StabilityActivity Tradeoffs Proximate vs Ultimate Causes Jeffrey Endelman

Stability-Activity Tradeoffs: Proximate vs. Ultimate Causes Jeffrey Endelman University of California, Santa Barbara

Causation in Biology • Proximate (physicochemical) • Ultimate (evolutionary) Mayr, E. (1997) This is Biology. Cambridge: Harvard Univ. Press.
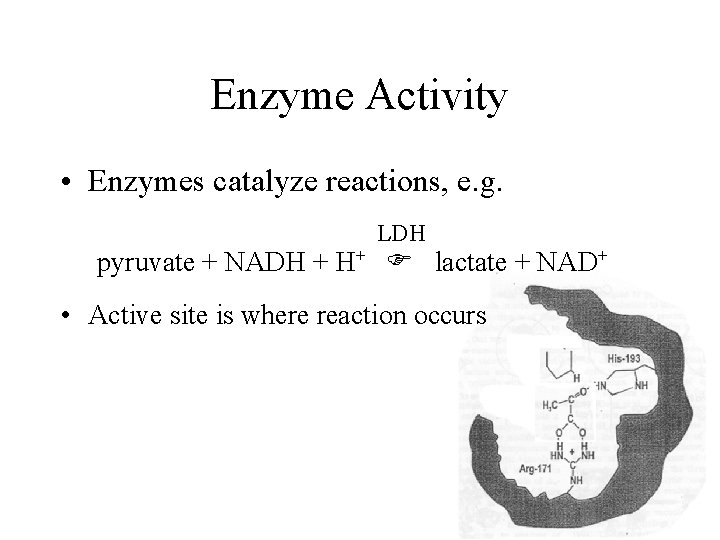
Enzyme Activity • Enzymes catalyze reactions, e. g. LDH pyruvate + NADH + H+ lactate + NAD+ • Active site is where reaction occurs
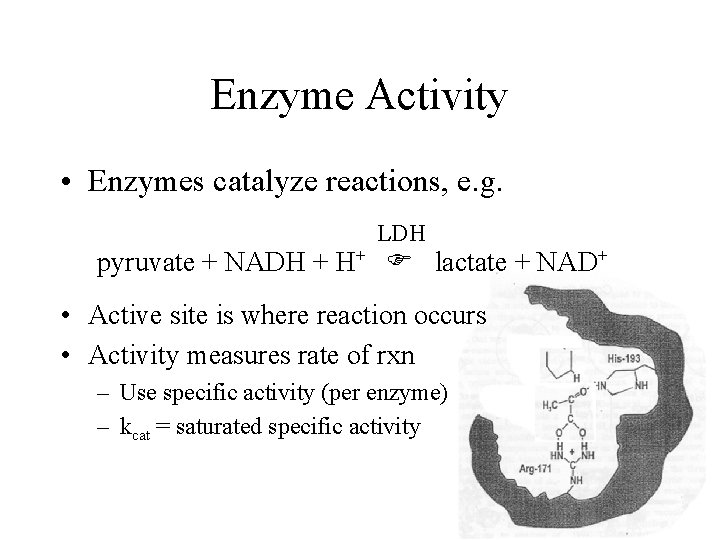
Enzyme Activity • Enzymes catalyze reactions, e. g. LDH pyruvate + NADH + H+ lactate + NAD+ • Active site is where reaction occurs • Activity measures rate of rxn – Use specific activity (per enzyme) – kcat = saturated specific activity
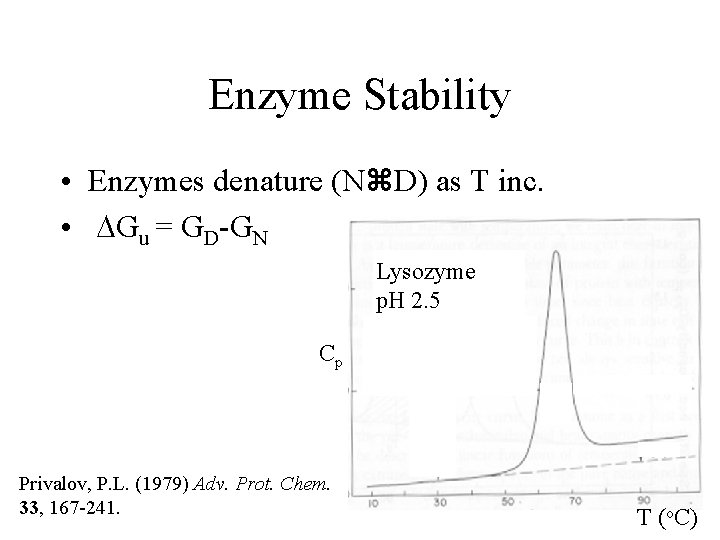
Enzyme Stability • Enzymes denature (N D) as T inc. • DGu = GD-GN Lysozyme p. H 2. 5 Cp Privalov, P. L. (1979) Adv. Prot. Chem. 33, 167 -241. T (o. C)
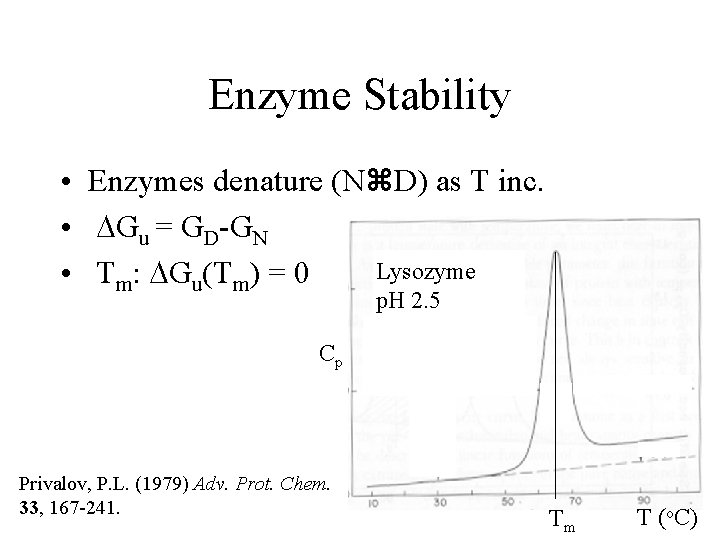
Enzyme Stability • Enzymes denature (N D) as T inc. • DGu = GD-GN Lysozyme • Tm: DGu(Tm) = 0 p. H 2. 5 Cp Privalov, P. L. (1979) Adv. Prot. Chem. 33, 167 -241. Tm T (o. C)
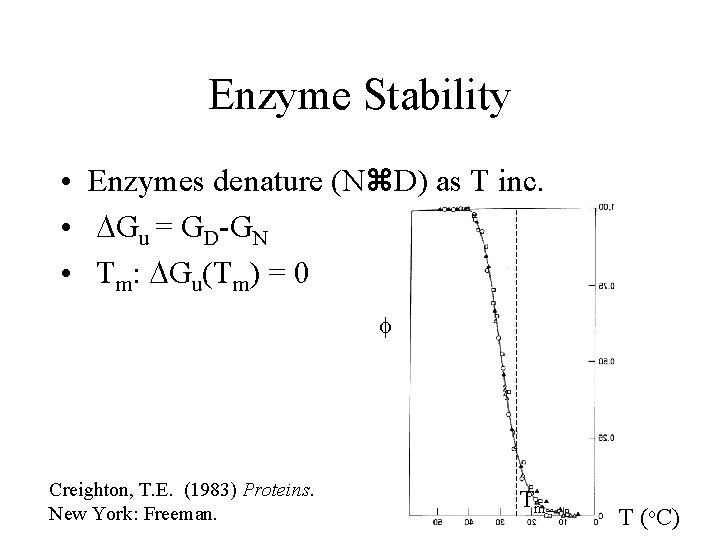
Enzyme Stability • Enzymes denature (N D) as T inc. • DGu = GD-GN • Tm: DGu(Tm) = 0 f Creighton, T. E. (1983) Proteins. New York: Freeman. Tm T (o. C)

