SPS 6 Students will investigate the properties of























- Slides: 25

SPS 6. Students will investigate the properties of solutions. a. Describe solutions in terms of -solute/solvent -conductivity -concentration b. Observe factors affecting the rate a solute dissolves in a specific solvent. c. Demonstrate that solubility is related to temperature by constructing a solubility curve. d. Compare and contrast the components and properties of acids and bases. e. Determine whether common household substances are acidic, basic, or neutral.

Solutions • Solution – a mixture of two or more substances

• The substance to be dissolved. • The one doing the dissolving.

Example: Salt Water • Solution: Salt Water • Solute: Salt • Solvent: Water

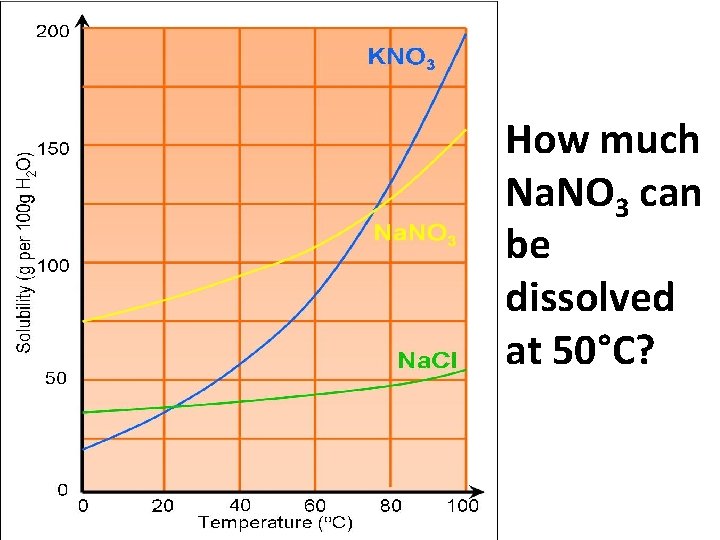
VOCAB • Solubility: the amount of solute that will dissolve in a given amount of solvent • Solubility Curve: graph that expresses solubility vs. temperature

How much Na. NO 3 can be dissolved at 50°C?

Factors Affecting Solubility • Particle size: smaller particles are more soluble • Temperature: many solids substances become more soluble as the temp of a solvent increases; however, gases are less soluble in liquids at higher temps. • Agitation: Increases the solubility of the solution

VOCAB • Concentration: a measure of the amount of solute dissolved in a certain volume of solvent

MORE VOCAB Saturated solution: any solution that contains all the solute it can hold (can no longer dissolve anymore) Unsaturated: any solution that can dissolve more solute Supersaturated: any solution that contains more solute than a saturated solution

p. H Scale • p. H is a measure of how acidic or basic a solution is. • The p. H scale ranges from 0 to 14. • A solution with a p. H of 7 is neutral. • Pure water has a p. H of 7.

Definition of p. H: • Concentration of hydronium ions in solution

ACIDS • p. H below 7 • A solution with a p. H of 0 is the most acidic. • Excess H+ ions • Strong Acid = Strong Conductor • Corrosive • Turn blue litmus paper red • Ex. Coffee, soda, fruit, sour candies

Bases (alkalis) • p. H above 7 • A solution with a p. H of 14 is the most basic • Excess OH- ions • Strong base = strong conductor • Slippery • Turn red litmus paper blue • Ex. Soap, cleaning products

Neutralization Reaction Chemical reaction in which an acid and a base react in a water solution i. Not acidic or basic so p. H is 7 Equal concentrations of H+ and OHions

Flashcard VOCAB Words • • Solution Solute Solvent Solubility Concentration Saturated Solution Unsaturated Solution Supersaturated Solution • • p. H Acid Base Neutralization Reaction

SPS 7. Students will relate transformations and flow of energy within a system. • a. Identify energy transformations within a system (e. g. lighting of a match). • b. Investigate molecular motion as it relates to thermal energy changes in terms of conduction, convection, and radiation. • c. Determine the heat capacity of a substance using mass, specific heat, and temperature. • d. Explain the flow of energy in phase changes through the use of a phase diagram.

Types of Energy • • • Potential Energy: Stored Kinetic Energy: Movement Chemical Energy: Chemical Bonds Electrical Energy: Charged Ions Other types: thermal (heat), light, wind, etc.

Three Examples of Energy Transformations • Swing Set/Pendulum – Potential to Kinetic to Potential • Lighting a Match – Chemical to light to heat • A remote control – Chemical to electrical

Thermal Energy: 3 Types • Conduction: touch • Convection: fluid • Radiation: electromagnetic waves (ex sun)

Heat Capacity • Amount of thermal energy needed to raise 1 kg of that material 1°C • The higher the heat capacity, the more energy needed to raise the temperature

Calculating Heat Capacity • Q = mΔTC OR Q = m (Tf-Ti)C – Q = Heat (Joules) – m = Mass (grams or kg) – T = Temperature (°C) – C = Heat Capacity (J/Kg°C)

Example Problem • How much heat is gained when 114. 3 g of water at 14. 8°C is raised to 18. 0°C? (Cwater =4. 18 J/kg°C)

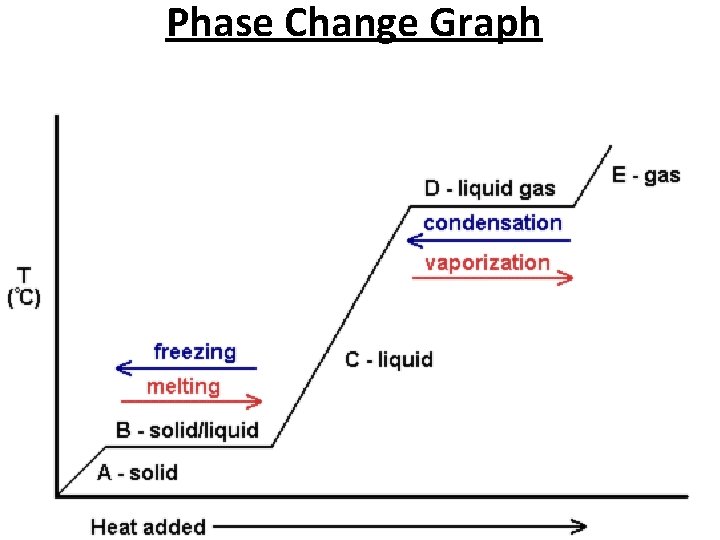
Phase Changes Exothermic (losing heat) Endothermic (gaining heat) • Gas Liquid : Condensation • Liquid Solid : Freezing • Gas Solid : Deposition • Solid Liquid : Melting • Liquid Gas : Vaporization • Solid Gas : Sublimation

Phase Change Graph

Flashcard VOCAB Words • • Potential Energy Kinetic Energy Chemical Energy Electrical Energy Conduction Convection Radiation Heat Capacity • • Melting Freezing Vaporization Deposition Sublimation Condensation Exothermic Endothermic