Stable Free Radical Polymerization Peter Kazmaier Manager New

Stable Free Radical Polymerization Peter Kazmaier Manager New Materials Design CH 3 N O TEMPO CH 3

Agenda n Introduction n SFRP Chemistry and it’s Development History n SFRP Monomers and Process Latitude n Scale-Up n Applications n Conclusions and Wrap-Up n Q&A

Introduction to Stable Free Radical Polymerization n A New Radical Polymerization Process that Allows the Preparation CH 3 of Polymers with Controlled Architecture n Block copolymers n Comb shape polymers n Brush type polymers n Reactive polymers CH 3 N O TEMPO CH 3

History of SFRP Development in Xerox n Identified need to develop a novel class of polymers with improved performance in 1990’s. n n n Polymer architecture plays a key role in polymer properties along with monomer choice as they pertained to imaging materials. Anionic polymerization (AP) Issues n The AP process is too expensive for many applications. n Incompatible with many functional monomers not compatible with AP Consequently, Xerox launched a research effort to control polymer architecture. n Resulted in a free radical process called SFRP

What is SFRP Conventional Radical Polymerization Initiator + Monomer Polymer - Broad polydispersities - Poor Mw control Stable Free Radical Polymerization Initiator + Monomer + Nitroxide I-P-N - Narrow polydispersities - Good Mw control
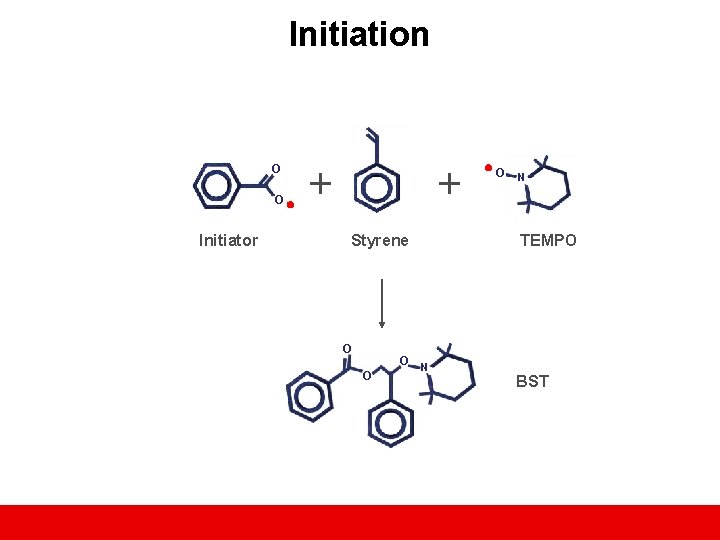
Initiation O O Initiator + + Styrene O O N TEMPO N BST
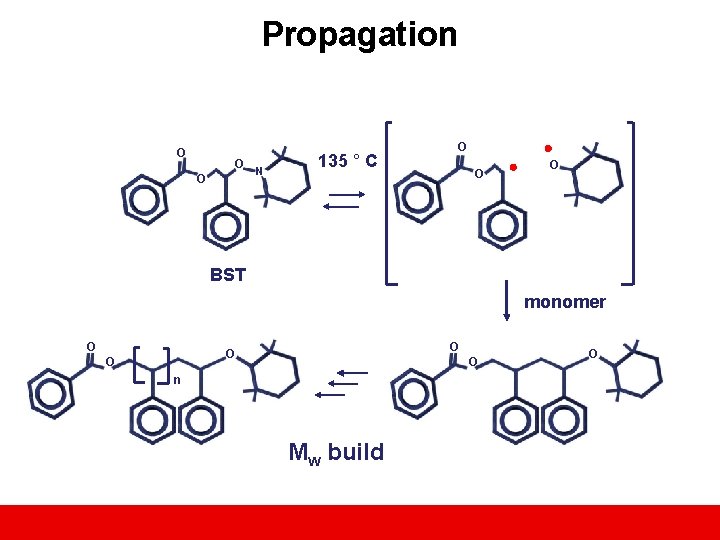
Propagation O O O N 135 ° C O O O BST monomer O O O n Mw build O