Enzyme Stability • • Enzymes denature (N D) as T inc. DGu = GD-GN Tm: DGu(Tm) = 0 Residual activity (Ar /Ai)
Wintrode, P. L & Arnold, F. H. (2001) Adv. Prot. Chem. 55, 161 -225.
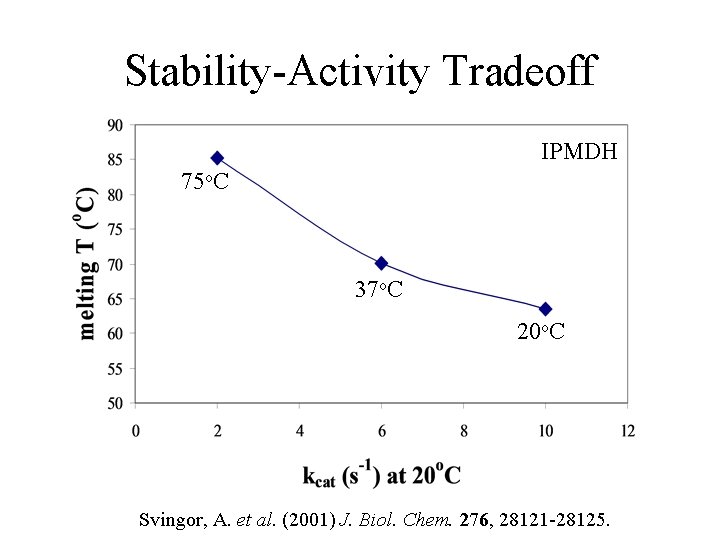
Stability-Activity Tradeoff IPMDH 75 o. C 37 o. C 20 o. C Svingor, A. et al. (2001) J. Biol. Chem. 276, 28121 -28125.
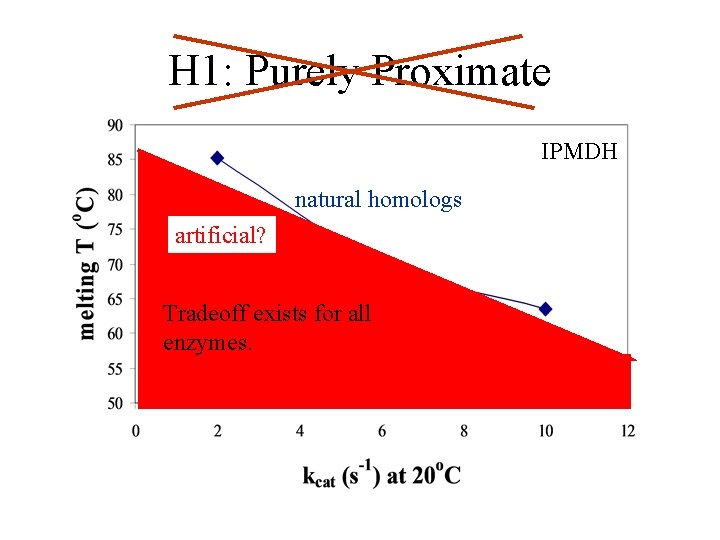
H 1: Purely Proximate IPMDH natural homologs artificial? Tradeoff exists for all enzymes.
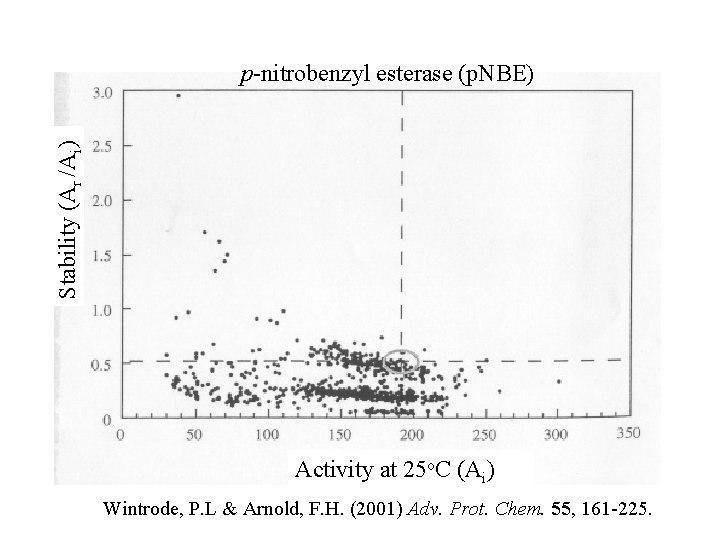
Stability (Ar /Ai) p-nitrobenzyl esterase (p. NBE) Activity at 25 o. C (Ai) Wintrode, P. L & Arnold, F. H. (2001) Adv. Prot. Chem. 55, 161 -225.
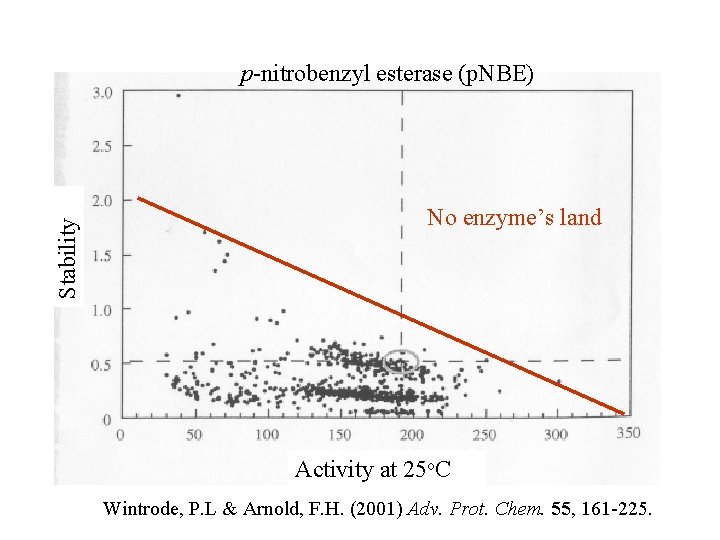
Stability p-nitrobenzyl esterase (p. NBE) No enzyme’s land Activity at 25 o. C Wintrode, P. L & Arnold, F. H. (2001) Adv. Prot. Chem. 55, 161 -225.

