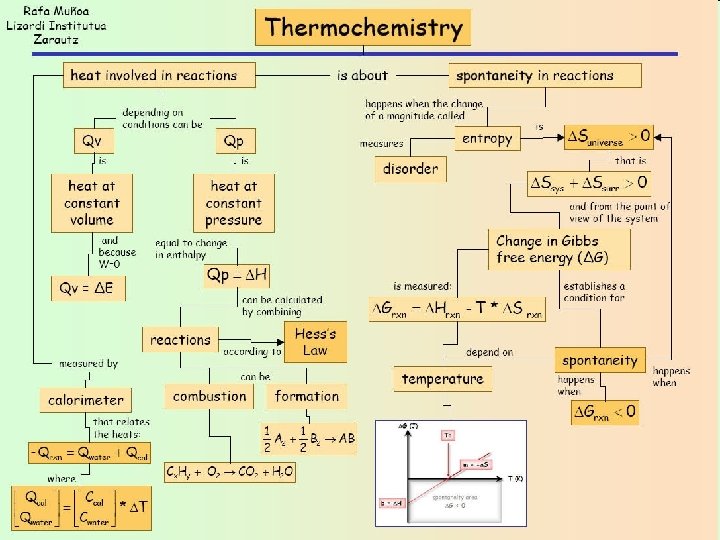
Spontaniety Spontaneous reactions are reactions that once started

















- Slides: 20

Spontaniety � Spontaneous reactions are reactions, that once started, continues by itself without further input of energy from the outside. � If a reaction is spontaneous under a given set of conditions, then the reverse reaction is considered non-spontaneous.

Identifying Spontaneity • Identify whether the reactions below are spontaneous or not: – H 2 O (s) H 2 O (l) – 2 H 2 (g) + O 2 (g) 2 H 2 O (l) *requires a spark to begin – 2 H 2 O (l) 2 H 2 (g) + O 2 (g) *requires consistent electric current

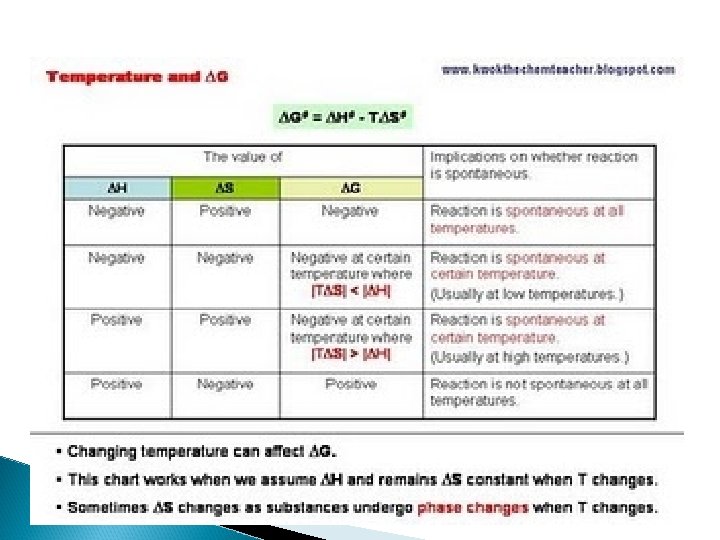
Enthaply and Spontaneity: • Almost all exothermic reactions are considered to be spontaneous (at 25 *C and 1 atm). – ΔH for a spontaneous reactions tends to be negative. • • • However, some endothermic reactions at specific temperatures may be considered spontaneous, for example the melting of ice at 1 atm above 0 *C: H 2 O (s) H 2 O (l) ΔH = 6. 0 k. J Endothermic reactions that are non-spontaneous at room temperature often become spontaneous when the temperature is raised. The Randomness Factor: In general, nature tends to move spontaneously from more ordered to more random states (order to disorder).

Entropy � The randomness factor discussed is treated quantitatively as entropy (S). Basically the greater the disorder (more random the distribution of molecules) the greater the entropy. � -Entropy, like enthalpy is a state property so that ΔS = Sfinal - Sinitial ◦ unit for S = J/mol K

Factors that influence entropy that a system has in a particular state: • • A liquid has higher entropy than the solid from which it is formed. A gas has a higher entropy than the liquid from which it is formed. Increasing the temperature of a substance increases its entropy. A completely ordered pure crystalline solid has an entropy of 0 K (3 rd law of thermodynamics).

Standard molar entropies of elements, compounds, and aqueous ions: • • • Elements have nonzero standard entropies. Standard molar entropies of pure substances are always positive quantities. Aqueous ions may have negative entropy values. As a group, gases tend to have higher entropies than liquids. An increase in the number of moles of a gas also leads to a higher entropy and vice versa. As a molecule becomes more complex, the higher the entropy (more ways for the atoms to move about with respect to one another).

