Squalene is a linear triterpene 6 isoprene units
- Slides: 12

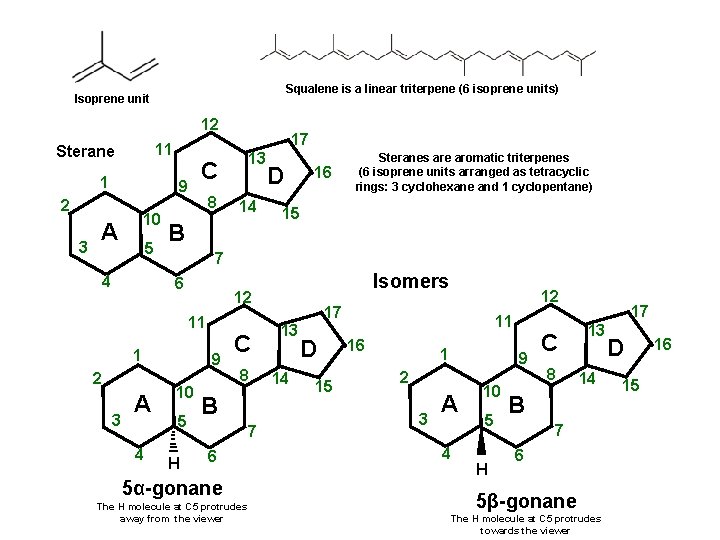
Squalene is a linear triterpene (6 isoprene units) Isoprene unit 12 11 Sterane 1 3 10 A 5 4 8 4 16 15 9 10 5 H Isomers 12 1 3 D Steranes are aromatic triterpenes (6 isoprene units arranged as tetracyclic rings: 3 cyclohexane and 1 cyclopentane) 7 11 A 14 B 6 2 13 C 9 2 17 C 8 B 7 6 5α-gonane The H molecule at C 5 protrudes away from the viewer 12 17 13 14 D 15 11 16 1 9 2 3 A 4 10 5 H C 8 17 13 14 B 7 6 5β-gonane The H molecule at C 5 protrudes towards the viewer D 15 16

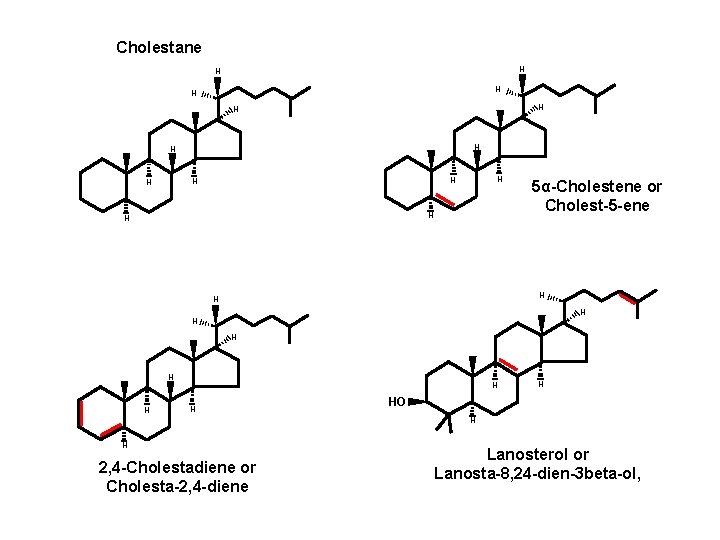
Cholestane H H H H 5α-Cholestene or Cholest-5 -ene H H HO H H 2, 4 -Cholestadiene or Cholesta-2, 4 -diene Lanosterol or Lanosta-8, 24 -dien-3 beta-ol,