S/A Tradeoff Hypotheses 1. All enzymes have proximate tradeoff 2. Ultimate: Selection for high S&A Proximate: Highly optimized enzymes have S/A tradeoff

Proximate Tradeoff: Flexibility • Enzymes achieve greater stability by reducing flexibility. • Flexible motions are important for catalysis in many enzymes. • Thus thermostability through reduced flexibility decreases activity. Somero, G. N. (1995) Annu. Rev. Physiol. 57, 43 -68.

Flexibility & Activity • Large motions (hinge bending, shear) – Pyruvate dehydrogenase – Triosephosphate isomerase – Lactate dehydrogenase – Hexokinase • Small motions (vibrational, breathing, internal rotations) – No evidence, but not unlikely Fersht, A. (1999) Structure and Mechanism in Protein Science. New York: Freeman.

Proximate Tradeoff: Flexibility • Enzymes achieve greater stability by reducing flexibility. • Flexible motions are important for catalysis in many enzymes. • Thus thermostability through reduced flexibility decreases activity. Somero, G. N. (1995) Annu. Rev. Physiol. 57, 43 -68.

Flexibility & Stability • Stabilization involves all levels of protein structure • Experiments typically probe small motions via amide hydrogen exchange • Some thermophiles are more rigid than mesophile, others are not • “. . . hypothesis [that] enhanced thermal stability … [is] the result of enhanced conformational ridigity…. has no general validity. ” Jaenicke, R. (2000) PNAS 97, 2962 -2964.

Proximate Tradeoff: Flexibility • Enzymes achieve greater stability by reducing flexibility. • Flexible motions are important for catalysis in many enzymes. • Thus thermostability through reduced flexibility decreases activity. Somero, G. N. (1995) Annu. Rev. Physiol. 57, 43 -68.