Example 17. 1 � Predict whether ΔS is positive or negative for each of the following processes: ◦ Taking dry ice from a freezer where its temperature is -80°C and allowing it to warm to room temperature ◦ Dissolving bromine in hexane ◦ Condensing gaseous bromine to liquid bromine

Which of the following reactions results in the largest increase in entropy? (A) CO 2(s) CO 2(g) (B) H 2(g) + Cl 2(g) 2 HCl(g) (C) KNO 3(s) KNO 3(l) (D) C(diamond) C(graphite)

Calculating Entropy � To calculate the standard entropy change, ΔS, use the following relation: ΔS = ∑ ΔS products - ∑ ΔS reactants � The 2 nd Law of Thermodynamics: In a spontaneous process, there is a net increase in entropy, taking into account both the system and surroundings, ΔS > 0

Example � Calculate for the entropy change at 25ºC in J/K 2 SO 2(g) + O 2(g) 2 SO 3(g) given the following data: ◦ SO 2(g): 248. 1 J/mol-K ◦ O 2(g): 205. 3 J/mol-K ◦ SO 3(g): 256. 6 J/mol-K � [2(256. 6)] - [2(248. 1) + 1(205. 3)] = -188. 3 J/K

Example 17. 2 � Calculate ΔS for ◦ (1) dissolving one mole of calcium hydroxide in water ◦ (2) the combustion of one mole of methane, CH 4, to form carbon dioxide and liquid water

Gibbs Free Energy (G) • Gibbs free energy is a quantity that basically allows us to put the two quantities, enthalpy and entropy, together in such a way as to arrive at a single function whose sign will determine whether the reaction is spontaneous. The basic definition of Gibbs free energy is: ΔG = ΔH - TΔS (The Gibbs-Helmholtz Equation) – G = Gibbs Free Energy (J) – H = Enthalpy (J/mol) – T = Temperature (K) – S = Entropy (J/mol K -- please be aware that the mole in the unit tends to cancel out from the ΔS equation)

Understanding ΔG � The Gibb’s free energy equation combines all the information that we have learned thus far. But what does the Gibb’s free energy value tell us about a reaction? It tells us the following: ◦ If ΔG is negative, the reaction is spontaneous in the forward direction. ◦ If ΔG is equal to zero, the reaction is at equilibrium. ◦ If ΔG is positive, then the reaction is nonspontaneous in the forward direction, but the reverse reaction will be spontaneous. ◦ For elements at standard state (pure elements at 25ºC and 1 atm are assigned a value of zero).

Example 17. 3 � For the reaction Ca. SO 4 (s) Ca 2+ calculate: ◦ ΔH° ◦ ΔS° ◦ ΔG° at 25°C (aq) + SO 42 - (aq)

Gibbs Free Energy of Formation � The standard free energy of formation, ΔGf, can also be used to solve for the free energy of a reaction (reference Appendix 1 in text): ΔGf rxn = ∑ ΔGf products - ∑ ΔGf reactants � If it is a negative quantity then the compound can be formed spontaneously from the elements, like in the formation of water: H 2 (g) + ½ O 2 (g) � Elements ΔGf = 0 H 2 O (l) ΔGf = -237. 2 k. J in their elemental state will have a

Example 17. 4 � Using ΔG°f values from Appendix 1, calculate the standard free energy change at 25°C for the reaction Ca. SO 4 (s) Ca 2+ (aq) + SO 42 - (aq) � Using ΔG°f values from Appendix 1, calculate the standard free energy change at 25°C for the dissolution of 1 mole of calcium chloride.


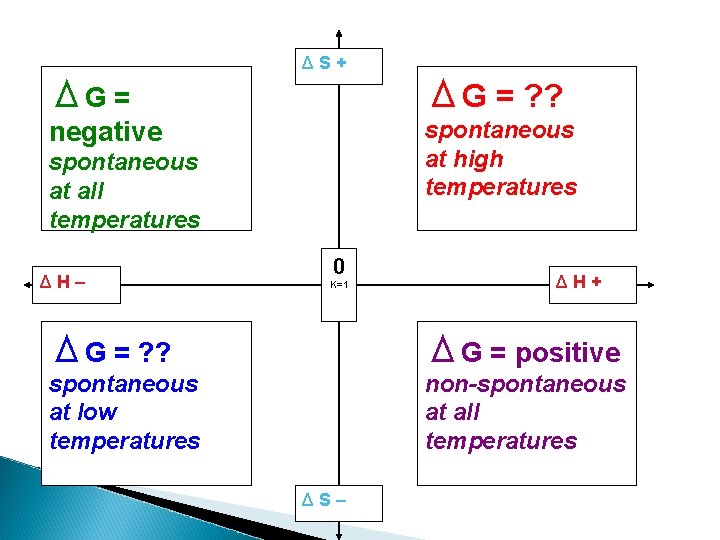
ΔS+ ΔG = ? ? negative spontaneous at high temperatures spontaneous at all temperatures ΔH– 0 K=1 ΔH+ ΔG = ? ? ΔG = positive spontaneous at low temperatures non-spontaneous at all temperatures ΔS–

Example 17. 6 � At what temperature does ΔG° become zero for the reaction Fe 2 O 3 (s) + 3 H 2 (g) 2 Fe (s) + 3 H 2 O (g)