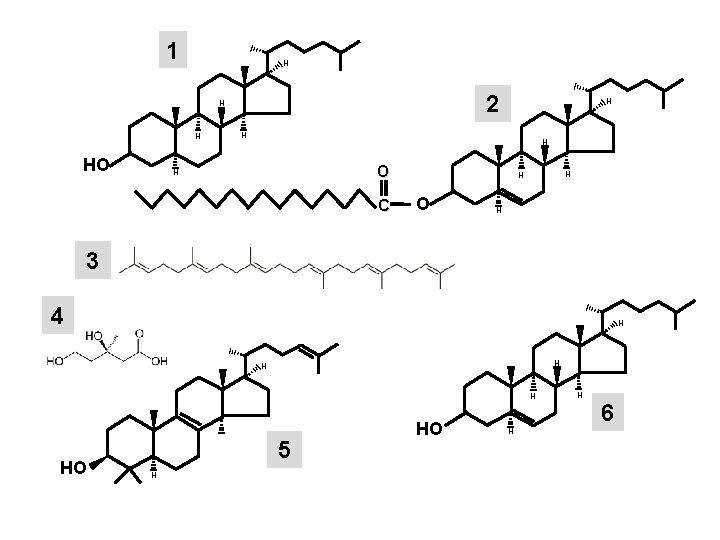
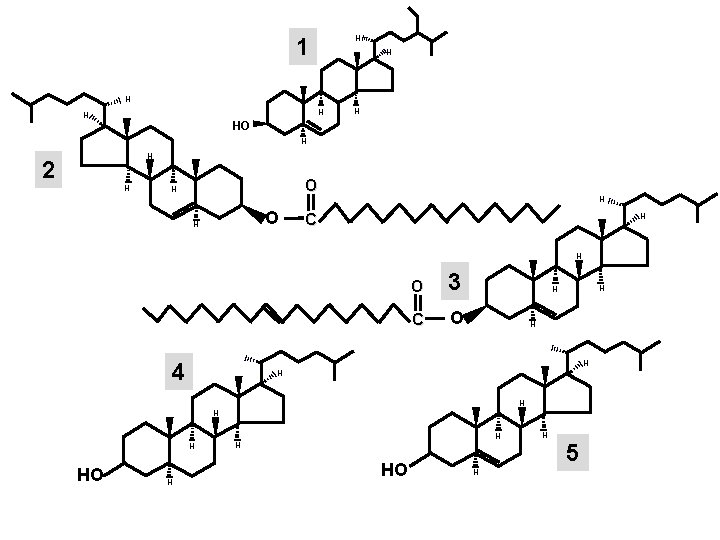
1 H 2 H H HO H H H O H C H H O H 3 4 H H HO 5 H HO H H 6

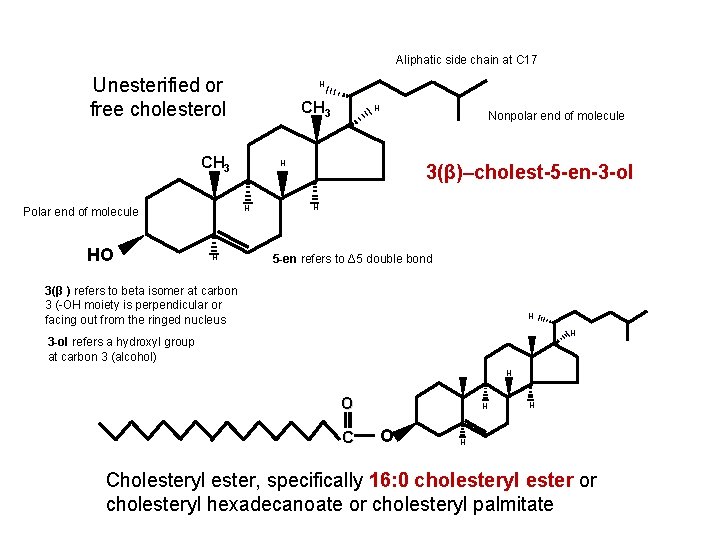
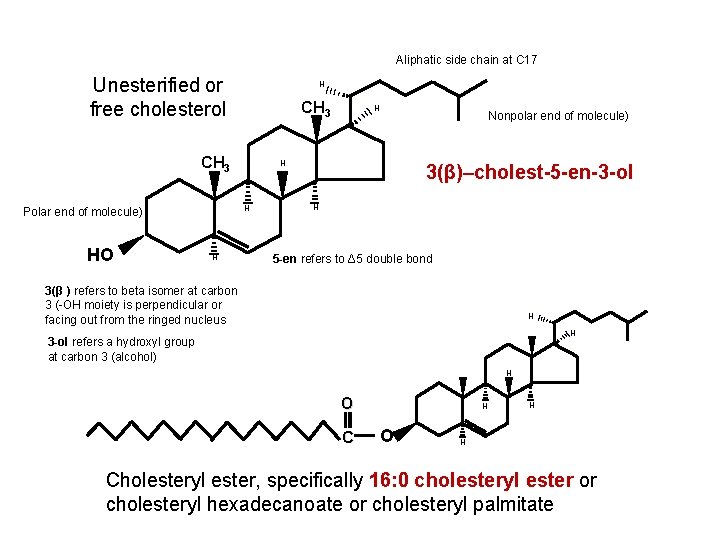
Aliphatic side chain at C 17 Unesterified or free cholesterol H CH 3 HO H Nonpolar end of molecule H H Polar end of molecule H 3(β)–cholest-5 -en-3 -ol H 5 -en refers to Δ 5 double bond 3(β ) refers to beta isomer at carbon 3 (-OH moiety is perpendicular or facing out from the ringed nucleus H H 3 -ol refers a hydroxyl group at carbon 3 (alcohol) H O C H O H H Cholesteryl ester, specifically 16: 0 cholesteryl ester or cholesteryl hexadecanoate or cholesteryl palmitate