Flexibility is Weak Link • Protein flexibility is complex – Spans picoseconds to milliseconds – Varies spatially • Only meaningful to discuss particular motions and how they affect stability and activity • Stability and activity often involve different regions and different time scales Lazaridis, T. , Lee, I. & Karplus, M. (1997) Prot. Sci. 6, 2589 -2605.

S/A Tradeoff Hypotheses 1. All enzymes have proximate tradeoff 2. Ultimate: Selection for high S&A Proximate: Highly optimized enzymes have S/A tradeoff – No known generic mechanism, e. g. flexibility – Experiments do not support notion
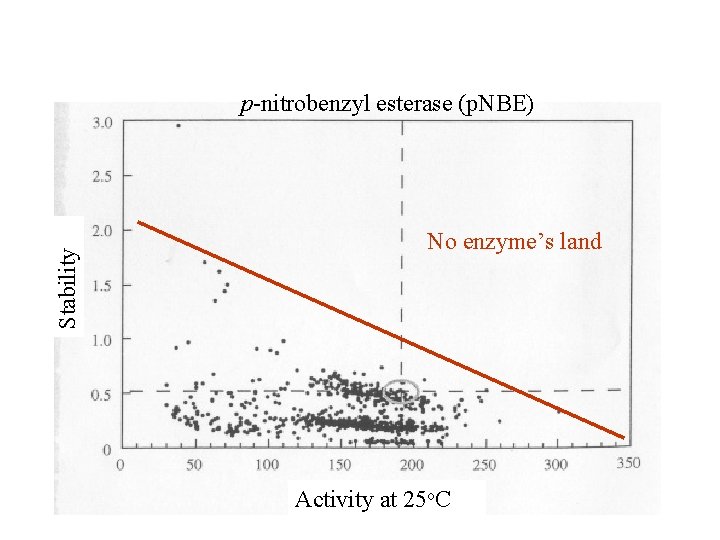
Stability p-nitrobenzyl esterase (p. NBE) No enzyme’s land Activity at 25 o. C
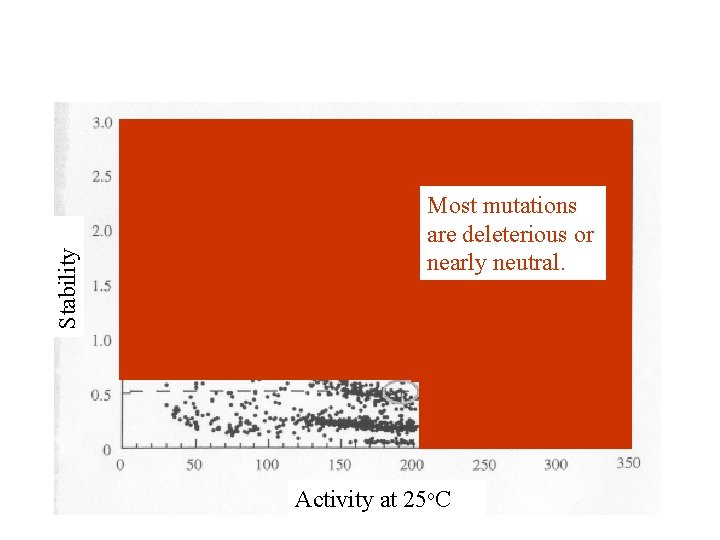
Stability Most mutations are deleterious or nearly neutral. Activity at 25 o. C
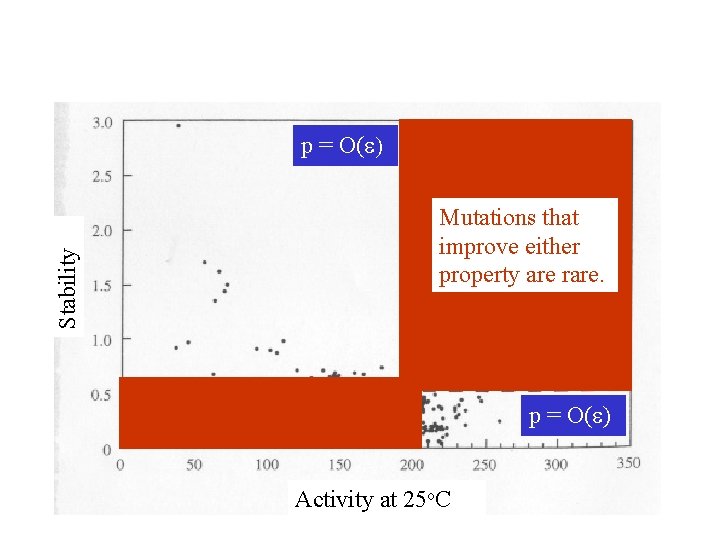
Stability p = O(e) Mutations that improve either property are rare. p = O(e) Activity at 25 o. C

