Spinal Cord Stimulators and Failed Back Syndrome Mark

Spinal Cord Stimulators and Failed Back Syndrome Mark Stern, MD Neurosurgeon

Topics • Part 1: Chronic Pain Conditions • Part 2: Spinal Cord Stimulation (SCS)/ Neurostimulation • Part 3: Prevention and Management of Neurostimulation Complications • Part 4: Intrathecal Drug Delivery (IDD)

Types of Neuropathic Pain • Direct nerve root injury: radiculopathy – Battered root syndrome – Perineural fibrosis – Intrafascicular fibrosis – Adhesive arachnoiditis • Peripheral deafferentation – Phantom limb pain – Sympathetic-mediated pain syndrome – Herpetic neuralgia – Diabetic polyneuropathy • Central deafferentation-thalamic stroke Cole AJ. In Low Back Pain Handbook, 2 nd ed. 2003; pg 361 -374

Types of Nociceptive Pain • Mechanical low back pain – Discogenic pain – Joint pain • Facet joint • Sacroiliac joint – Pseudoarthrosis – Osteoporosis – Musculoskeletal trauma Cole AJ. In Low Back Pain Handbook, 2 nd ed. 2003; pg 361 -374

Combined Nociceptive and Neuropathic Pain • Failed Back Surgery Syndrome (FBSS)* • Idiopathic chronic pain syndrome • Cancer pain *Also referred to as “post-surgical chronic back pain” Cole AJ. In Low Back Pain Handbook, 2 nd ed. 2003; pg 361 -374

Chronic Pain Conditions • Post-surgical chronic back pain or failed back • Radicular pain syndrome or radiculopathies resulting in pain secondary to failed back or herniated disk • Postlaminectomy pain • Multiple back operations • Unsuccessful disk surgery • Degenerative disk disease, herniated disk pain refractory to conservative and surgical interventions • Peripheral causalgia • Epidural fibrosis • Arachnoiditis or lumbar adhesive arachnoiditis • Complex regional pain syndrome, reflex sympathetic dystrophy or causalgia

Pain Treatment Ladder Device therapies are now considered earlier in the treatment continuum Stamatos JM, et al. Live Your Life Pain Free, October 2005. Based on the interventional pain management experience of Dr. John Stamatos

Part 2: Spinal Cord Stimulation / Neruostimulation
Spinal Cord Stimulation: Indication To aid in the management of chronic, intractable pain of the trunk and/or limbs-including unilateral or bilateral pain. MR 15993 © 2016 Medtronic, Inc. All Rights Reserved.

Choosing Candidates for Spinal Cord Stimulation Indicated for spinal cord stimulation (SCS) system as an aid in the management of chronic, intractable pain of the trunk and/or limbs-including unilateral or bilateral pain associated with the following conditions: § § § § § Failed Back Syndrome (FBS) or low back syndrome or failed back Radicular pain syndrome or radiculopathies resulting in pain secondary to FBS or herniated disk Postlaminectomy pain Multiple back operations Unsuccessful disk surgery Degenerative Disk Disease (DDD)/herniated disk pain refractory to conservative and surgical interventions Peripheral causalgia Epidural fibrosis Arachnoiditis or lumbar adhesive arachnoiditis Complex Regional Pain Syndrome (CRPS), Reflex Sympathetic Dystrophy (RSD), or causalgia

Choosing Candidates for Spinal Cord Stimulation § § § Chronic, intractable pain 1 Objective evidence of pathology 2 Lack of adequate relief from conventional treatments 3 Initial or further surgical intervention not indicated 3 At least 18 years of age 2 § § § Patient can properly operate the system 2 Patient understands therapy risks 2 Satisfactory screening test results 2 Patient is not pregnant 2 Completion of psychological evaluation 3 1. Medtronic, Inc. Indications, Implantable Neurostimulation Systems. 2004. 2. Medtronic, Inc. Medtronic Pain Therapy: Information for Prescribers. 2012. 3. Centers for Medicare & Medicaid Services. National Coverage Determination (NCD) for Electrical Nerve Stimulators (160. 7). Published August 7, 1995. Available at: https: //www. cms. gov/medicare-coverage-database/details/ncddetails. aspx? NCDId=240&ncdver=1&bc=AAAAg. AAAAA%3 d%3 d&. Accessed May 19, 2016.

Failed Back Surgery Syndrome (FBSS) • • • Defined as persistence or development • of low back or leg pain following surgery on the lumbosacral spine Most common indication for • neurostimulation therapy for chronic • pain in the United States FBSS occurs in patients who have typically undergone multiple lumbosacral spine operations for conditions such as disk herniation, lumbar stenosis, or spinal instability 1, 2, 3, 4 Approximately 300, 000 back surgeries are performed in USA/year to treat chronic lumbar pain 5 Failure rates as high as 40%5 Success rate decreases significantly with each subsequent back surgery 6