Aliphatic side chain at C 17 Unesterified or free cholesterol H CH 3 HO H Nonpolar end of molecule) H H Polar end of molecule) H 3(β)–cholest-5 -en-3 -ol H 5 -en refers to Δ 5 double bond 3(β ) refers to beta isomer at carbon 3 (-OH moiety is perpendicular or facing out from the ringed nucleus H H 3 -ol refers a hydroxyl group at carbon 3 (alcohol) H O C H O H H Cholesteryl ester, specifically 16: 0 cholesteryl ester or cholesteryl hexadecanoate or cholesteryl palmitate

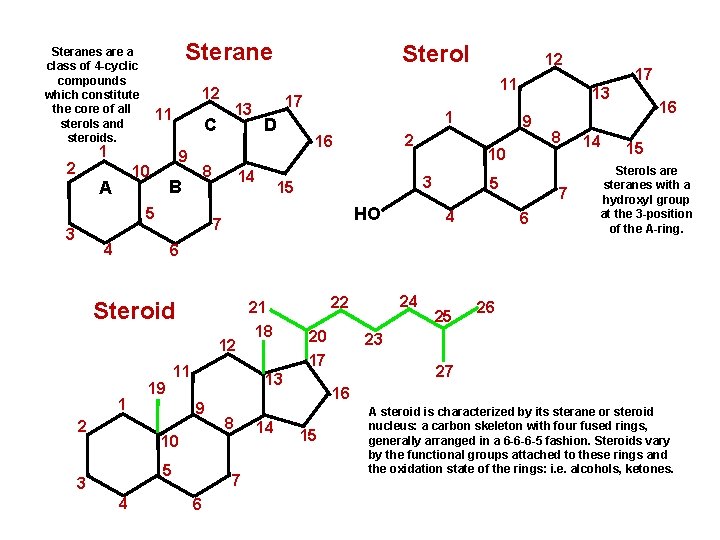
Steranes are a class of 4 -cyclic compounds which constitute the core of all sterols and steroids. 12 11 1 2 9 10 A 8 B 5 3 D 14 1 2 16 15 HO 9 5 4 17 13 10 3 7 4 12 11 17 13 C Sterol 8 7 6 14 16 15 Sterols are steranes with a hydroxyl group at the 3 -position of the A-ring. 6 Steroid 12 1 2 19 11 9 5 4 20 8 7 6 14 25 26 23 17 13 10 3 18 24 22 21 27 16 15 A steroid is characterized by its sterane or steroid nucleus: a carbon skeleton with four fused rings, generally arranged in a 6 -6 -6 -5 fashion. Steroids vary by the functional groups attached to these rings and the oxidation state of the rings: i. e. alcohols, ketones.