Stability p = O(e 2) Mutations that improve both properties are very rare Activity at 25 o. C
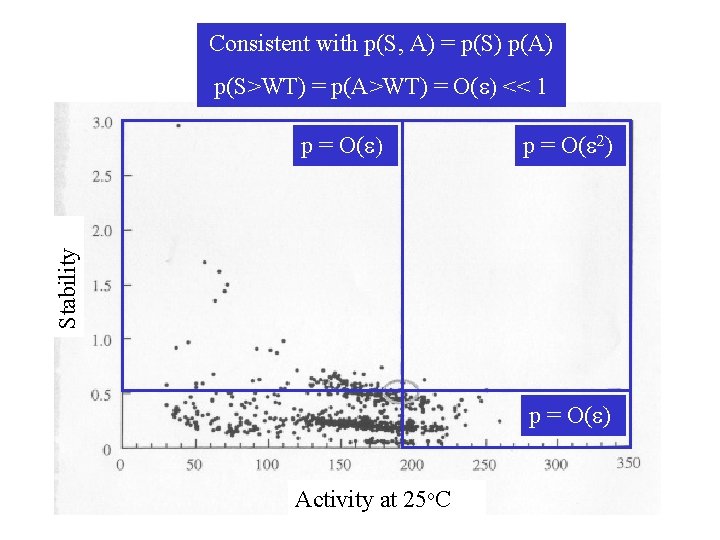
Consistent with p(S, A) = p(S) p(A) p(S>WT) = p(A>WT) = O(e) << 1 p = O(e 2) Stability p = O(e) Activity at 25 o. C
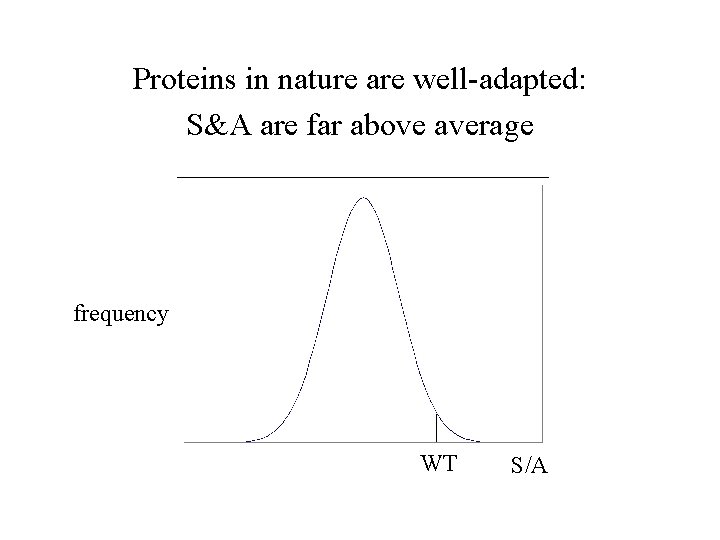
Proteins in nature are well-adapted: S&A are far above average frequency WT S/A

Buffering/Evolvability • More mutations are nearly neutral than might be expected for random tinkering of complex system • Compartmentalization – protein domains • Redundancy – Hydrophobicity – Steric requirements Gerhart, J. & Kirschner, M. (1997) Cells, Embryos, & Evolution. Malden: Blackwell Science.
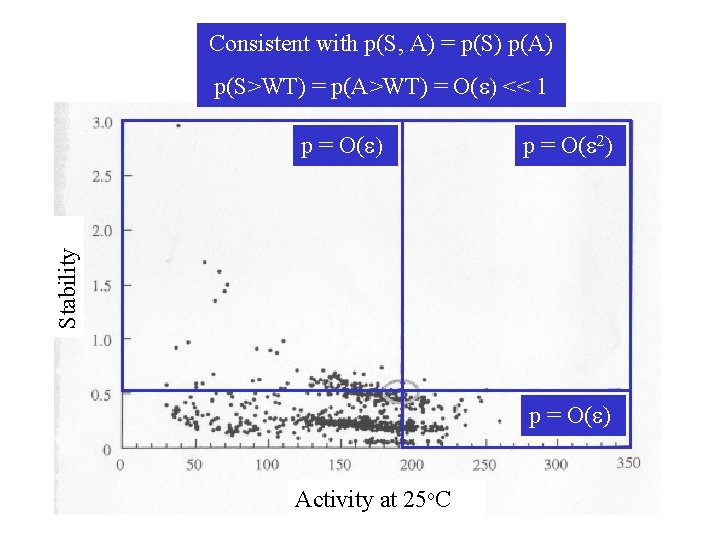
Consistent with p(S, A) = p(S) p(A) p(S>WT) = p(A>WT) = O(e) << 1 p = O(e 2) Stability p = O(e) Activity at 25 o. C
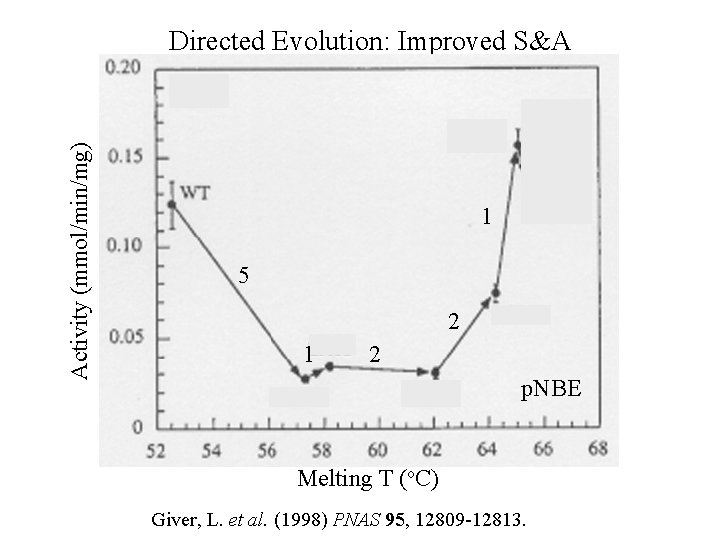
Activity (mmol/min/mg) Directed Evolution: Improved S&A 1 5 2 1 2 p. NBE Melting T (o. C) Giver, L. et al. (1998) PNAS 95, 12809 -12813.