Conventional Free Radical Polymerization vs SFRP Gives Control of Molecular Weight Build and PD Over Time Conventional Mn Time SFRP Increasing Molecular Weight
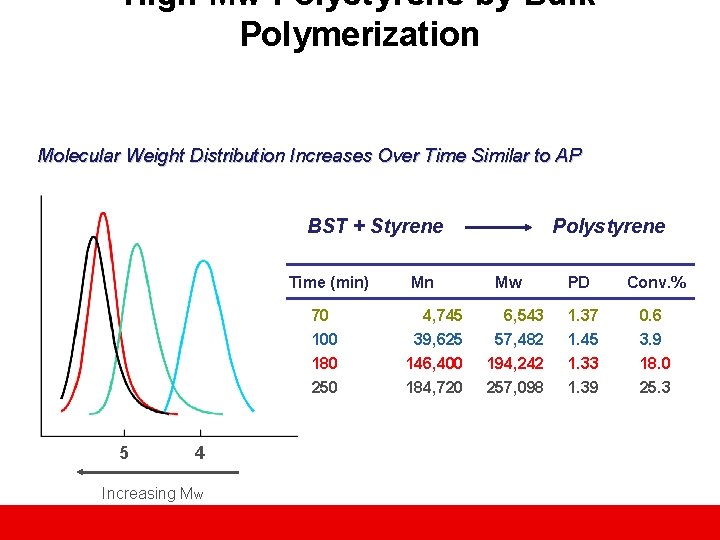
High Mw Polystyrene by Bulk Polymerization Molecular Weight Distribution Increases Over Time Similar to AP BST + Styrene Time (min) 70 100 180 250 5 4 Increasing Mw Mn 4, 745 39, 625 146, 400 184, 720 Polystyrene Mw 6, 543 57, 482 194, 242 257, 098 PD Conv. % 1. 37 1. 45 1. 33 1. 39 0. 6 3. 9 18. 0 25. 3

Blocks by SFRP Reactive end used to add new monomers: n At room temperature free radical is completely stable. I n At elevated temperature in the presence of a second monomer one can then build block-copolymers I n N N And in the presence of a third monomer or the original one can then build block-Ter-polymers. I N
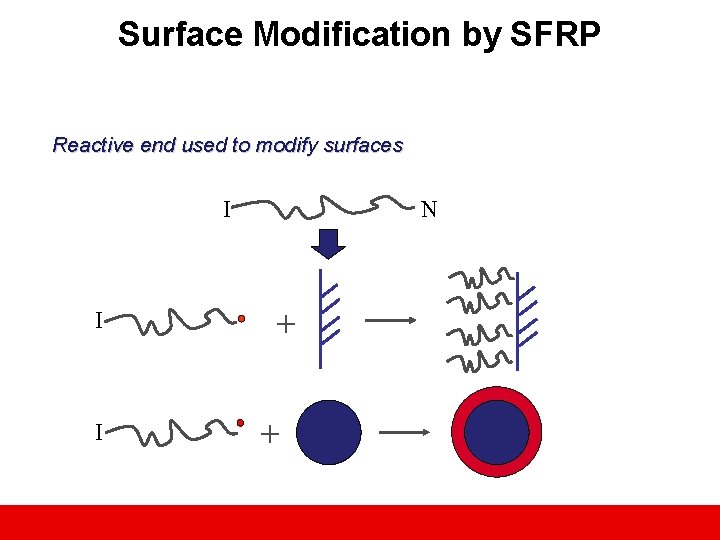
Surface Modification by SFRP Reactive end used to modify surfaces I I I N + +