Complex Regional Pain Syndrome (CRPS) • • Second most common use of neurostimulation therapy for chronic pain in the United States is for the symptomatic management of complex regional pain syndrome (CRPS) CRPS is a neuropathic pain syndrome precipitated most commonly by minor limb trauma 1, 2, 4 Continuous, severe pain, disproportionate to the inciting event, occurs in the limb and may be accompanied by allodynia, hyperalgesia, skin color changes, edema, joint stiffness, and bone demineralization 1, 2, 3, 4 Type I (reflex sympathetic dystrophy) Minor injuries or fractures of a limb precede the onset of symptoms Type II (causalgia) Develops after injury to a major peripheral nerve
Contraindications for Spinal Cord Stimulation Diathermy- Do not use shortwave diathermy, microwave or therapeutic ultrasound diathermy on patients implanted with a neurostimulation system. Energy from diathermy can be transferred through the implanted system and cause tissue damage at the locations of the implanted electrodes, resulting in severe injury or death. Refer to the package labeling for a complete list of indications and contraindications for comprehensive details regarding product safety.
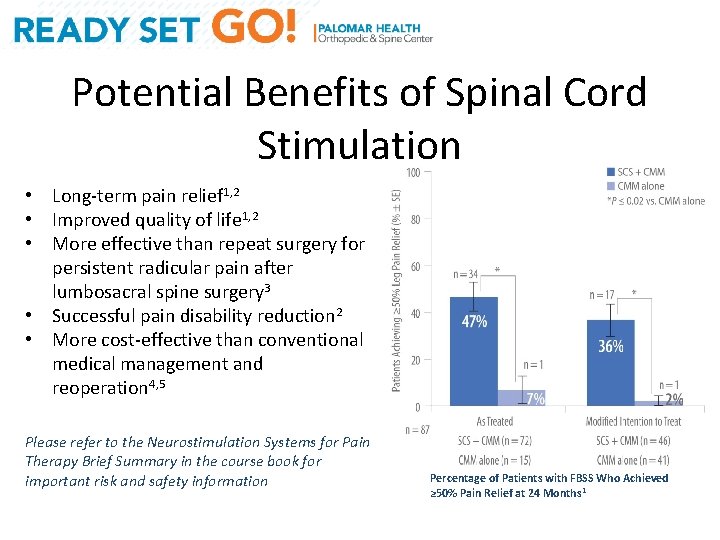
Potential Benefits of Spinal Cord Stimulation • Long-term pain relief 1, 2 • Improved quality of life 1, 2 • More effective than repeat surgery for persistent radicular pain after lumbosacral spine surgery 3 • Successful pain disability reduction 2 • More cost-effective than conventional medical management and reoperation 4, 5 Please refer to the Neurostimulation Systems for Pain Therapy Brief Summary in the course book for important risk and safety information Percentage of Patients with FBSS Who Achieved ≥ 50% Pain Relief at 24 Months 1

Medtronic Registry Product 1 Performance-related Events for SCS Leada, b § § § § Lead migration/dislodgements (n=314) High impedance (n=78) Lead fracture (n=46) Undesirable change in stimulation (n=43) Medical device complication (n=25) Low impedance (n=17) Device malfunction (n=11) Neurostimulatorc § § § § Medical device complication (n=3) Device malfunction (n=2) Undesirable change in stimulation (n=2) High impedance (n=1) Paresthesia (n=1) Recharging unable to recharge (n=1) Broken bond wire (n=1) a. 4962 leads followed from June 2004 through July 31, 2013 b. Includes events with 5 or more occurrences c. 2841 neurostimulators followed from June 2004 through July 31, 2013 1. Medtronic Product Performance Report 2013. For a complete list of adverse events which have been associated with SCS, refer to the manufacturer labeling for the specific device.

Importance of Patient Selection § Appropriate patient selection is critical for: – Therapy success – Patient satisfaction § Goal of patient selection – Choose patients most likely to experience therapeutic success – Minimize likelihood of risks, complications, and adverse events

Part 3: Prevention and Management of Neurostimulation Complications
Complications in Neurostimulation for Chronic Pain § Preoperative considerations § Intraoperative considerations § Postoperative considerations

PREOPERATIVE

Informed Consent: Risks § Undesirable change in stimulation described by some patients as uncomfortable, jolting or shocking § Hematoma § Epidural hemorrhage § Paralysis § Seroma § CSF leakage § § § § Infection Erosion Allergic response Hardware malfunction or migration Pain at implant site Loss of pain relief Chest wall stimulation Surgical risks For additional safety information, please refer to the brief summary of safety information at www. professional. medtronic. com For current product advisories, refer to http: //professional. medtronic. com/pt/neuro/scs/ind/productadvisories/index. htm

Informed Consent Discussion § Risk-based discussion § Discuss both early and late complications § Provide information on the frequency and severity of event, identifying those which are most common, and how the events may be prevented or managed

Physiological Considerations • Difficult Anatomy – Scoliosis, Morbid Obesity, Prior Surgery – Inability to access the epidural space or place the lead • Pacemaker Implantation – Need for intra-op and post-op cardiac monitoring – Recommendation for pacemaker representative involvement • Infection – – History of MRSA Systemic vs. local infection Diabetes SMOKING • Anticoagulation • Future medical requirements (ie, MRI compatibility)

INTRAOPERATIVE

Intraoperative Considerations – Needle insertion angle (< 45) – Dural puncture – LOR to air • Pneumocephalus – Lead placement – – • Intrathecal • Anterior epidural • Lateral epidural Anchoring Strain relief loop of lead Strict hemostasis IPG placement • Depth and anchoring – Wound closure

