SPONTANEOUS CHANGE ENTROPY AND GIBBS FREE ENERGY Spontaneous

SPONTANEOUS CHANGE, ENTROPY AND GIBB’S FREE ENERGY

Spontaneous Change ■ Spontaneous change/reaction: one that will occur without any outside intervention. Spontaneous doesn’t mean rapid. ■ non-spontaneous event: can occur only if it receives some sort of outside assistance

Enthalpy and Spontaneity ■ exothermic reactions tend to be spontaneous since lowering the energy of the system tends to be favored ■ but the following reactions are also spontaneous at room temperature: H 2 O(s) H 2 O(l) ∆H° = +6. 0 k. J/mol as is: NH 4 NO 3(s) NH 4+(aq) + NO 3 -(aq) k. J/mol ∆H° = +25

Entropy ■

Entropy ■ Processes that increase entropy tend to be spontaneous Think: Ø does a room stay organized if you do not put energy into maintaining it? Ø what happens to your notes if you do not organize them?

Gibb’s Free Energy ■ We have now seen that two factors – enthalpy and entropy – will determine whether, or not, a given physical or chemical event will be spontaneous. Sometimes these two factors work together. For example, when a stone wall crumbles, its enthalpy (potential energy) decreases and its entropy increases. Ø Since a decrease in enthalpy and an increase in entropy both favor a spontaneous change, the two factors complement one another. ■ In other situations, the effects of enthalpy and entropy are in opposition. Such is the case in the melting of ice or the vaporization of water.

Gibb’s Free Energy ■ When ENTHALPY AND ENTROPY EFFECTS OPPOSE EACH OTHER, the result of the interaction between them is not immediately obvious. However, we can discover the NET EFFECT of the two factors through the use of anothermodynamic quantity, called the GIBB’S FREE ENERGY, G. It is defined as: G = H – TS. For a CHANGE at CONSTANT TEMPERTURE and PRESSURE, the equation becomes: ■ What is of particular importance is that a CHANGE CAN ONLY BE SPONTANEOUS IF IT IS ACCOMPANIED BY A DECREASE IN FREE ENERGY. In other words, for a change to be spontaneous, G must be NEGATIVE.
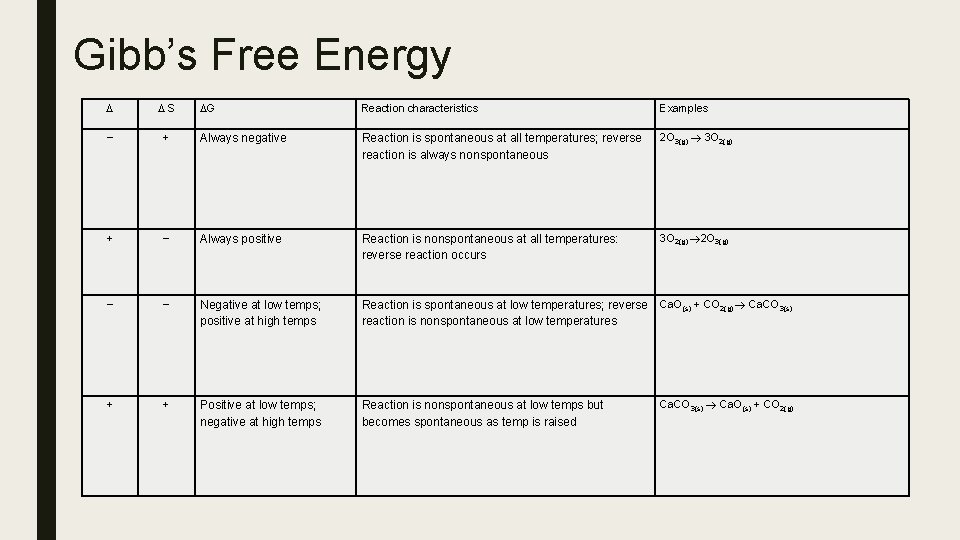
Gibb’s Free Energy S G Reaction characteristics Examples + Always negative Reaction is spontaneous at all temperatures; reverse reaction is always nonspontaneous 2 O 3(g) 3 O 2(g) + Always positive Reaction is nonspontaneous at all temperatures: reverse reaction occurs 3 O 2(g) 2 O 3(g) Negative at low temps; positive at high temps Reaction is spontaneous at low temperatures; reverse reaction is nonspontaneous at low temperatures Ca. O(s) + CO 2(g) Ca. CO 3(s) + + Positive at low temps; negative at high temps Reaction is nonspontaneous at low temps but becomes spontaneous as temp is raised Ca. CO 3(s) Ca. O(s) + CO 2(g)

Example: ■ Will a reaction with H = -256 k. J/mol and S = - 45. 0 J/mol. K be spontaneous at 67 o. C?

Example 2: ■ At what temperature will the melting of ice ( H = 6. 01 k. J/mol and S = 22. 0 J/mol. K) become spontaneous?
- Slides: 10