Spin Hamiltonian for a Pair H B B

Spin Hamiltonian for a Pair H= B B. g 1. S 1+ S 1. D 1. S 1+ j S 1. A 1 j. Ij+. . + B B. g 2. S 2+ S 2. D 2. S 2+ j S 2. A 2 j. Ij+. . +S 1. J 12. S 2 = J 12 S 1. S 2+ S 1. D 12. S 2+ d 12. S 1 x. S 2 isotropic anisotropic Spin-spin interaction antisymmetric

SH Parameters for Pairs In the strong exchange limit, J>>D, d the total spin S=S 1+S 2 is a good quantum number: g S= c 1 g 1+ c 2 g 2 A S= c 1 A 1+ c 2 A 2 DS= d 1 D 1+ d 2 D 2+ d 12 D 12 c 1=(1+c)/2; c 2= (1 -c)/2; d 1= (c++c-)/2; d 2= (c+-c-)/2; d 12= (1 -c+)/2

Coupling coefficients

Some numerical coefficients S 1 S 2 S c 1 c 2 d 1 d 2 d 12 1/2 1 1/2 0 0 1/2 1/2 -1/2 1 1 1 2 1/2 1/6 1/3 3/2 1 1/2 -6/5 17/10 3/2 2 1/2 0 0 1/2 3/2 3 1/2 1/5 3/10
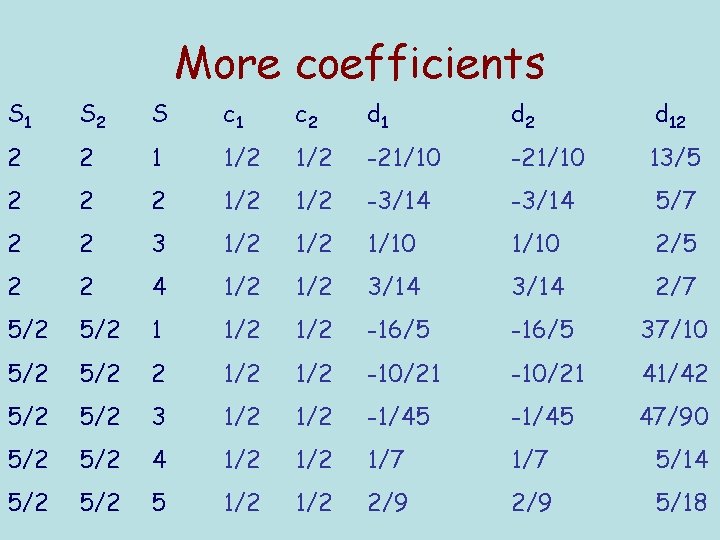
More coefficients S 1 S 2 S c 1 c 2 d 1 d 2 d 12 2 2 1 1/2 -21/10 13/5 2 2 2 1/2 -3/14 5/7 2 2 3 1/2 1/10 2/5 2 2 4 1/2 3/14 2/7 5/2 1 1/2 -16/5 37/10 5/2 2 1/2 -10/21 41/42 5/2 3 1/2 -1/45 47/90 5/2 4 1/2 1/7 5/14 5/2 5 1/2 2/9 5/18
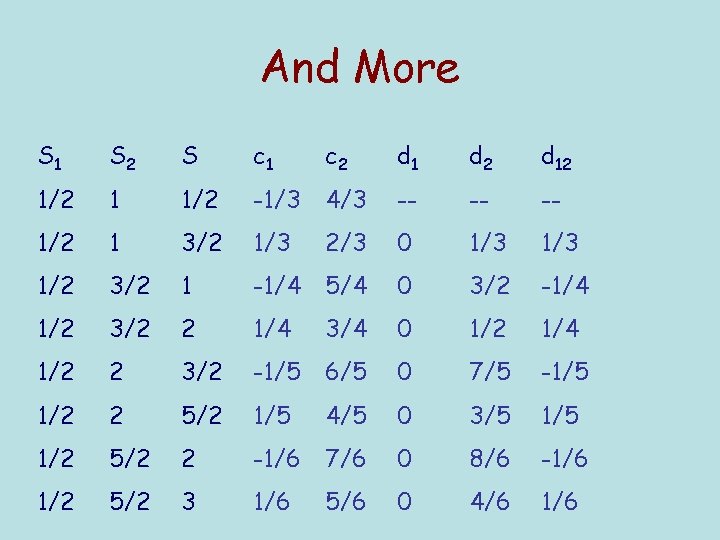
And More S 1 S 2 S c 1 c 2 d 1 d 2 d 12 1/2 1 1/2 -1/3 4/3 -- -- -- 1/2 1 3/2 1/3 2/3 0 1/3 1/2 3/2 1 -1/4 5/4 0 3/2 -1/4 1/2 3/2 2 1/4 3/4 0 1/2 1/4 1/2 2 3/2 -1/5 6/5 0 7/5 -1/5 1/2 2 5/2 1/5 4/5 0 3/5 1/2 5/2 2 -1/6 7/6 0 8/6 -1/6 1/2 5/2 3 1/6 5/6 0 4/6 1/6

