STABILITY TESTING PROTOCOLS ACCORDING TO UNITED STATES Presented

STABILITY TESTING PROTOCOLS ACCORDING TO UNITED STATES Presented By : Ch. Mahesh Babu. M. PHARMACY Guided By : Mrs. Mcthel Asst. professor Dept of pharmaceutics NIRMALA COLLEGE OF PHARMACY

INTRODUCTION Ø Stability testing is an integral part of pharmaceutical development. Ø The primary purpose of stability testing is to provide supporting evidence on stability behavior of pharmaceutical drug products. Ø Many factors drive the process of industrial stability testing for pharmaceutical development.

STABILITY TESTING AND PRODUCT DEVELOPMENT Ø Ø Ø It is an evolutionary concept covering the life cycle of pharmaceutical product development. In early discovery phase the primary focus is to generate stability characteristics of a chemical /biological entity. In later stages , the goal is to establish shelf life formulations packaged in final package intended for commercial introduction.

STABILITY TESTING PRINCIPLES CAN BE SUBDIVIDED IN TO VARIOUS STAGES OF DEVELOPMENT Ø Discovery phase Ø Pre clinical stage Ø Pre-IND stage Ø Product development stage Ø NDA stage Ø Approved product stage Ø Revised product stage

DISCOVERY PHASE Ø Ø To help select the most satisfactory chemical entity possessing the right pharmacological, toxicological & pharmaceutical profile. The pharmaceutical profile is mostly focused towards the optimum chemical and physical stability characteristics. To select the right physical form(base, salt , ester) These studies help establish the boundaries with in which one must operate to design formulations.

PRE CLINICAL STAGE � The development of early dosage form for pre clinical testing in humans require an extensive stability evaluation. � Preliminary stability testing on all formulations must be carried out using stability indicating assays in accordance with GLPs. � It requires an entrance assay prior to the initiation of toxicological testing and an exit assay, must be performed at the end of the studies.

PRE-IND STAGE � Pre-formulation & stability evaluation of chemical entity is carried out according to ICH guidelines. � In DOSAGE FORM PARAMETERS SOLID Temperature, humidity, photo degradation. solutions p. H, ionic strength, additives addition to normal preformulation evaluations , forced degradation studies under highly stressed stability conditions is under taken.

IND STAGE � Accelerated and normal storage temperature testing of drug substance and for clinical formulation must be initiated. � The goal of these studies should be to generate information to insure that the clinical formulations are likely to remain stable during the planned clinical studies.

PRODUCT DEVELOPMENT STAGE � Intermediate stage. � Interim stability testing is done in this stability testing is conducted to establish the maximum time for which a drug product can be stored in interim containers for further processing.

NDA STAGE � Formal stability program is established for generation of stability data for registration applications. � The stability of drugs should be evaluated in containers used for marketing. � Care should be exercised in selection of the size, surface-to volume ratio of the container.

APPROVED PRODUCT STAGE � The goal of the stability evaluation program during this phase is to confirm or extending the expiration date. � The commitment in this stage mandates that any batch that is found out of specification will be with drawn from the market.

REVISED PRODUCT STAGE � Most products undergo post approval changes. � They may be internally driven or externally driven. � Internally driven : changing size & shape of dosage forms, changes in package design and others. � Externally driven : deletion of dyes, formulation changed and others.

DEVELOPMENT OF STABILITY TESTING FUNCTION � Establishment of stability testing function requires an extensive development of documentation to maximize the compliance and to minimize the regulatory citations.

WRITTEN STABILITY TESTING PROGRAM � The guidelines in section 211. 166 of 21 CFR states “ there shall be a written stability testing program designed to assess the stability characteristics of drug product”. �a written stability testing program is critical for the establishment of the stability testing function.

STABILITY PROTOCOL AND COMMITMENT � The stability protocol should record the purpose for conducting the stability test the method used, the testing frequency , storage conditions and several other factors. � The commitment requires submission of stability data at periodic intervals as specified in the application.

STANDARD OPERATING PROCEDURES � Current good manufacturing practices mandate that every organization develops a set of SOP’s to describe their operations. � It is almost impossible to develop a stability management function with out having a comprehensive set of SOP’s

TRAINING OF PERSONNEL � Regulations require that “each person engaged in the manufacturing processing should have education or training to enable that person to perform the assigned functions”. � Training shall be in the particular operations that the employees performs. � Training must be conducted by qualified individual on a continuous basis with sufficient frequency.