Postoperative • Hardware • Biological • Stimulation

n= 42 to 2972 Incidence of Hardware Complications 1 Complication Rate* Lead migration 1, 2, 4, 5, 7, 8, 9 4. 89% to 22. 6% Loose connection 2, 7, 10 0. 58% - 9. 5% Lead wire breakage 1, 2, 4, 5, 7, 8, 10 0. 58% - 9. 1% Hardware malfunction 1, 2, 5, 7, 10 2. 0% - 13% Battery failure 1, 2, 9 0. 92% - 1. 6% *Rate is based on the number of subjects implanted.
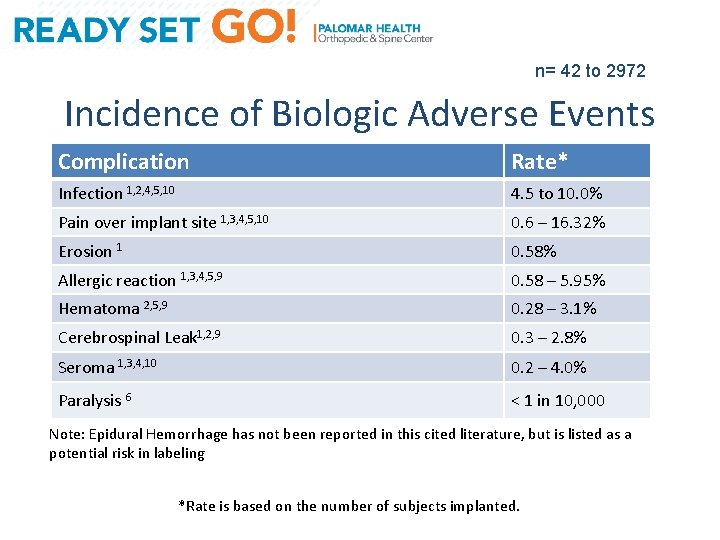
n= 42 to 2972 Incidence of Biologic Adverse Events Complication Rate* Infection 1, 2, 4, 5, 10 4. 5 to 10. 0% Pain over implant site 1, 3, 4, 5, 10 0. 6 – 16. 32% Erosion 1 0. 58% Allergic reaction 1, 3, 4, 5, 9 0. 58 – 5. 95% Hematoma 2, 5, 9 0. 28 – 3. 1% Cerebrospinal Leak 1, 2, 9 0. 3 – 2. 8% Seroma 1, 3, 4, 10 0. 2 – 4. 0% Paralysis 6 ˂ 1 in 10, 000 Note: Epidural Hemorrhage has not been reported in this cited literature, but is listed as a potential risk in labeling *Rate is based on the number of subjects implanted.
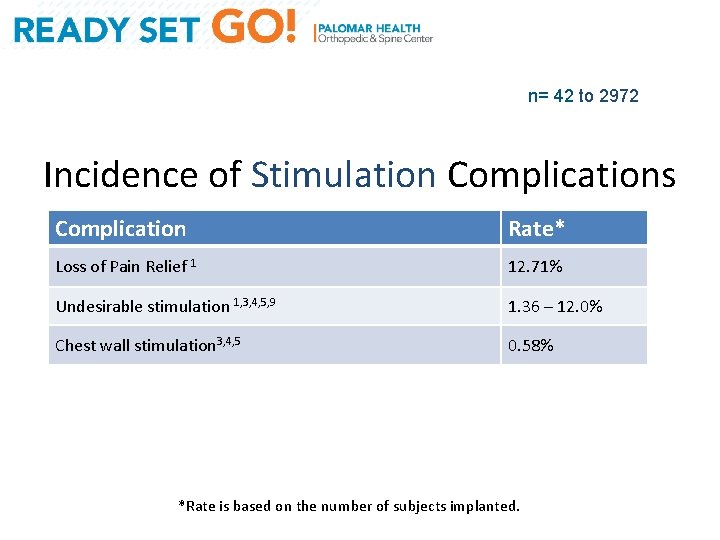
n= 42 to 2972 Incidence of Stimulation Complications Complication Rate* Loss of Pain Relief 1 12. 71% Undesirable stimulation 1, 3, 4, 5, 9 1. 36 – 12. 0% Chest wall stimulation 3, 4, 5 0. 58% *Rate is based on the number of subjects implanted.

BEST PRACTICES TO MINIMIZE AND MANAGE COMPLICATIONS

Minimizing Infection • Several weeks before surgery – Manage coexisting conditions & risks • Control blood glucose • Recognize and treat remote infections – Consider consultation with an infectious disease specialist • Reduce or eliminate smoking • Manage anticoagulation – Discuss surgical risks and management with the patient • 1 -2 days before surgery – Patient instructed to bath/clean surgical sites with a antimicrobial scrub Follett KA, Boortz-Marx RL, Drake JM, et al. 2004.

Minimizing Infection Perioperative: 1 hour, administer prophylactic antibiotic 1 Intraoperative 1, 2 • Operating room • During procedure – Drape fluoroscopic C-arm within – Minimize number of people sterile field – Double-glove, minimal touch – Limit entry & exit to OR – Strict hemostasis – Minimize tissue trauma • Patient prep – Scrub with antiseptic solution – Use antimicrobial incision drape – Drape large sterile field 1 Follett 2 Deer • Closing the incision – Suture in anatomical layers – Apply sterile occlusive dressing KA, Boortz-Marx RL, Drake JM, et al. 2004. TR, Stewart CD. 2008.

Device Selection and Implant Techniques • Complications that may be minimized through device selection and implant technique best practices – Undesirable stimulation – Lead migration – Lead wire breakage – Connection problems Kumar K, Buchser E, Linderoth B, et al. 2007.