Origin of the Spin-spin interaction • Through space (magnetic dipolar) • Through bonds (exchange)
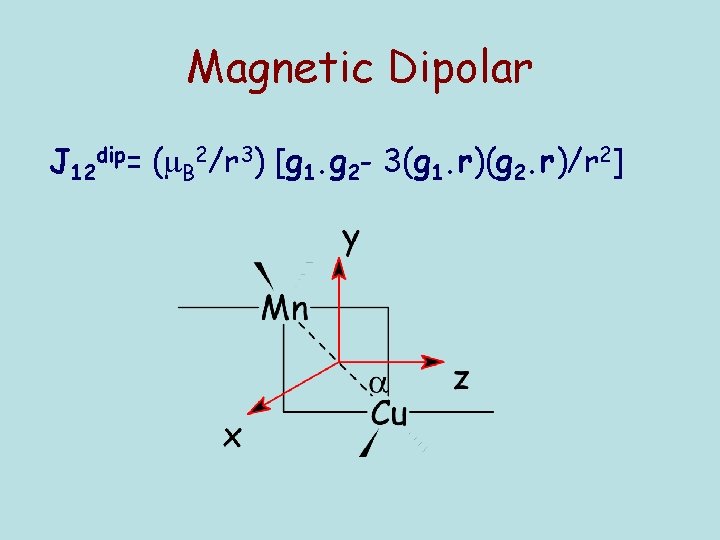
Magnetic Dipolar J 12 dip= ( B 2/r 3) [g 1. g 2 - 3(g 1. r)(g 2. r)/r 2]

Dipolar matrix in B 2/r 3 units gxxge 0 0 0 gyyge(1 -3 sin 2 ) -3 sin cos gyyge 0 -3 sin cos gzzge(1 -3 cos 2 )

Decomposition of the interaction matrix J= (1/3)(Jxx+Jyy+Jzz) dxx=(Jyz-Jzy)/2 Dij=(Jij+Jji)/2

Dipolar interaction calculated r=2. 5 Å r=3. 5 Å r=4. 5 Å 7 3 J 18 D -3519 E 28 11 5 dx -83 -30 -14 -1283 -603 The values are given in 10 -4 cm-1. gxx=gyy=2. 2; gzz=2. 0. The principal direction of D is parallel to the Mn-Cu direction

Origin of the Exchange Contributions J <g 1 g 2 Hex g 1 g 2> D <n 1 g 2 Hex n 1 g 2> 2/ 2 D ( g/g)2 J d <n 1 g 2 Hex g 1 g 2> / d ( g/g)J

Potential exchange- the case of non-degenerate terms 1. One half-filled orbital per ion: s-s molecule: A B 2. Non-degenerate terms: The effective Hamiltonian of the exchange interaction: one orbital per center: Exchange integral (potential energy): Many-electron exchange parameter (all bi-orbital interactions, half-filled orbitals): The effective Hamiltonian of the exchange interaction: many orbitals per center: Heisenberg-Dirac- Van Vleck model (HDVV model)
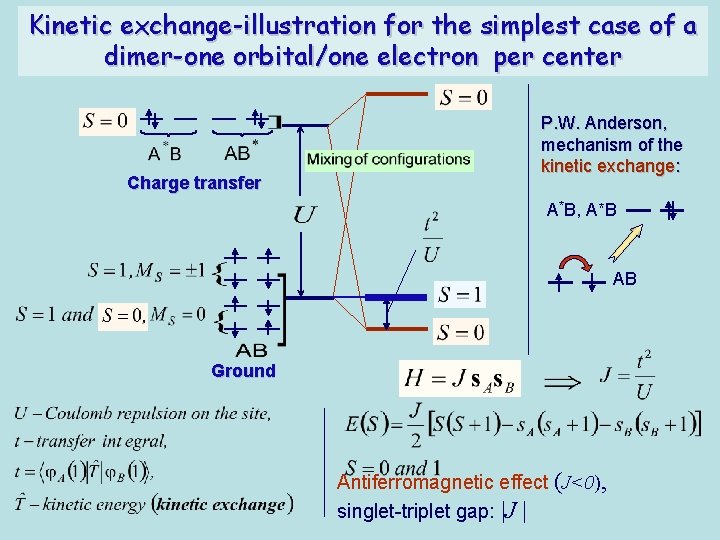
Kinetic exchange-illustration for the simplest case of a dimer-one orbital/one electron per center Charge transfer P. W. Anderson, mechanism of the kinetic exchange: A*B, A*B AB Ground Antiferromagnetic effect (J<0), singlet-triplet gap: |J |

Heisenberg-Dirac-Van Vleck (HDVV) model -parameter of the isotropic exchange, incorporates contributions of all mechanisms: Full spin S numerates the energy levels (“good” quantum number): Lande’s rule for the intervals: Further generalization: summation over all pairwise interactions ij in many-electron ions with full spins Si and Sj Zeeman interaction (orbital part disappears in HDVV model): This result holds for any direction of the magnetic field H HDVV- isotropic model
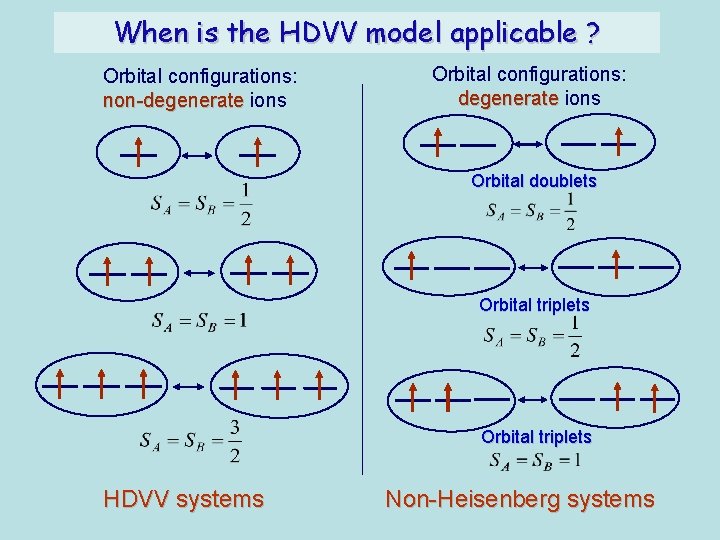
When is the HDVV model applicable ? Orbital configurations: non-degenerate ions Orbital configurations: degenerate ions Orbital doublets Orbital triplets HDVV systems Non-Heisenberg systems