SELECTION OF BATCHES AND SAMPLES � During discovery phase stability evaluation is used in the conduct of pre-clinical safety. � This data helps to establish retest period for drug substances. � Selection of three pilot batches is used for statistical evaluation.

STABILITY STORAGE TEST CONDITIONS AND STABILITY ROOMS � Based on the analysis the world is divided in to 4 climatic zones.
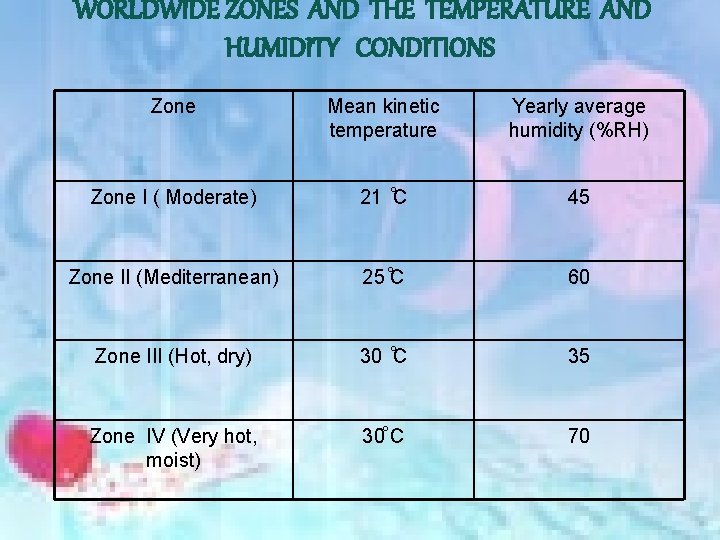
WORLDWIDE ZONES AND THE TEMPERATURE AND HUMIDITY CONDITIONS Zone Mean kinetic temperature Yearly average humidity (%RH) Zone I ( Moderate) 21 C 45 Zone II (Mediterranean) 25 C 60 Zone III (Hot, dry) 30 C 35 Zone IV (Very hot, moist) 30 C 70
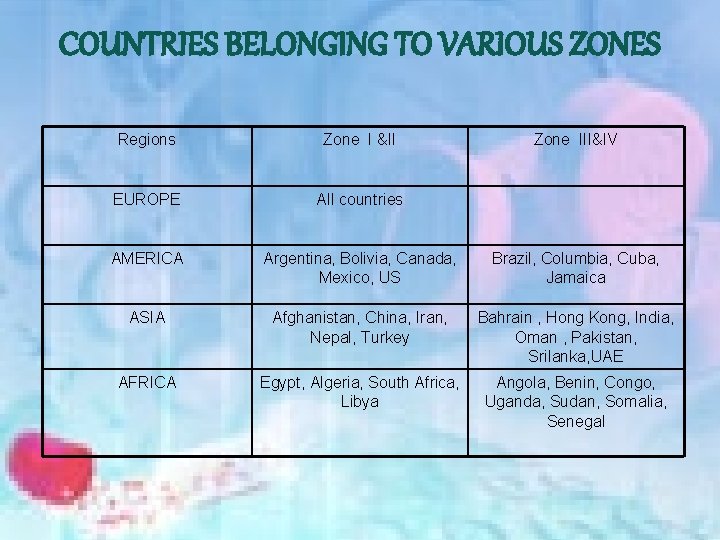
COUNTRIES BELONGING TO VARIOUS ZONES Regions Zone I &II Zone III&IV EUROPE All countries AMERICA Argentina, Bolivia, Canada, Mexico, US Brazil, Columbia, Cuba, Jamaica ASIA Afghanistan, China, Iran, Nepal, Turkey Bahrain , Hong Kong, India, Oman , Pakistan, Srilanka, UAE AFRICA Egypt, Algeria, South Africa, Libya Angola, Benin, Congo, Uganda, Sudan, Somalia, Senegal

Large stability chambers

Small stability chamber

Stability protocol Study Storage condition Minimum time period covered by data at submission Long Term (Ambient) 25º C ± 2º C 60%RH ± 5% 12 months Intermediate (controlled) 30º C ± 2º C 60%RH ± 5% 6 months Accelerated 40º C ± 2º C 75%RH ± 5% 6 months

Thermal cycling � To simulate the transportation and shipment conditions in the stability studies thermal cycling is done � Testing parameters for those studies should include not only the chemical analysis but also physical changes.