Minimizing Undesirable Stimulation with Restore. Sensor ® and Adaptive. Stim ® § Adaptive. Stim technology addresses these needs by automatically changing amplitude as needed in appropriate patients § Restore. Sensor with Adaptive. Stim § Automatically detects changes in body position § Adapts stimulation settings • to patient preferences • up to 6 positions

Implant Technique: Minimizing Lead Migration and Wire Breakage • Lead placement 1 – Patient prone, pillow – Conscious sedation – Both AP & lateral images • Adequate length for lead & extension • Percutaneous lead – Needle oentry: paramedian oblique, less than 45 angle 1 – Soft silicone anchor 2 • Anchor end is pushed through fascia 2 1 Kumar K, Buchser E, Linderoth B, et al. 2007. JM, Schade CM, Sasaki J, et al. 2006. 2 Henderson Injex. TM Anchors

Implant Technique: Minimizing Lead Migration and Wire Breakage (continued) • Strain relief loop 1 – Nonabsorbable sutures 2 – Circular loop 1, greater than 2 cm diameter • Surgical lead: instead of anchor 1 • Percutaneous lead: between anchor and extension 1 • Neurostimulator in abdominal wall 1 1 Kumar K, Buchser E, Linderoth B, et al. 2007. JM, Schade CM, Sasaki J, et al. 2006. 2 Henderson

Implant Technique: Minimizing Connection Problems • Use adequate length for lead & extension – Some physicians prefer a longer lead to avoid a lead-extension connection 1 • Tighten setscrews securely; use appropriate wrench • Protect connections from body fluids – Lead to extension - use tissue adhesive in boot 2 – Neurostimulator • eg, Make sure self-sealing grommet closes after wrench removed 1 Renard 2 Kumar VM, North RB. 2006. K, Wilson JR, Taylor RS, et al. 2006.
Minimizing Pain and Skin Erosion at Anchor & Extension Connection • Select appropriate anchor 1 • Anchor at unobstructed location 1 – Not over bony structures • Secure anchor to deep fascia • Bury extension connector in an existing pocket (anchor or neurostimulator)2 Injex. TM Bi-wing Anchor – Not in a new pocket 1 Kumar K, Wilson JR, Taylor RS, et al. 2006. VM, North RB. 2006. 2 Renard
Minimizing Pain and Skin Erosion at Neurostimulator Site • Neurostimulator location – Away from obstructions 1, 2 • eg, not over rib cage; avoid belt line and other clothing restrictions – No deeper than specified in product labeling • Neurostimulator pocket – Blunt dissection – Snug fit 2 – Secure neurostimulator 1 Kumar 2 Follett K, Wilson JR, Taylor RS, et al. 2006. KA, Boortz-Marx RL, Drake JM, et al. 2004.

Management of Postsurgical Complications Complication Management • CSF leak: dural puncture • Conservative management 1 – Flat bed rest, hydrate, analgesics – Caffeine – Abdominal binder • Some physicians perform epidural blood patch 1 • Subcutaneous hematoma (or seroma) • Apply binder 2 • May resolve spontaneously 3 • May need to aspirate 2, 3 1 Eldrige JS, Weingarten TN, Rho RH. 2006. TR, Stewart CD. 2008. 3 Kumar K, Wilson JR, Taylor RS, et al. 2006. 2 Deer

Management of Postsurgical Complications 1 Complication Management • Pain over component • May improve with conservative measures 1 – Lidocaine patch – Cushion site • Surgically revise component 1 – Deeper, if appropriate – Away from irritation • Revise component 2 • Skin erosion over component – Deeper, if appropriate 1 – Away from irritation • Consider antibiotics 1 Deer TR, Stewart CD. 2008. KA, Boortz-Marx RL, Drake JM, et al. . 2004. 2 Follett

Management of System Complications Complication Management • Lead migration • Reprogram • Revise surgically • Lead wires broken • Replace lead • Hardware malfunction • Replace component • Undesirable paresthesia • Depending on cause: • • Reprogram Educate patient Replace component Revise surgically • Battery failure • Replace component • Revise surgically • Loose connection • Revise surgically

Part 4: Intrathecal Drug Delivery (IDD)
• Intrathecal Drug Delivery (IDD) Therapy IDD therapy involves the delivery of pain medicine in the intrathecal space • The pump is connected to a thin, flexible catheter; both are implanted under the skin • Smaller doses of medication are needed for effective pain relief because drug is delivered directly to the pain receptors

Synchromed® II Drug Infusion System Indications • Chronic intrathecal infusion of preservative-free morphine sulfate sterile solution in the treatment of chronic intractable pain • Also indicated for chronic intrathecal infusion of Lioresal® Intrathecal (baclofen injection) for severe spasticity, chronic epidural infusion of preservativefree morphine sulfate sterile solution in the treatment of chronic intractable pain, and chronic intrathecal infusion of preservative-free ziconotide sterile solution for the management of severe chronic pain Lioresal® is a registered trademark of Novartis Pharmaceuticals, Inc.

Approximate Equivalent Daily Doses of Morphine Route of Administration Relative Potency (mg)* Oral Intravenous Epidural Intrathecal *Relative approximations based on clinical observations 300 100 20 1

Benefits of IDD Therapy • Pain relief for patients who have not received adequate relief with conventional therapies • Reduction in adverse effects from oral opioids such as nausea, vomiting, sedation, and constipation • Decreased or elimination of oral analgesics • Increased ability to perform activities of daily living • Patient control within physician-set limits • May be effective for patients who do not experience relief from neurostimulation therapy Lamer TJ: Mayo Clin Proc. May 1994; 69(5): 473 -80. ; Paice JA, et al: J Pain Symptom Manage. 1996; 11(2): 71 -80.