S/A Tradeoff Hypotheses 1. All enzymes have proximate tradeoff 2. Ultimate: Selection for high S&A Proximate: Highly optimized enzymes have S/A tradeoff 3. Proximate: Most mutations are deleterious or nearly neutral Ultimate: Selection for threshold S&A Wintrode, P. L & Arnold, F. H. (2001) Adv. Prot. Chem. 55, 161 -225.
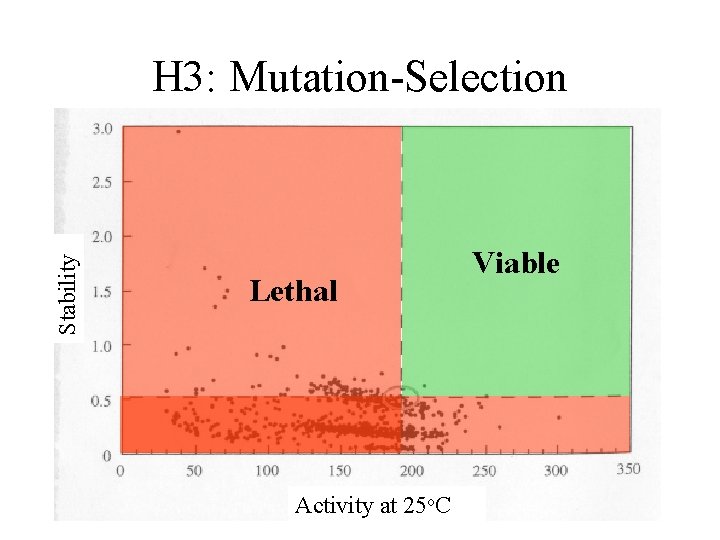
Stability H 3: Mutation-Selection Lethal Activity at 25 o. C Viable

Threshold Selection • DGu(Th) = c k. Th – KD/N = e-c – Proteins typically have c > 7 – No reason (or evidence) to believe higher S has selective advantage

Threshold Selection • DGu(Th) = c k. Th – KD/N = e-c – Proteins typically have c > 5 – No reason (or evidence) to believe higher S has selective advantage • A(Th) = a – With low flux control coefficient, higher A may offer no advantage – When important for control, higher A may be disadvantageous
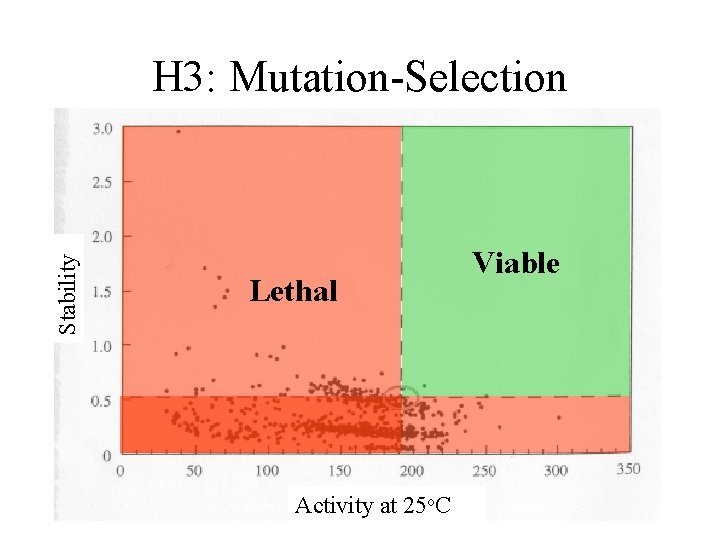
Stability H 3: Mutation-Selection Lethal Activity at 25 o. C Viable