Unique Aspects of SFRP Styrene / Maleic Anhydride Polymerization O O + O Excess O O Conventional: Three types of polymer with poor blocks • P(S/MA) • P(S/MA-b-S) poor block • P(S) SFRP: Only one polymer with good block • P(S/MA-b-S) O
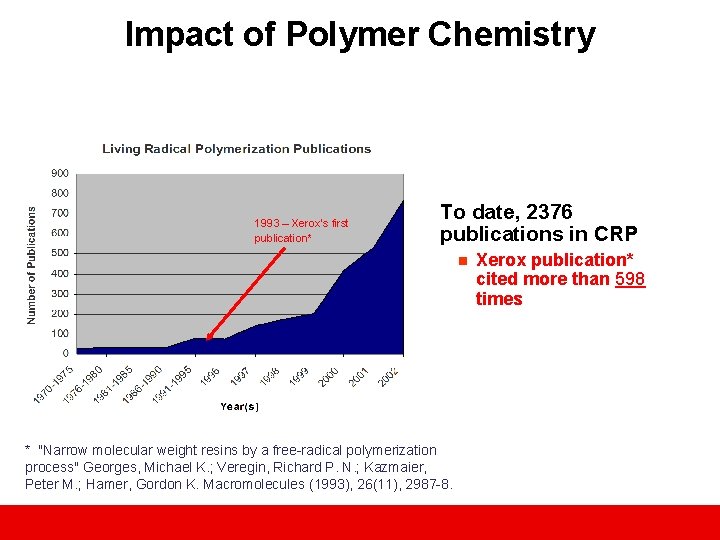
Impact of Polymer Chemistry 1993 – Xerox’s first publication* To date, 2376 publications in CRP n * "Narrow molecular weight resins by a free-radical polymerization process" Georges, Michael K. ; Veregin, Richard P. N. ; Kazmaier, Peter M. ; Hamer, Gordon K. Macromolecules (1993), 26(11), 2987 -8. Xerox publication* cited more than 598 times

Monomer Range Styrenics Alkylacrylates Dienes CH 2=CH C=C COOX X H SO 3 Na X= CH 2 Cl COOH X= Bu t-Bu Butadiene and Isoprene

Polymerization Range n Bulk Polymerization n n Random Copolymers of: n styrene/acrylates including acrylic acid n styrene/dienes n styrene/acrylonitrile Block copolymers of: n styrene-b-acrylates n styrene-b-dienes

Misconceptions of SFRP n External Literature Indicates SFRP: n Has long reactions times upwards of 36 hours n Has low conversions in bulk polymerization of 50% n Is not a scalable process n Has low loadings (~10%) in emulsion polymerizations with bimodal distributions and significant fouling
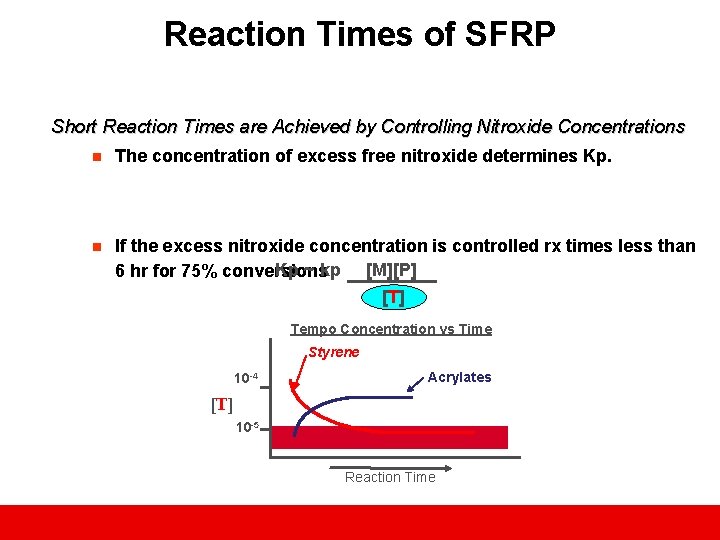
Reaction Times of SFRP Short Reaction Times are Achieved by Controlling Nitroxide Concentrations n The concentration of excess free nitroxide determines Kp. n If the excess nitroxide concentration is controlled rx times less than Kp ~ kp [M][P] 6 hr for 75% conversions [T] Tempo Concentration vs Time Styrene 10 -4 Acrylates [T] 10 -5 Reaction Time