Photostability testing of new drug substances & products Photostability testing studies include: • Test on drug substance. • Test on exposed drug product outside the immediate pack. • Test on drug product in the marketing pack. Light source Option 1: Artificial daylight lamp combining both visible & UV output similar to D 65 & ID 65. Option 2: Cool white fluorescent & near UV lamp output max. energy emitted

Procedure Sample exposed to light source/actinometric system Eg: Quinine chemical actinometry 2%w/v aq. solution of quinine monohydrochloride dihydrate Ø Option 1: In 20 ml colourless ampoule at 400 nm Option 2: In 1 cm quartz cell-(sample) and control wrapped with Aluminium foil Change in absorbance calculated by A = AT-A 0


Photostability testing Forced degradation Evaluate the photosensitivity Its used alone or in solution Placed in chemically inert & transparent containers. Variety of exposure condition Confirmatory testing Information necessary for handling, packaging and labeling To study, identify precautionary measures needed in manufacturing or in formulation

CONTAINER CLOSURE SYSTEMS � The containers should be tested in all directions i. e. . up right , inverted , on the side positions. � This is done for long term and accelerated stability testing. � This is to ensure that there are no adverse effects from any interaction is produced.

Testing frequency To establish the stability profile of drug substance For long-term storage 12 month study ◦ Testing frequency 0 3 6 9 12 For accelerated storage 6 month study ◦ Testing frequency 0 3 6 (initial) (final) If significant change occur ◦ Increase the testing by adding sample at final time point ◦ Include 4 th time point in study design For intermediate storage 12 month study - Testing frequency 0 6 12 If significant change occur - A 4 th time point can be included

STABILITY OF RECONSTITUTED PRODUCTS � According to the section 211. 166 of 21 CFR states that “testing of drug products for re constitution at the time of dispensing as well as after they are reconstituted”. � For reconstituted products two distinct stability periods are in operation. � First period : long term and accelerated stability testing prior to reconstitution. � Second period: short term stability after reconstitution
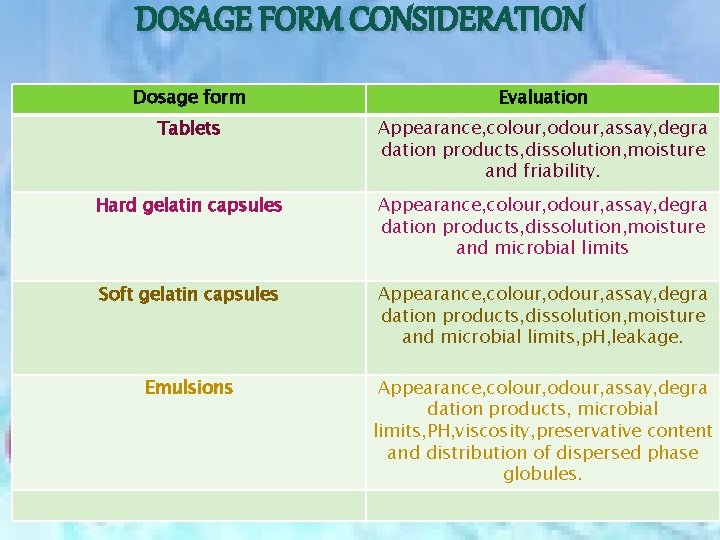
DOSAGE FORM CONSIDERATION Dosage form Evaluation Tablets Appearance, colour, odour, assay, degra dation products, dissolution, moisture and friability. Hard gelatin capsules Appearance, colour, odour, assay, degra dation products, dissolution, moisture and microbial limits Soft gelatin capsules Appearance, colour, odour, assay, degra dation products, dissolution, moisture and microbial limits, p. H, leakage. Emulsions Appearance, colour, odour, assay, degra dation products, microbial limits, PH, viscosity, preservative content and distribution of dispersed phase globules.
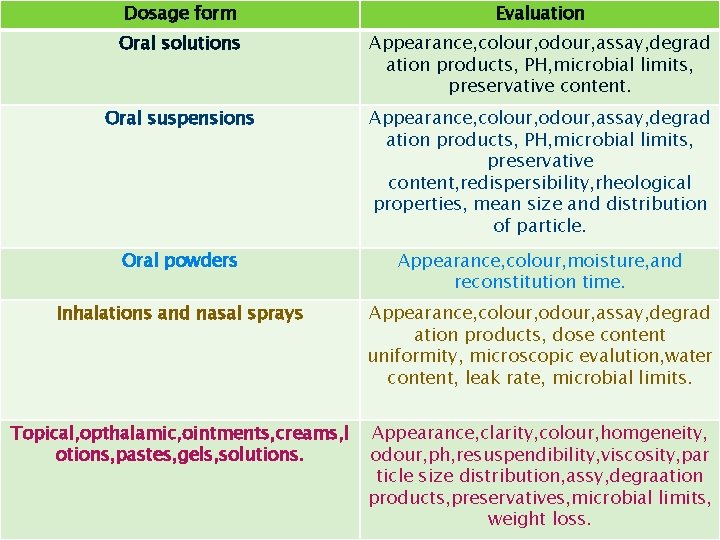
Dosage form Evaluation Oral solutions Appearance, colour, odour, assay, degrad ation products, PH, microbial limits, preservative content. Oral suspensions Appearance, colour, odour, assay, degrad ation products, PH, microbial limits, preservative content, redispersibility, rheological properties, mean size and distribution of particle. Oral powders Appearance, colour, moisture, and reconstitution time. Inhalations and nasal sprays Appearance, colour, odour, assay, degrad ation products, dose content uniformity, microscopic evalution, water content, leak rate, microbial limits. Topical, opthalamic, ointments, creams, l otions, pastes, gels, solutions. Appearance, clarity, colour, homgeneity, odour, ph, resuspendibility, viscosity, par ticle size distribution, assy, degraation products, preservatives, microbial limits, weight loss.