Heisenberg-Dirac-Van Vleck (HDVV) model The main condition of applicability-orbitally nondegenerate well isolated ground term in crystal field Under this condition the orbital angular momentum is strongly reduced and the anisotropic terms are relatively small (second and higher order corrections): Antisymmetric exchange: Local anisotropy: High order isotropic terms: biquadratic exchange, symmetric part of the anisotropic exchange tensor, etc HDVV model isotropic interactions >>anisotropic interactions

Il modello di Anderson A-C-B →A+-C-B- Lo scambio cinetico favorisce il singoletto Lo scambio potenziale il tripletto

Regole di Goodenough-Kanamori • Se gli orbitali magnetici si sovrappongono l’accoppiamento è antiferromagnetico • Se gli orbitali magnetici sono ortogonali ed hanno ragionevoli zone di sovrapposizione lo scambio è ferromagnetico • Se un orbitale magnetico sovrappone con un orbitale vuoto l’accoppiamento è ferromagnetico

Interazione di scambio Orbitali magnetici (quelli che hanno l’elettrone spaiato) con sovrapposizione diversa da zero: accoppiamento antiferromagnetico
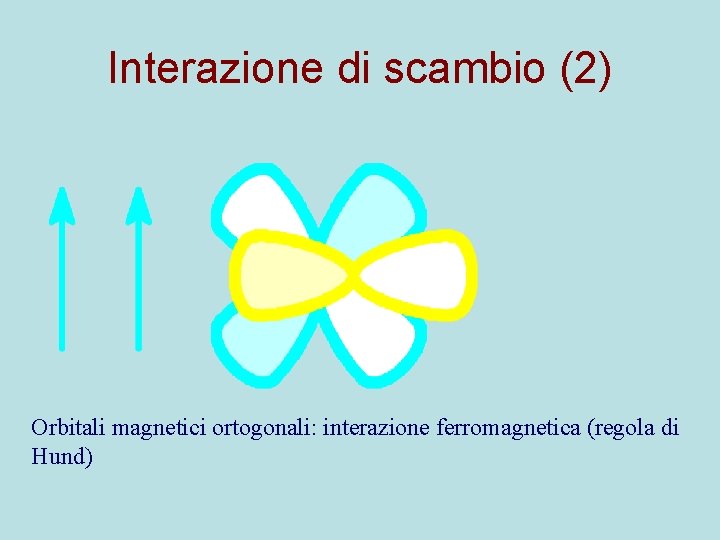
Interazione di scambio (2) Orbitali magnetici ortogonali: interazione ferromagnetica (regola di Hund)
Interazione di superscambio