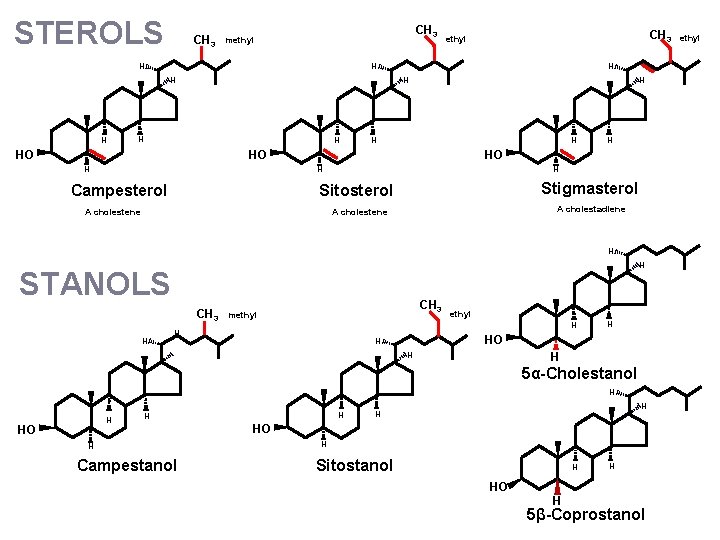
STEROLS CH 3 methyl H H CH 3 ethyl H H HO H HO H Campesterol H H Stigmasterol Sitosterol A cholestene A cholestadiene A cholestene H H STANOLS CH 3 H CH 3 methyl H H H HO H H H 5α-Cholestanol H H HO H H H Campestanol H H HO H Sitostanol H HO H H 5β-Coprostanol ethyl

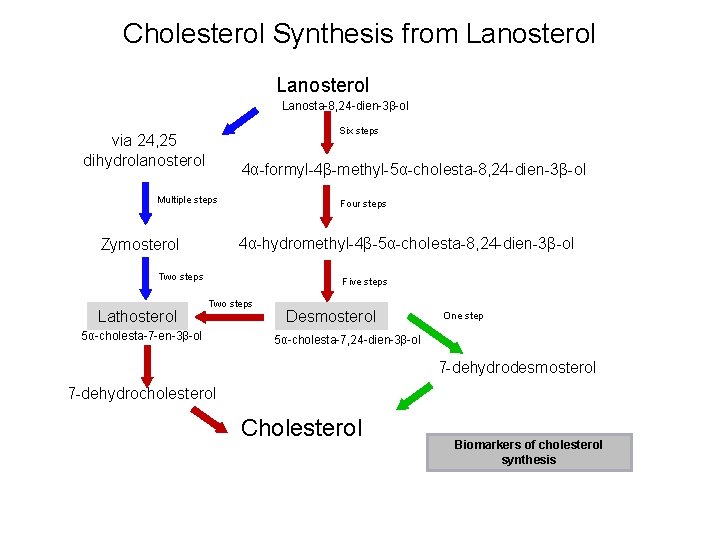
Cholesterol Synthesis from Lanosterol Lanosta-8, 24 -dien-3β-ol Six steps via 24, 25 dihydrolanosterol 4α-formyl-4β-methyl-5α-cholesta-8, 24 -dien-3β-ol Multiple steps Four steps 4α-hydromethyl-4β-5α-cholesta-8, 24 -dien-3β-ol Zymosterol Two steps Lathosterol Five steps Two steps 5α-cholesta-7 -en-3β-ol Desmosterol One step 5α-cholesta-7, 24 -dien-3β-ol 7 -dehydrodesmosterol 7 -dehydrocholesterol Cholesterol Biomarkers of cholesterol synthesis

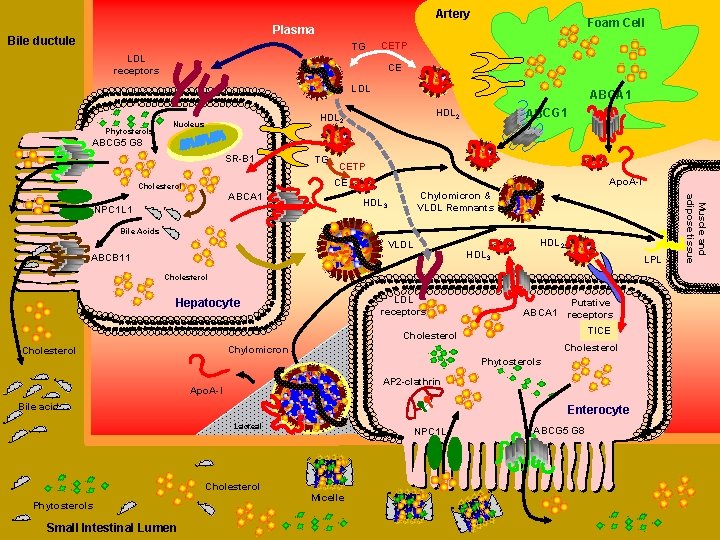
Artery Foam Cell Plasma Bile ductule TG CETP LDL receptors CE LDL Phytosterols ABCA 1 ABCG 1 HDL 2 Nucleus ABCG 5 G 8 SR-B 1 TG CETP Apo. A-I CE Cholesterol HDL 3 NPC 1 L 1 Bile Acids VLDL HDL 3 ABCB 11 HDL 2 LPL Cholesterol LDL receptors Hepatocyte ABCA 1 Putative receptors TICE Cholesterol Chylomicron Cholesterol Phytosterols AP 2 -clathrin Apo. A-I Bile acids Enterocyte Lacteal Cholesterol Phytosterols Small Intestinal Lumen Lacteal Micelle NPC 1 L 1 ABCG 5 G 8 Muscle and adipose tissue ABCA 1 Chylomicron & VLDL Remnants