Conversions of SFRP n Initial research utilized bulk polymerization: n Conversions limited to 75% n Bulk polymerization conversion rates still not high enough for commercialization n Led to consideration of other polymerization processes for SFRP

Scalability of SFRP n To commercialize, a higher conversion AND scalable process was required. n 3 options investigated: n Solution n Suspension n Emulsion – the preferred process! n environmentally better n most widely applicable n many existing commercial reactors

Emulsion Polymerization: Conventional vs SFRP Polymerization Conventional Free Features Radical Emulsion SFRP Initiation Continuous Over in <5 min Polymer chains High mol. wt Oligomeric at beginning Temperature 70 o. C 135 o. C Nitroxide level None Required Auto-initiation Non-issue Occurs but controllable Ability to make block copolymers Not possible “Holy Grail”- We can make block copolymers

Emulsion or Mini-Emulsion n Emulsion Initiation Stage n The nitroxide is asked to play too many roles n Too complicated - Mini-Emulsion is easier
SFRP by Mini-Emulsion-Initiation Stage n In Mini-Emulsion Reactions, the Nitroxide has only One Role n IM M Control the Polymerization T IM T M I M M M T M IM M M I Mn T M T M I Mn T M IM T I Mn T I M T I IM I Mn M
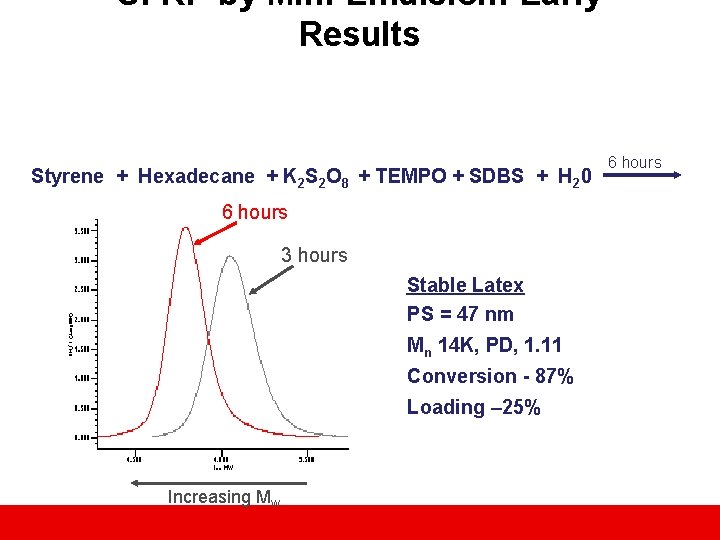