Interazione di superscambio (2)
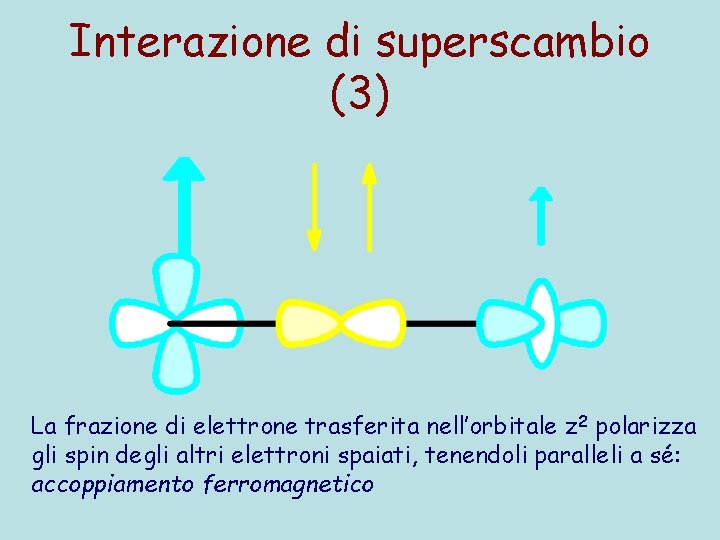
Interazione di superscambio (3) La frazione di elettrone trasferita nell’orbitale z 2 polarizza gli spin degli altri elettroni spaiati, tenendoli paralleli a sé: accoppiamento ferromagnetico
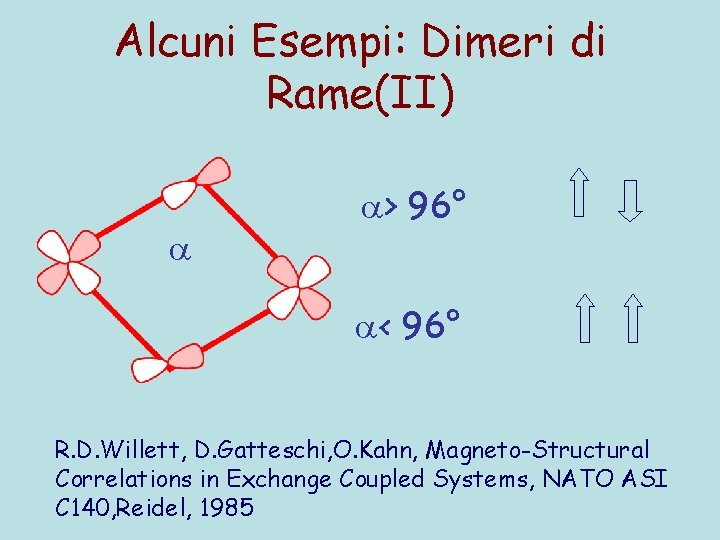
Alcuni Esempi: Dimeri di Rame(II) > 96° < 96° R. D. Willett, D. Gatteschi, O. Kahn, Magneto-Structural Correlations in Exchange Coupled Systems, NATO ASI C 140, Reidel, 1985
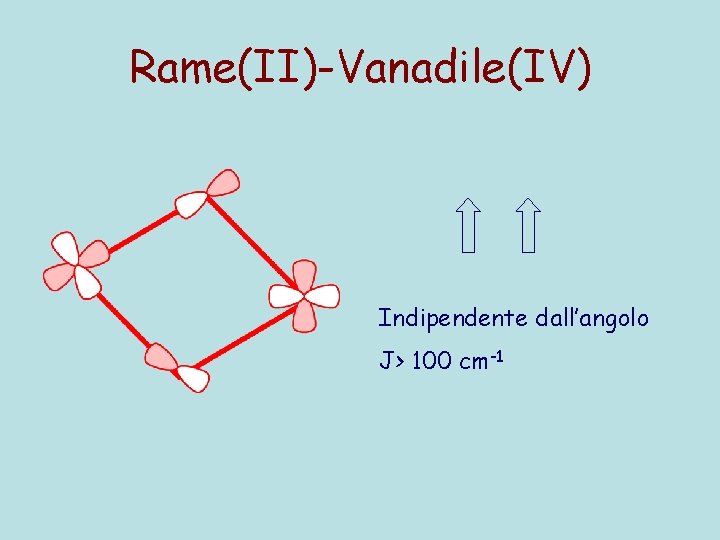
Rame(II)-Vanadile(IV) Indipendente dall’angolo J> 100 cm-1
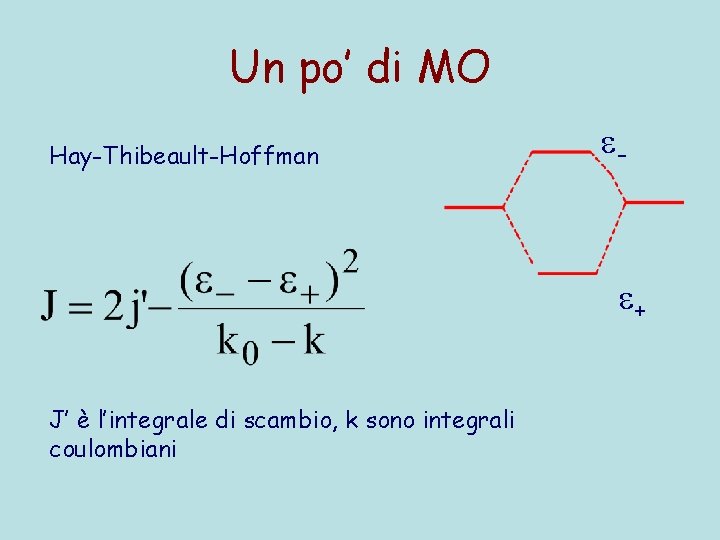
Un po’ di MO Hay-Thibeault-Hoffman - + J’ è l’integrale di scambio, k sono integrali coulombiani

Il modello di Kahn J=j-ks 2 J integrale di scambio s integrale di sovrapposizione
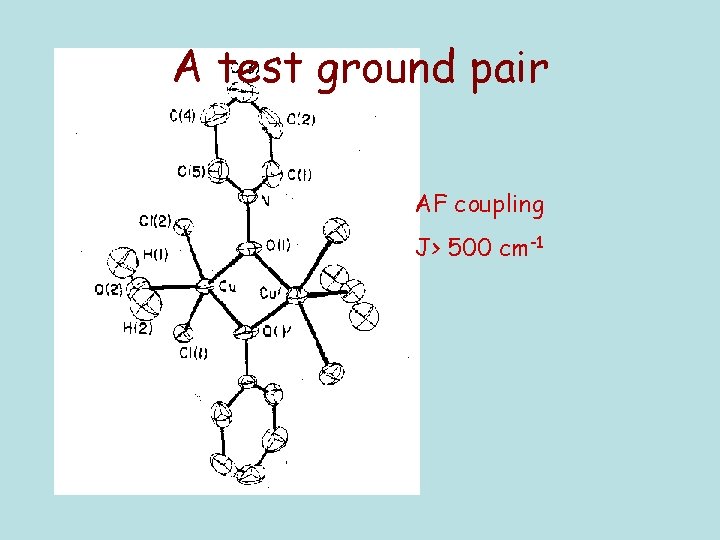
A test ground pair AF coupling J> 500 cm-1