IDD Trial • The purpose of the trial is to assess the efficacy and side effects of intrathecal morphine • Trialing methods include – Continuous epidural – Continuous intrathecal – Bolus epidural – Bolus intrathecal • Patients who report at least 50% reduction in pain may be candidates for long-term therapy

IDD Therapy Risks • • • Programming error Drug concentration error Surgical complications, such as infection Catheter could become dislodged or blocked The pump could stop working Other side effects may occur For complete list of risks/complications, refer to product package insert Onofrio BM, Yaksh TL. J Neurosurg 1990; 72: 200 -209. ; Winkelmueller M, Winkelmueller W. J Neurosurg 1996; 85: 458467. ; Paice JA, Penn RD, Shott S. J Pain Symptom Manage 1996; 11(2): 71 -80.

Patient Selection

Indications for Neurostimulation and Intrathecal Drug Delivery Therapy Cole AJ. In Low Back Pain Handbook, 2 nd ed. 2003; pg 362. Refer to full prescribing information for Medtronic Neurostimulation Systems and Synchromed® II and Isomed® Drug Infusion Systems

Patient Selection Considerations • Patients who have neuropathic pain in a concordant anatomic distribution respond best to neurostimulation (NS) therapy • Patients who have nociceptive pain in a concordant distribution respond best to Intrathecal Drug Delivery (IDD) • Patients who do not respond well to NS may be candidates for IDD therapy Cole AJ. In Low Back Pain Handbook, 2 nd ed. 2003; pg 362.

Patient Selection Checklist ü Failure of oral/transdermal opiate use or undesirable side effects ü More conservative therapies have failed ü An observable pathology exists that is concordant with the pain complaint ü Further surgical intervention is not indicated ü No serious untreated drug habituation exists ü Psychological evaluation and clearance for implantation has been obtained ü No contraindications to implantation exist ü Refer Patient for a Pain Therapy Trial Cole AJ. In Low Back Pain Handbook, 2 nd ed. 2003; pg 362.