SFRP by Mini-Emulsion: Early Results Styrene + Hexadecane + K 2 S 2 O 8 + TEMPO + SDBS + H 20 6 hours 3 hours Stable Latex PS = 47 nm Mn 14 K, PD, 1. 11 Conversion - 87% Loading – 25% Increasing Mw 6 hours
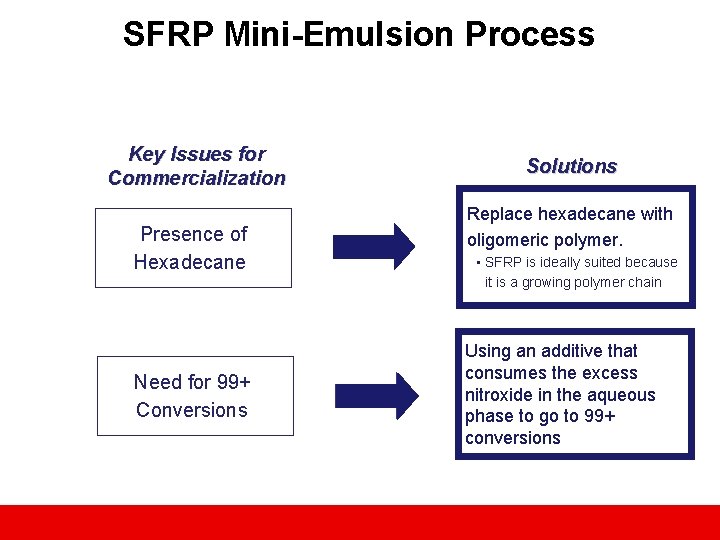
SFRP Mini-Emulsion Process Key Issues for Commercialization Presence of Hexadecane Need for 99+ Conversions Solutions Replace hexadecane with oligomeric polymer. • SFRP is ideally suited because it is a growing polymer chain Using an additive that consumes the excess nitroxide in the aqueous phase to go to 99+ conversions
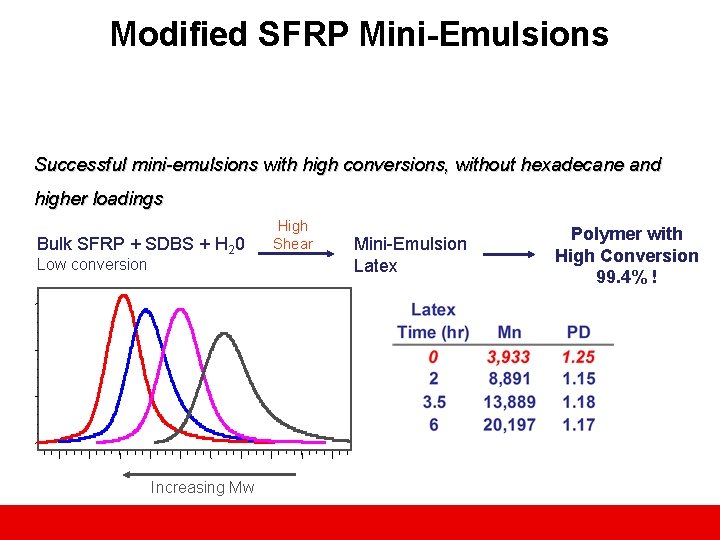
Modified SFRP Mini-Emulsions Successful mini-emulsions with high conversions, without hexadecane and higher loadings Bulk SFRP + SDBS + H 20 Low conversion Increasing Mw High Shear Mini-Emulsion Latex Polymer with High Conversion 99. 4% !

Scale-Up of Modified SFRP by Mini. Emulsion Lab Scale Factors Scale Down 5 Gallon 100 Gallon Heating Rates Stirring Mechanical Processes Raw Materials Conversions

Scale-Up Equipment 5 Gallon Reactor

Scale-Up Poly (Styrene / Acrylate) Bulk SFRP Consistency of Reaction 2. 50 2. 00 1. 50 1. 00 0. 50 0. 00 dwt/d(log. M) 2. 50 3. 80 3. 60 3. 40 3. 20 Slice Log MW 3. 00 2. 80
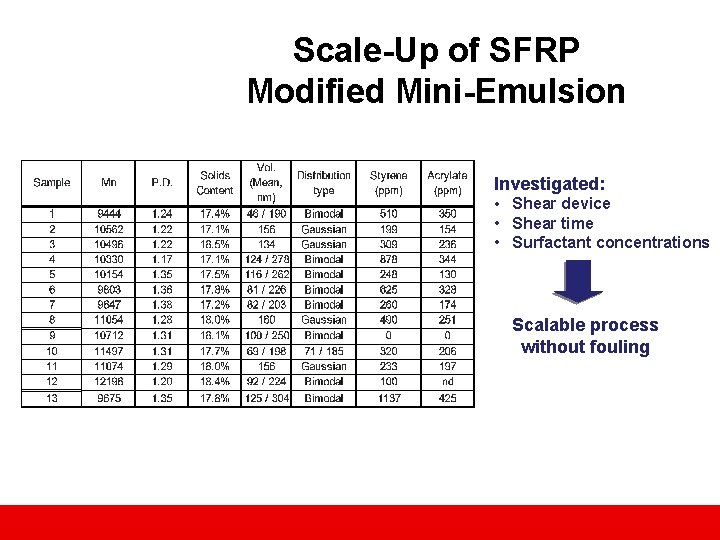
Scale-Up of SFRP Modified Mini-Emulsion Investigated: • Shear device • Shear time • Surfactant concentrations Scalable process without fouling