Single Xtal spectra of Mn(II) doped

Spin Hamiltonian Parameters
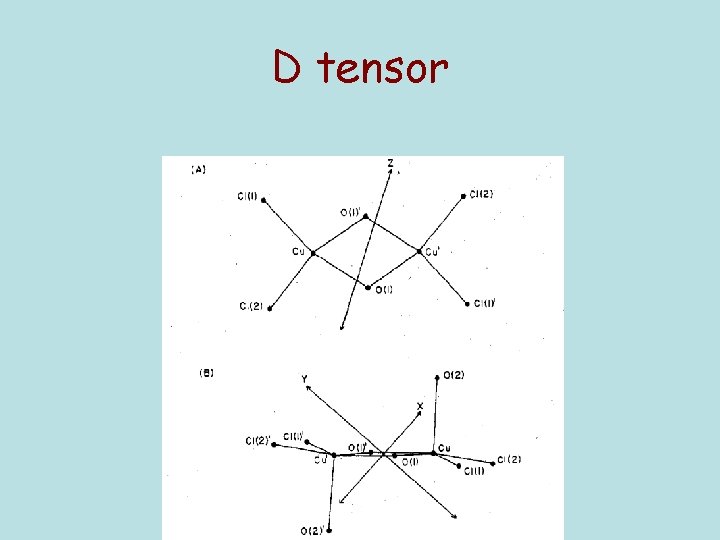
D tensor

g Tensor

Mixed Valence Manganese Dimers Manganese(III), d 4, S=2 Manganese(IV), d 3, S= 3/2 Antiferromagnetic coupling, S= 1/2
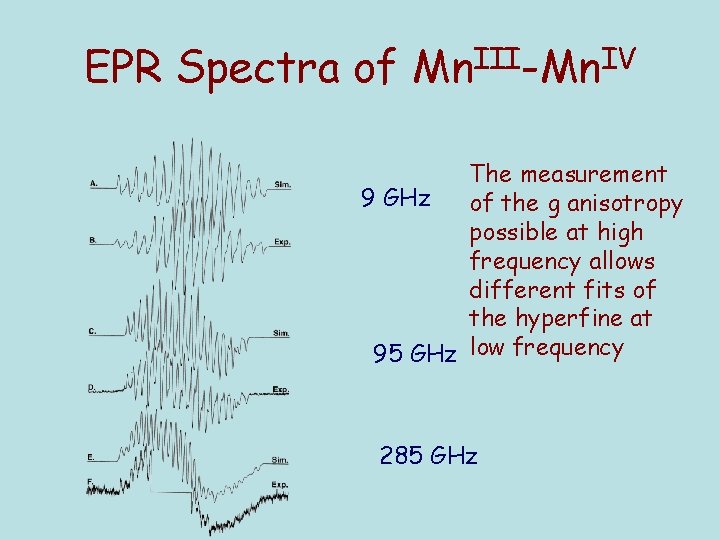
EPR Spectra of Mn. III-Mn. IV The measurement 9 GHz of the g anisotropy possible at high frequency allows different fits of the hyperfine at 95 GHz low frequency 285 GHz
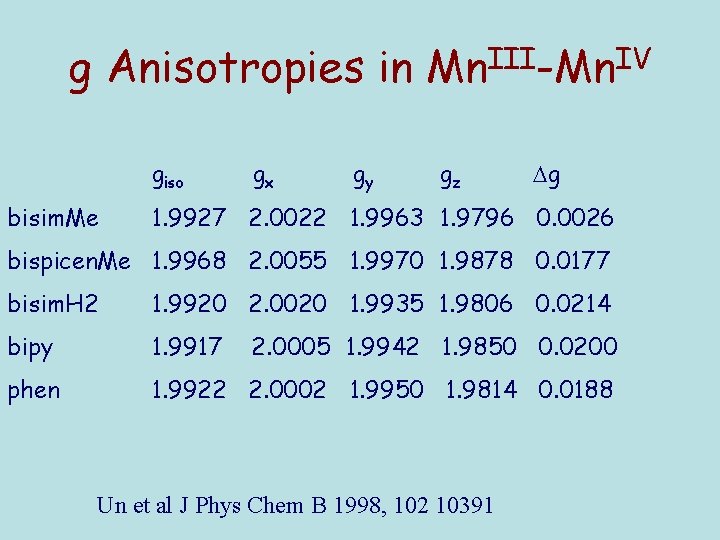
g Anisotropies in Mn. III-Mn. IV giso bisim. Me gx gy gz g 1. 9927 2. 0022 1. 9963 1. 9796 0. 0026 bispicen. Me 1. 9968 2. 0055 1. 9970 1. 9878 0. 0177 bisim. H 2 1. 9920 2. 0020 1. 9935 1. 9806 0. 0214 bipy 1. 9917 phen 1. 9922 2. 0002 1. 9950 1. 9814 0. 0188 2. 0005 1. 9942 1. 9850 0. 0200 Un et al J Phys Chem B 1998, 102 10391

Coefficients for Clusters In the assumption of dominant isotropic exchange the coefficients for the spin hamiiltonian in an S multiplet can be obtained using recurrence formulae The coefficients depend on the intermediate spins

