local terminology • Device • Model • Lot • Serial number 40

ISBT - 128 • International Society of Blood Transfusion • Now managed through the International Council for Commonality in Blood Banking Automation (ICCBBA) • American Association of Blood Banks • System for identification, labeling and processing of human blood, tissue, cellular therapy products. 41

Reference standards support SPL • Share some commonalities • Some overlap • Vary in management, content, and application • Multiple users • Reuse within and outside FDA 42

FDA information management “sins” • Overloaded value fields • Misnamed value “fields” • There are obvious pragmatic reasons overloading has occurred with time • Alignment with terminology and message standard becomes difficult 43

Overloaded “approved species” field • “Dairy replacement heifers growing on pasture” • • • 44 A common animal group (cattle) + Animal use classification (dairy) + Age range / repro status (heifer) + Sex / repro status (heifer) + Management practice (pasture) + Future use (replacement)

Overloaded routes of administration • Intramuscular is a route • “Intramuscular in neck” • Is route + site • “Topical with occlusive dressing” • Route + administration procedure 45

Mislabeled value field • Dose form • Solution in the bottle, administered intravenous as a solution then: • Dose form = “form of product” • Sterile powder for reconstitution as solution then: • Dose form ≠ “form of product” 46

Vocabulary drift • “bacterial pneumonia (shipping fever complex) caused by P. multocida susceptible to oxytetracycline” • Terramycin Soluble Powder (008 -622) • For the treatment of bacterial pneumonia (shipping fever) caused by Pasteurella multocida • Procaine Pen G (065 -010) 47

Vocabulary drift • “shipping fever, calf pneumonia and bovine pneumonia” • 055 -030 (Polyflex) • Bovine respiratory disease (BRD) • 140 -529 (Micotil) • 141 -244 (Draxxin) 48

Adaption to standards at CVM • Some of the historical and current practices may need to change to align with standards 49

SPL recipe • • • 50 1 part HL 7 CDA 1 part LOINC 2 parts SNOMED 4 parts NCI Thesaurus A dash of UCUM…
- Slides: 50