Applications: SFRP as Dispersants Reactive Polymer Mode Hypothesis: Modified carbon black would be dispersible in water and jettable in ink jet head. + PSST Heat TEMPO

Preparation of Carbon-Black Dispersions Carbon Black + PSST + Solvent 1) Attritor EG/H 2 O 130 °C Dispersion in water/EG 2) Microfluidizer H 2 O 130 °C Dispersion in water only
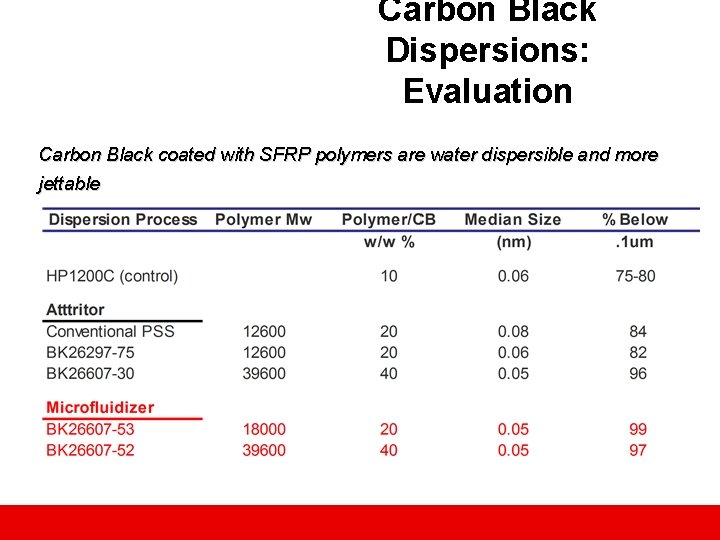
Carbon Black Dispersions: Evaluation Carbon Black coated with SFRP polymers are water dispersible and more jettable

Conclusion n Xerox’s SFRP Process: n Allows the preparation of a wider range of materials on an industrial scale compared to other free radical processes n Produces living polymers with complex architecture n Enables the manufacture of polymers with narrow molecular weight distributions n Is a scalable modified mini-emulsion SFRP process n n Demonstrated at the 5 gallon scale with stable latexes Is protected by over 35 US patents n Numerous Foreign Filings
Xerox SFRP Patents Applicable US Patents 5364723 (1993) 5266439 5346790 5322912 5852140 5530079 6258911 5773510 5449724 5545504 5412047 5498679 5608023 5739229 5932630 5728747 5723511 6075105 5817824 5744560 6156858 6121397 5891971 5401804 5487847 5529719 5549998 5552502 5575954 5610250 5714993 5907001 5919861 6087451 6320007 6114499 (2003) Xerox’s SFRP technology – patents and associated know how – is available for license

Applications n Adhesives n Viscosity modifiers n Particle formation n Resin rheology control n Compatibilizers

Value n Ciba license in place n Other companies have expressed interest and engaged XRCC n Miniemulsion has been excluded from licenses so far

- Slides: 37