A trinuclear cluster c 1(S 1 S 2 S 12 S 3 S)=c 1(S 12 S 3 S)c 1(S 1 S 2 S 12) c 2(S 1 S 2 S 12 S 3 S)=c 1(S 12 S 3 S)c 2(S 1 S 2 S 12) c 3(S 1 S 2 S 12 S 3 S)=c 2(S 12 S 3 S) d 1(S 1 S 2 S 12 S 3 S)=d 1(S 12 S 3 S)d 1(S 1 S 2 S 12) d 2(S 1 S 2 S 12 S 3 S)=d 1(S 12 S 3 S)d 2(S 1 S 2 S 12) d 3(S 1 S 2 S 12 S 3 S)=d 1(S 12 S 3 S) d 12(S 1 S 2 S 12 S 3 S)=d 1(S 12 S 3 S)d 12(S 1 S 2 S 12) d 13(S 1 S 2 S 12 S 3 S)=d 12(S 12 S 3 S)c 1(S 1 S 2 S 12) d 23(S 1 S 2 S 12 S 3 S)=d 12(S 12 S 3 S)c 2(S 1 S 2 S 12)
![Resonance fields for S states H(M M+1)=(ge/g)[H 0+(2 M+1)/D’/2]; D’=(3 cos 2 -1)D/(ge B) Resonance fields for S states H(M M+1)=(ge/g)[H 0+(2 M+1)/D’/2]; D’=(3 cos 2 -1)D/(ge B)](http://slidetodoc.com/presentation_image/eb15595bec525619c05052b4800bfb46/image-39.jpg)
Resonance fields for S states H(M M+1)=(ge/g)[H 0+(2 M+1)/D’/2]; D’=(3 cos 2 -1)D/(ge B)
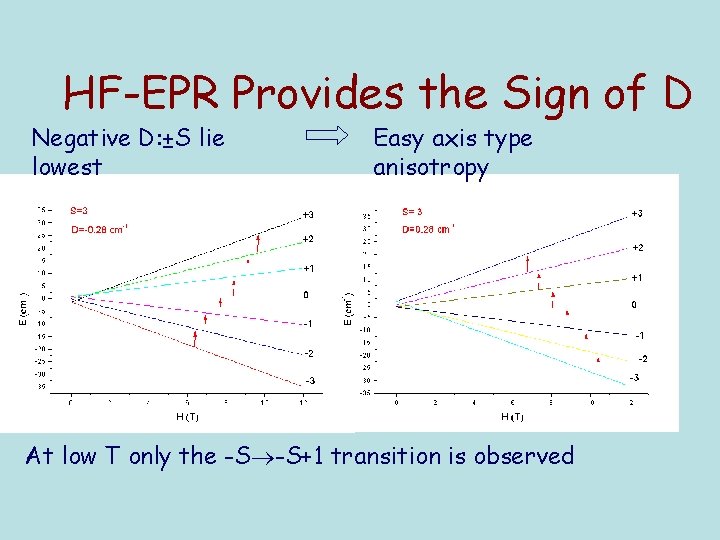
HF-EPR Provides the Sign of D Negative D: ±S lie lowest Easy axis type anisotropy At low T only the -S -S+1 transition is observed
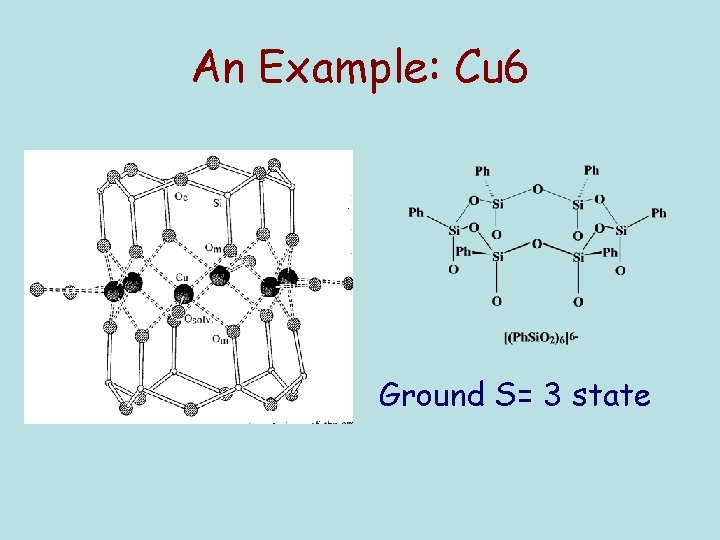
An Example: Cu 6 Ground S= 3 state
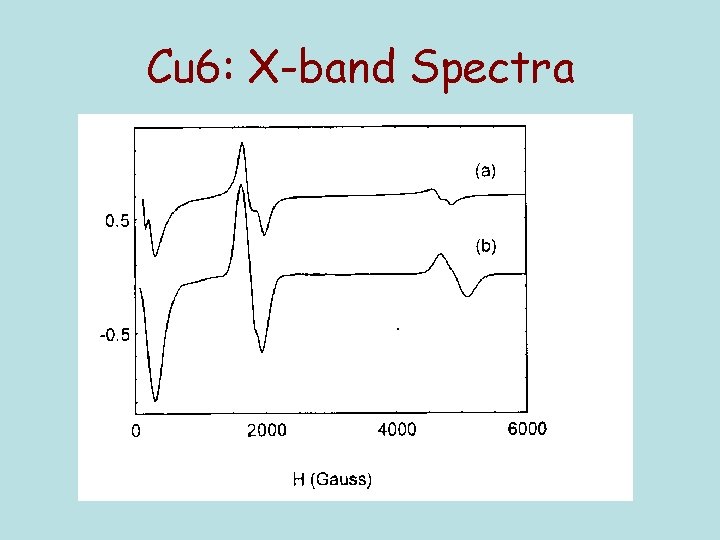
Cu 6: X-band Spectra
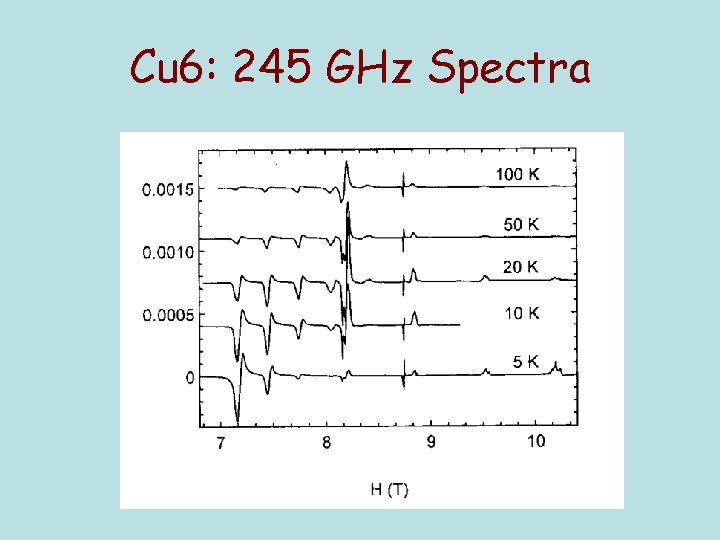
Cu 6: 245 GHz Spectra