Neurostimulation Clinical Evidence

Medtronic PROCESS Study • Primary outcome – Number of patients with ≥ 50% leg pain relief at 6 months (≥ 50% reduction in leg VAS) • Secondary outcomes evaluated at 1, 3, 6, 9, 12, 18 and 24 months – Pain relief (leg and axial back VAS) – Quality of life (SF-36 and EQ-5 D) – Function (Oswestry Disability Index) – Patient satisfaction – Need for drug or non-drug therapy for pain – Time away from work – Adverse events Kumar, et al. Pain 2007
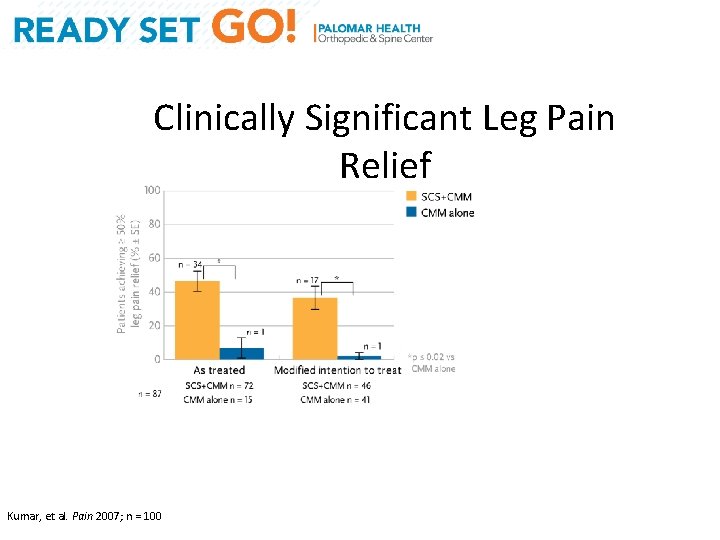
Clinically Significant Leg Pain Relief Key Findings: ≥ 50% leg pain relief at 24 months, continued greater effect with SCS* in the per treatment/ITT analyses over 24 months *SCS (spinal cord stimulation) is a term for neurostimulation Kumar, et al. Pain 2007; n = 100
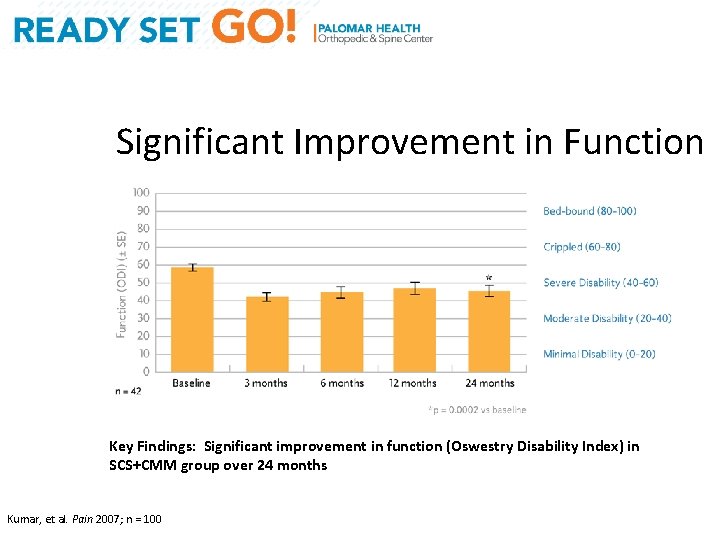
Significant Improvement in Function Key Findings: Significant improvement in function (Oswestry Disability Index) in SCS+CMM group over 24 months Kumar, et al. Pain 2007; n = 100
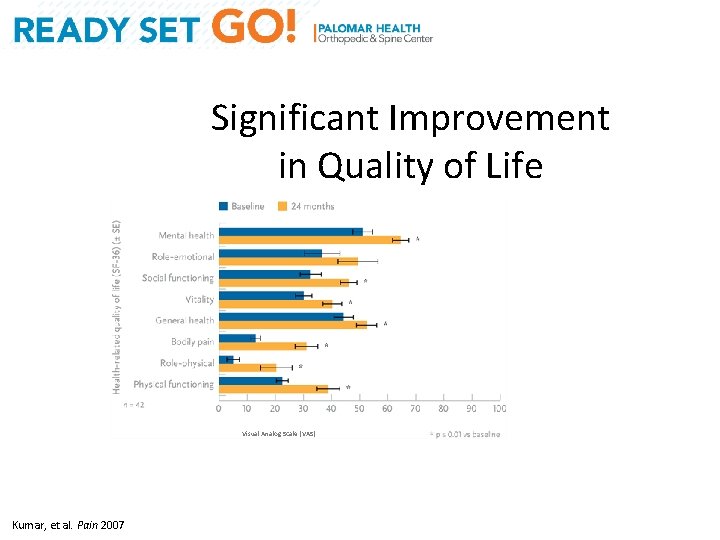
Significant Improvement in Quality of Life Key Findings: Significant improvement in SCS+CMM group in 7/8 domains of Qo. L (SF-36) over 24 months Visual Analog Scale (VAS) Kumar, et al. Pain 2007
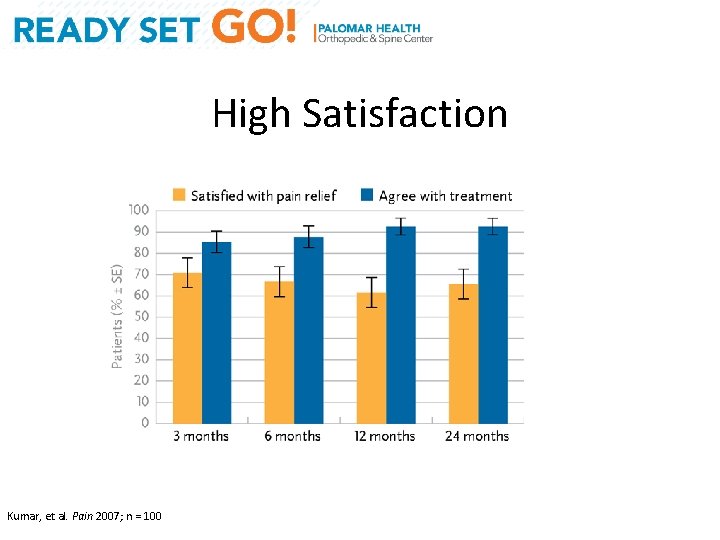
High Satisfaction Key Findings: Treatment satisfaction among patients continuing SCS+CMM at 24 months Kumar, et al. Pain 2007; n = 100
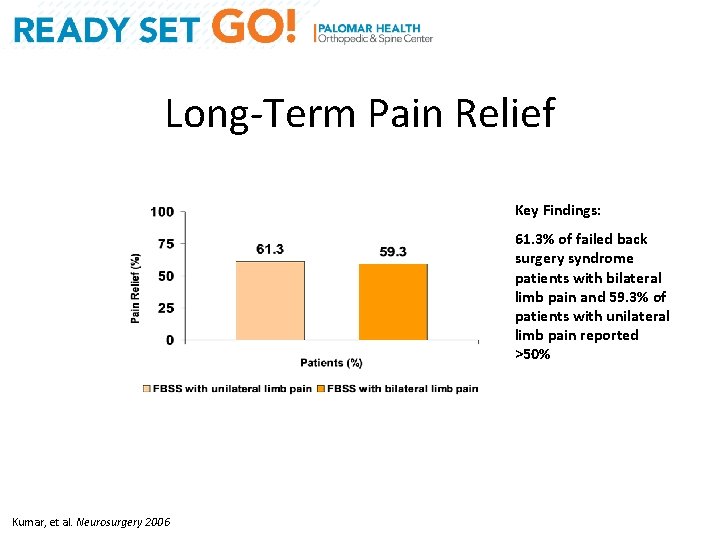
Long-Term Pain Relief Key Findings: 61. 3% of failed back surgery syndrome patients with bilateral limb pain and 59. 3% of patients with unilateral limb pain reported >50% Kumar, et al. Neurosurgery 2006
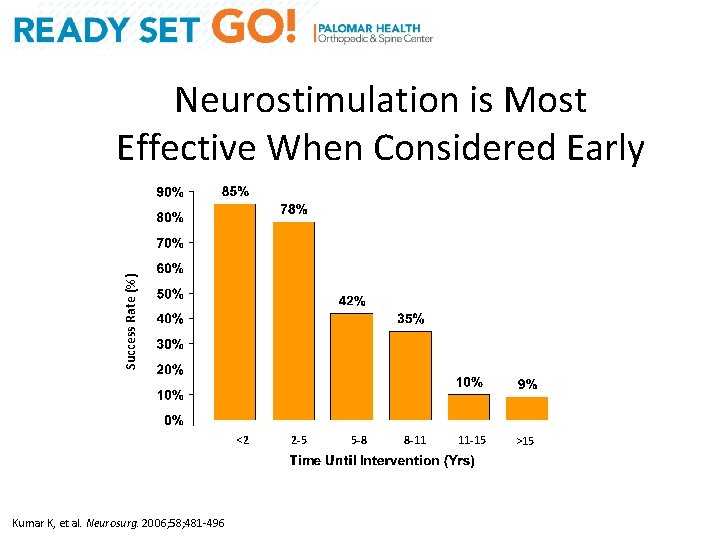
Success Rate (%) Neurostimulation is Most Effective When Considered Early <2 Kumar K, et al. Neurosurg. 2006; 58; 481 -496 2 -5 5 -8 8 -11 11 -15 >15
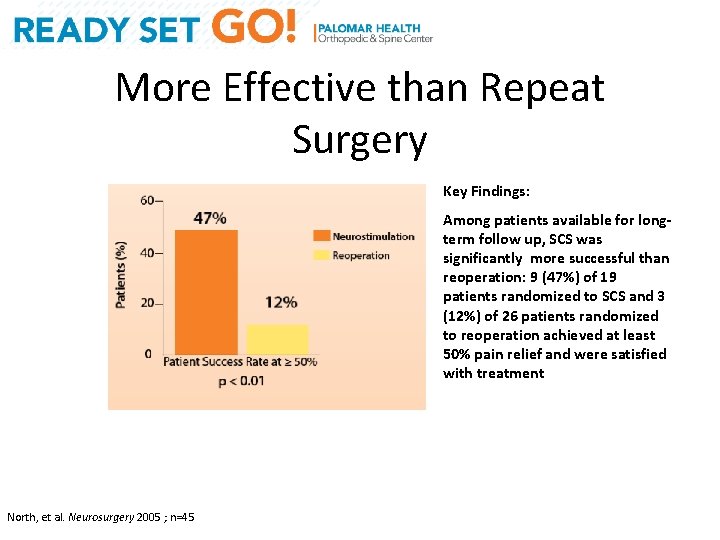
More Effective than Repeat Surgery Key Findings: Among patients available for longterm follow up, SCS was significantly more successful than reoperation: 9 (47%) of 19 patients randomized to SCS and 3 (12%) of 26 patients randomized to reoperation achieved at least 50% pain relief and were satisfied with treatment North, et al. Neurosurgery 2005 ; n=45
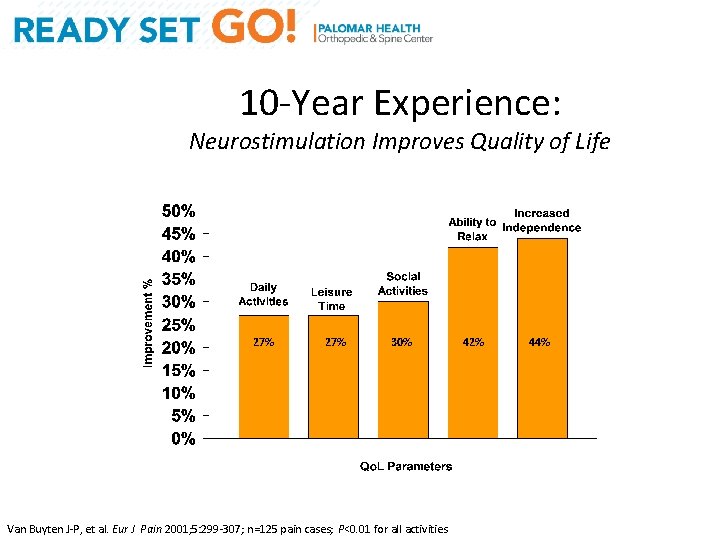
10 -Year Experience: Neurostimulation Improves Quality of Life 27% 30% Van Buyten J-P, et al. Eur J Pain 2001; 5: 299 -307; n=125 pain cases; P<0. 01 for all activities 42% 44%