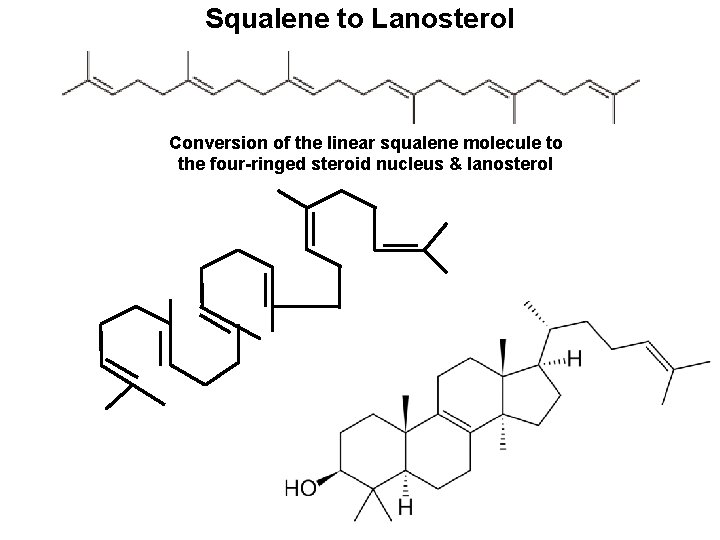
Squalene to Lanosterol Conversion of the linear squalene molecule to the four-ringed steroid nucleus & lanosterol

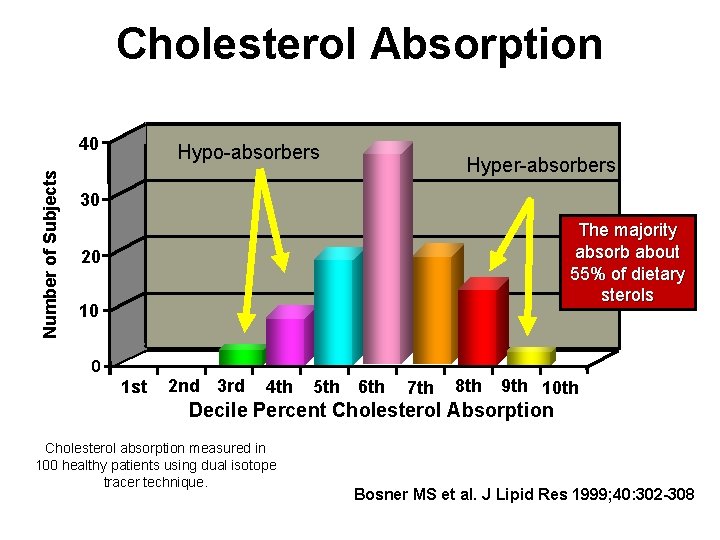
Cholesterol Absorption Number of Subjects 40 Hypo-absorbers Hyper-absorbers 30 The majority absorb about 55% of dietary sterols 20 10 0 1 st 2 nd 3 rd 4 th 5 th 6 th 7 th 8 th 9 th 10 th Decile Percent Cholesterol Absorption Cholesterol absorption measured in 100 healthy patients using dual isotope tracer technique. Bosner MS et al. J Lipid Res 1999; 40: 302 -308

H 1 H H HO H H 2 H O H H C H H 4 O 3 C O H H H H HO H H 5