Stability Mutation brings S&A to thresholds Lethal Activity at 25 o. C Viable
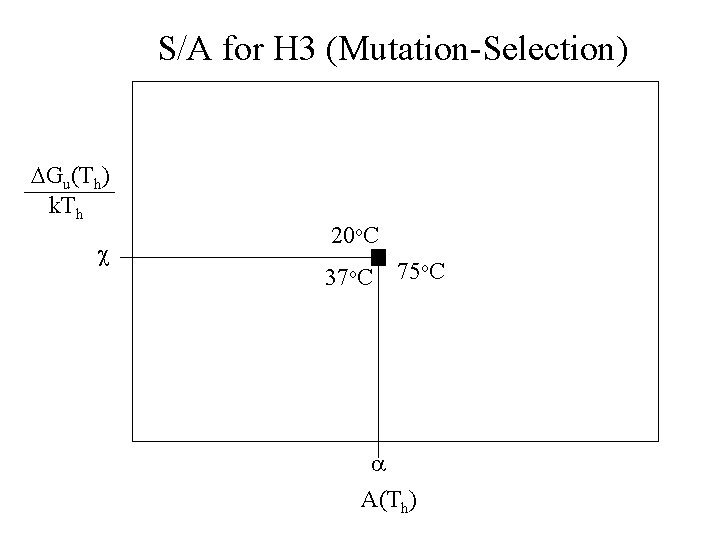
S/A for H 3 (Mutation-Selection) DGu(Th) k. Th c 20 o. C 37 o. C 75 o. C a A(Th)
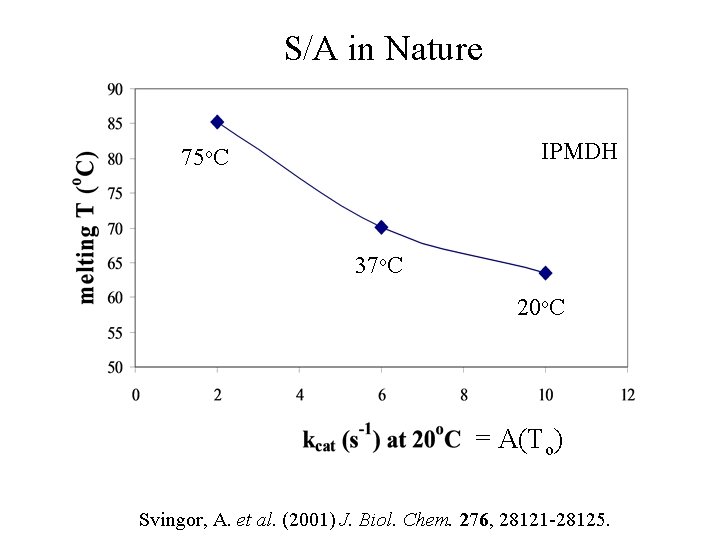
S/A in Nature IPMDH 75 o. C 37 o. C 20 o. C = A(To) Svingor, A. et al. (2001) J. Biol. Chem. 276, 28121 -28125.
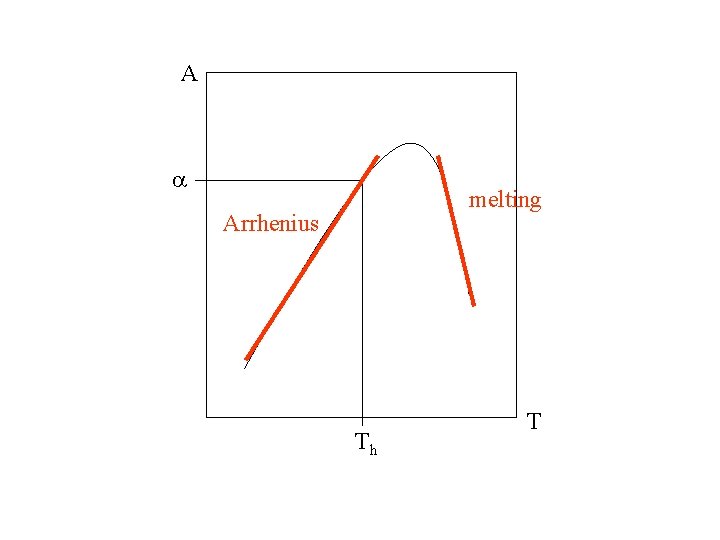
A a melting Arrhenius Th T

T 75 o. C Th 37 o. C 20 o. C a A
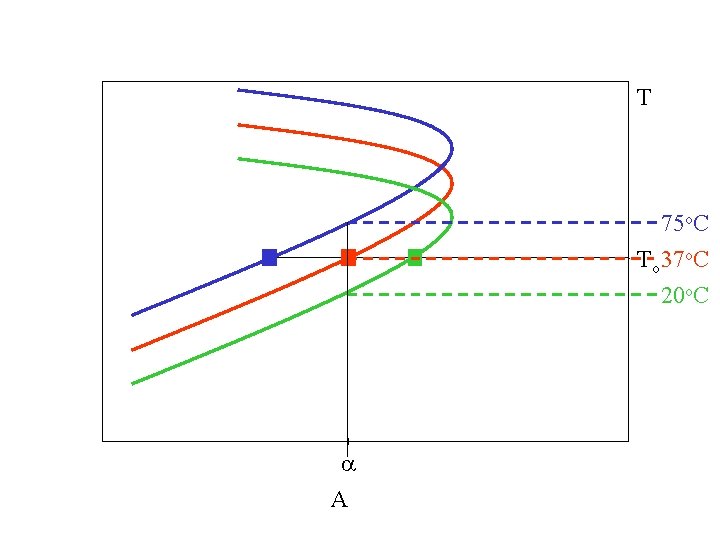
T 75 o. C To 37 o. C 20 o. C a A

S/A for H 3 (Mutation-Selection) DGu(Th) k. Th 75 o. C 37 o. C 20 o. C c a A(To)
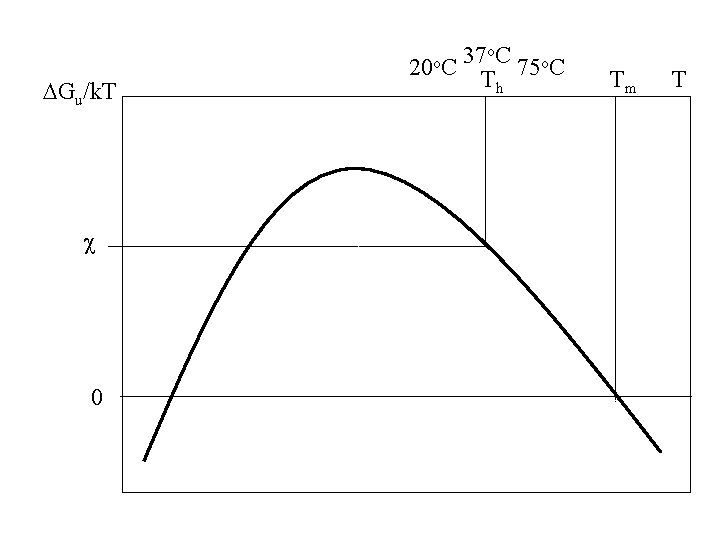
DGu/k. T c 0 20 o. C 37 o. C 75 o. C Th Tm T