Single-Molecule Magnets • In molecular clusters with large spin S and Ising type anisotropy the magnetization relaxes slowly at low temperature • Intermediate behavior between classic and quantum magnets • HF-EPR is unique tool for determining the axial and transverse magnetic anisotropy

The first single molecule magnet: Mn 12 -acetate top view S 4||z Prepared by a comproportionation reaction: T. Lis Acta Cryst. 1980, B 36, 2042. Mn(Ac. O)2 • 4 H 2 O + KMn. O 4 in 60% v/v Ac. OH/H 2 O [Mn 12 O 12(OAc)16(H 2 O)4]· 2 Ac. OH· 4 H 2 O lateral view z MS=-10 Manganese(IV) (s = 3/2, 3 d 3, ) Manganese(III) (s =2, 3 d 4) Oxygen Carbon Ground state S = 8*2 - 4*3/2 = 10 Msaturation = 2. S = 20 B Easy axis of magnetization MS= 10
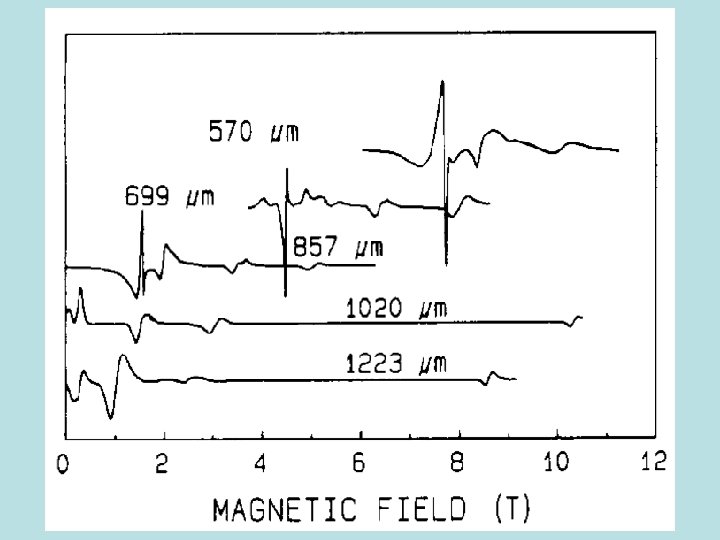

Very High Field EPR Spectra of Mn 12 acetate 525 GHz T= 30 K exp

Which are the conditions for tunneling? • The two wave functions must overlap • A transverse field must couple the two wavefunctions • The coupling splits the two states: tunnel splitting • The larger the tunnel splitting the higher the tunnelling probability
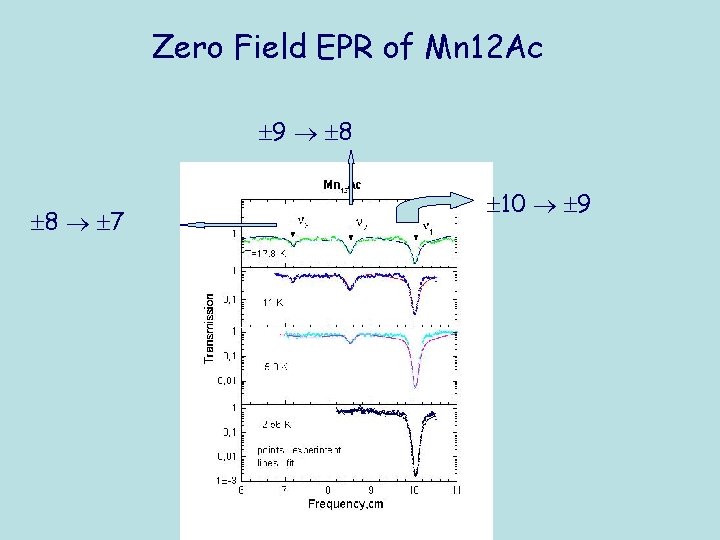
Zero Field EPR of Mn 12 Ac 9 8 8 7 10 9