Dosage form Evaluation Small volume parenterals Appearance, colour, clarity, assay, presarvative content, degradation products, particulate matter, sterility, Large volume parenterals Appearance, colour, clarity, assay, presarvative content, degradation products, particulate matter, sterility, p. H, pyrogeni city, volume.

BRACKETING Bracketing is the design of a stability schedule such that samples on the extremes of certain design factors are tested at all time points. Protocols for bracketing designs should be endorsed by the FDA prior to initiation of primary stability studies (FDA 1998). Bracketing design is suitable and applicable to identical or closely related formulations packaged in different size identical container/closure system.
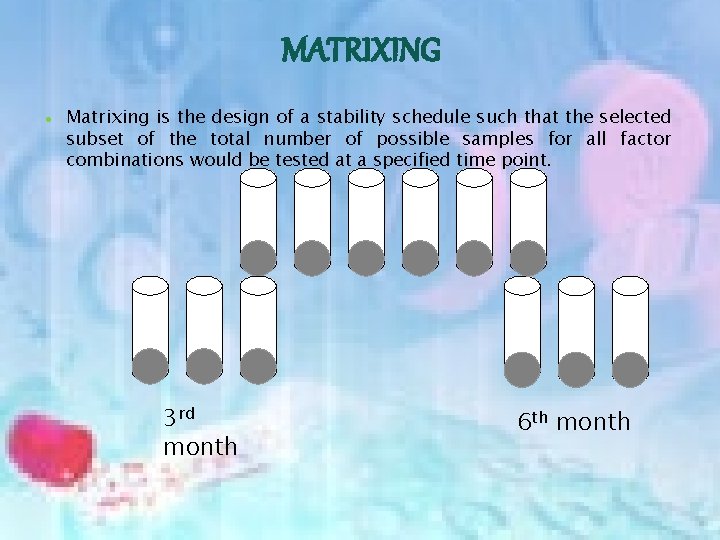
MATRIXING Matrixing is the design of a stability schedule such that the selected subset of the total number of possible samples for all factor combinations would be tested at a specified time point. 3 rd month 6 th month
- Slides: 37