Intrathecal Drug Delivery Clinical Evidence
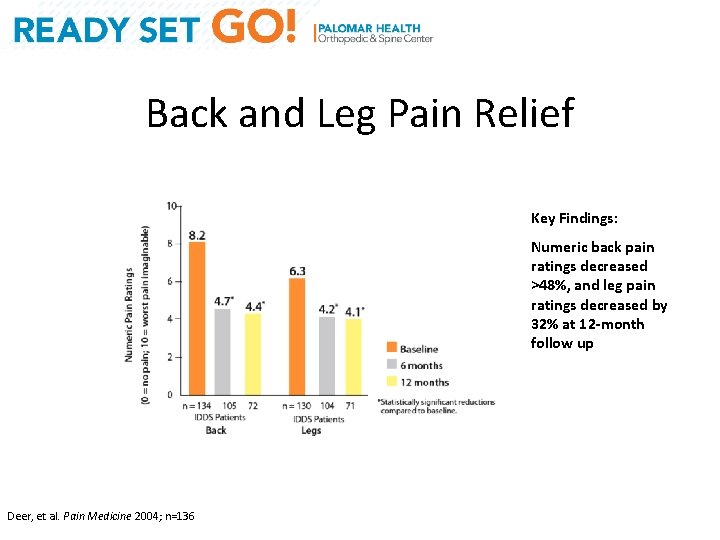
Back and Leg Pain Relief Key Findings: Numeric back pain ratings decreased >48%, and leg pain ratings decreased by 32% at 12 -month follow up Deer, et al. Pain Medicine 2004; n=136
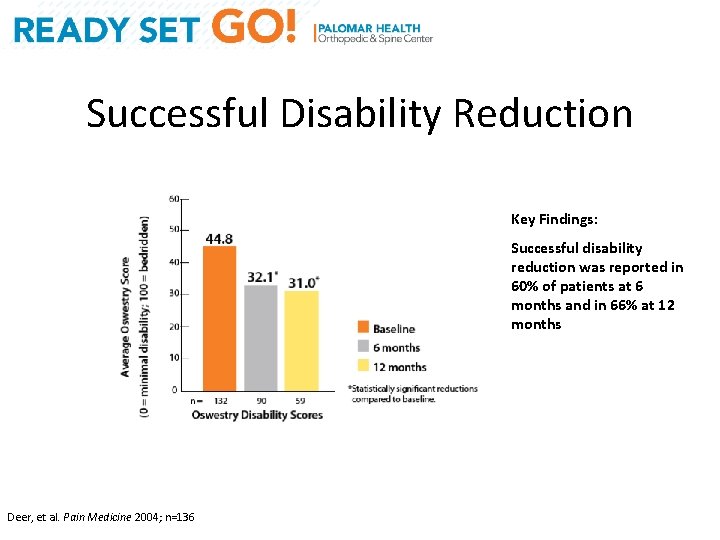
Successful Disability Reduction Key Findings: Successful disability reduction was reported in 60% of patients at 6 months and in 66% at 12 months Deer, et al. Pain Medicine 2004; n=136
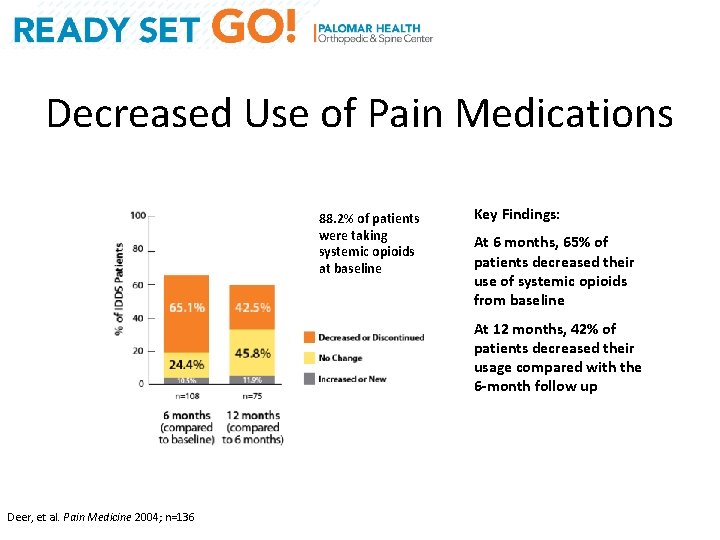
Decreased Use of Pain Medications 88. 2% of patients were taking systemic opioids at baseline Key Findings: At 6 months, 65% of patients decreased their use of systemic opioids from baseline At 12 months, 42% of patients decreased their usage compared with the 6 -month follow up Deer, et al. Pain Medicine 2004; n=136
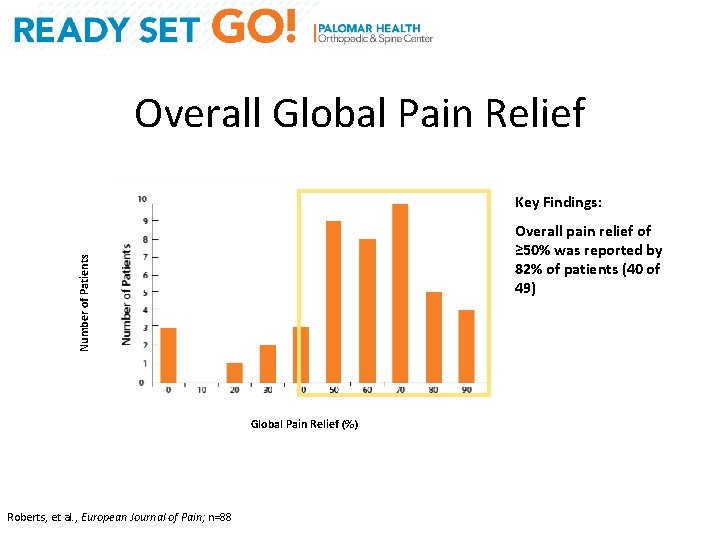
Overall Global Pain Relief Key Findings: Number of Patients Overall pain relief of ≥ 50% was reported by 82% of patients (40 of 49) Global Pain Relief (%) Roberts, et al. , European Journal of Pain; n=88