DGu/k. T c 0 20 o. C 37 o. C 75 o. C T
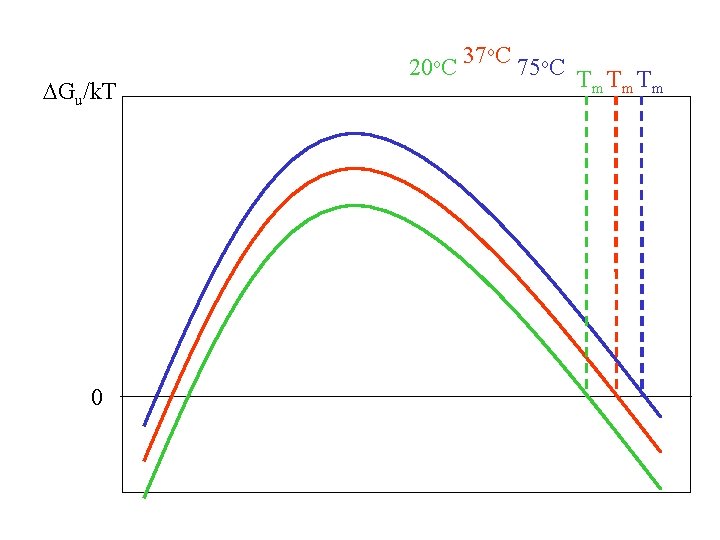
DGu/k. T 0 20 o. C 37 o. C 75 o. C Tm Tm Tm
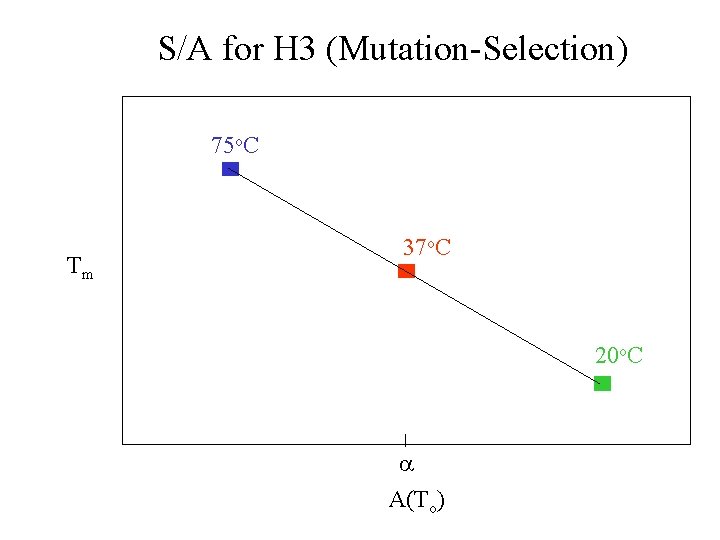
S/A for H 3 (Mutation-Selection) 75 o. C Tm 37 o. C 20 o. C a A(To)
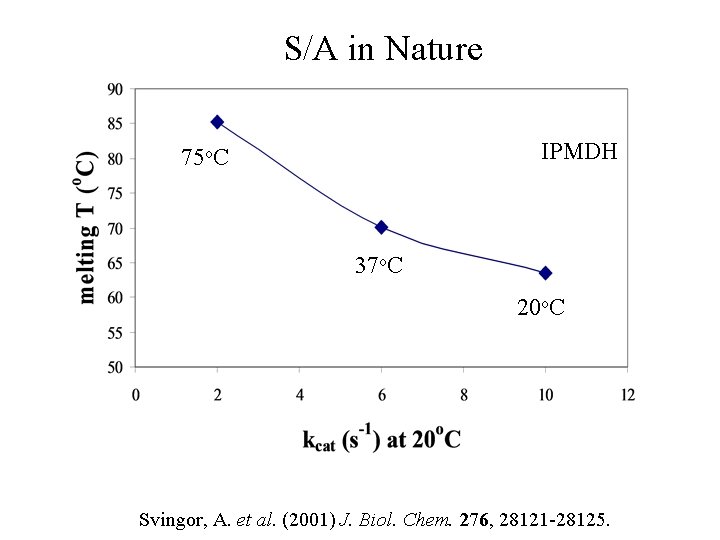
S/A in Nature IPMDH 75 o. C 37 o. C 20 o. C Svingor, A. et al. (2001) J. Biol. Chem. 276, 28121 -28125.

Conclusions • Because biological phenotypes are well-adapted, most mutations are deleterious • This mutational pressure pushes phenotypes to the thresholds of selection • Selection that requires homologs to have comparable S&A at physiological temperatures creates the appearance of S/A tradeoffs at a reference temperature • The proximate causes for S&A among homologs are unlikely to be universal

Performance Tradeoffs • • • Pervasive in biological thinking Resource allocation (time, energy, mass) Design tradeoffs Biochemistry: Stability/Activity Behavior: Foraging, Fight/Flight Physiology: Respiration, Biomechanics
- Slides: 49