Clinical Evidence Risks As with any surgical procedure, neurostimulation and IDD involve the risk of infection. Lead migration is the most common complication associated with neurostimulation, 1 while intrathecal catheters can fracture, kink, and migrate. 2 For a complete list of adverse events for implantable therapies, see the appropriate product labeling. 1. Cameron T. Safety and efficacy of spinal cord stimulation for the treatment of chronic pain: a 20 -year literature review J Neurosrug (Spine 3) 100: 254 -267, 2004; 2. Staats P. Complications of intrathecal therapy. Pain Medicine 2008; 9(S 1): S 102 -S 107

Summary • With today’s treatments, patients should not have to wait for effective pain relief • By partnering, we can help patients find the right pain treatment • Together we can improve the quality of life for chronic pain patients • Our decisions may change the course of a patient’s life

Case Study Male, 45, office manager, no major psychosocial issues • • • One spine surgery to treat herniated disc Referred from primary care physician to address axial back pain and secondary radicular pain that persists six months following anatomically corrective surgery Average back pain score (VAS) of 80/100 with diminished functional capacity Relief from physical rehabilitation therapy was not satisfactory Unsuccessfully treated with neuropathic pain agents and two systemic opioids, patient experienced extreme sedation and constipation Treated with nerve block series but relief was temporary
- Slides: 71