Local Probes • • Electron spin → EPR Endogenous Nuclear spin → NMR, NQR Muon spin → μSR Exogenous Neutron spin → PND, INS
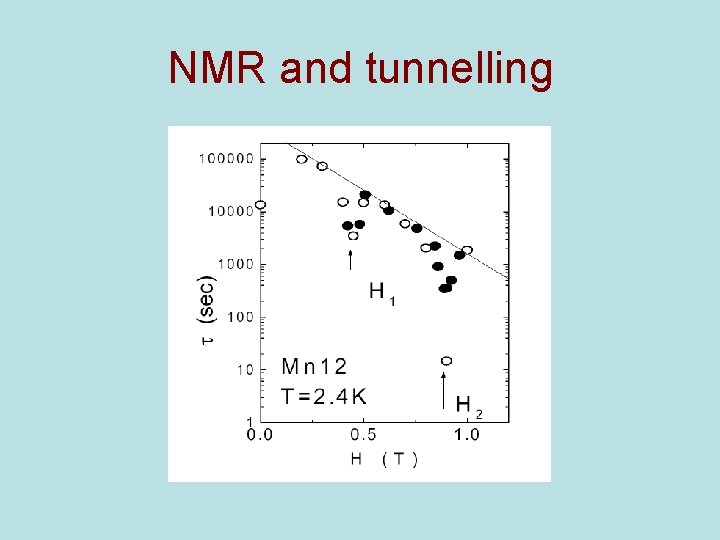
NMR and tunnelling
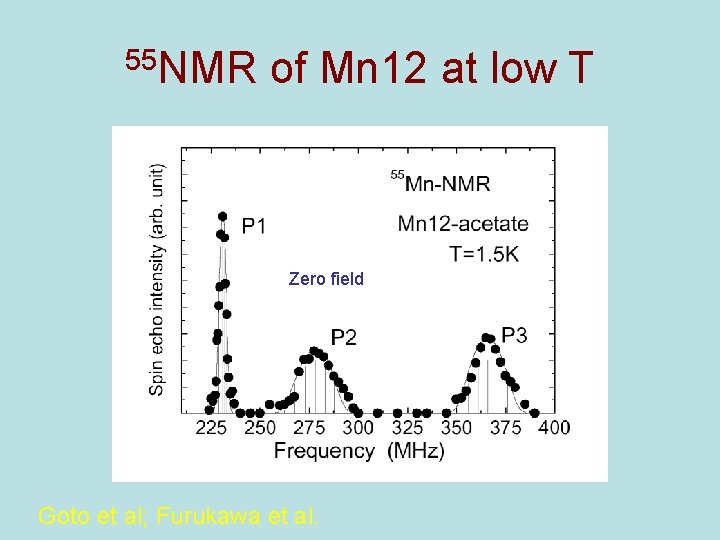
55 NMR of Mn 12 at low T Zero field Goto et al; Furukawa et al.

Parallel field dependence of 55 NMR in Mn 12 1. 5 K
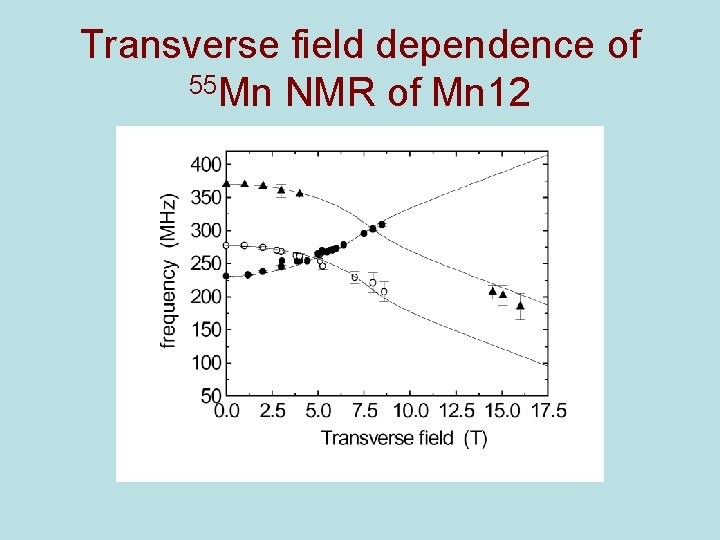
Transverse field dependence of 55 Mn NMR of Mn 12

A tetragonal Mn 12: t. Bu. Ac Wernsdorfer et al. PRL 2006, 96 057208; Hill et al. Polyhedron 2005 24 2284

The strategy EPR Spectra GSH S= 10 MSH

The SH for giant spin
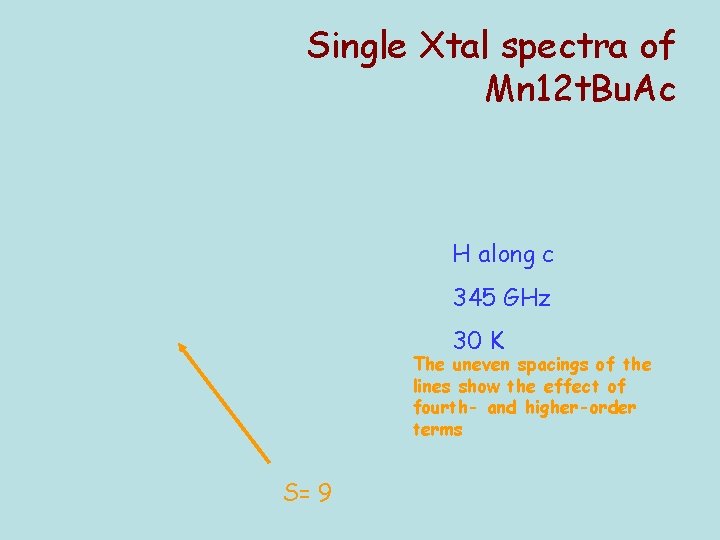
Single Xtal spectra of Mn 12 t. Bu. Ac H along c 345 GHz 30 K The uneven spacings of the lines show the effect of fourth- and higher-order terms S= 9

Single Xtal spectra in the ab plane 115 GHz 5 K The resonance oscillations indicate high order tetragonal terms

Angular dependence of the resonance fields Parameters in cm-1 B 20=-0. 15 B 40=-2. 2 x 10 -5 B 44=+1. 9 x 10 -5 B 60=-1. 0 x 10 -8 B 64=-1. 16 x 10 -7

Physical origin of the parameters • Projection of individual spin high order parameters • Spin admixture • A comparison with a Multi Spin Hamiltonian is needed But the Hilbert space is 100, 000 x 100, 000

A tractable model system 3125 x 3125
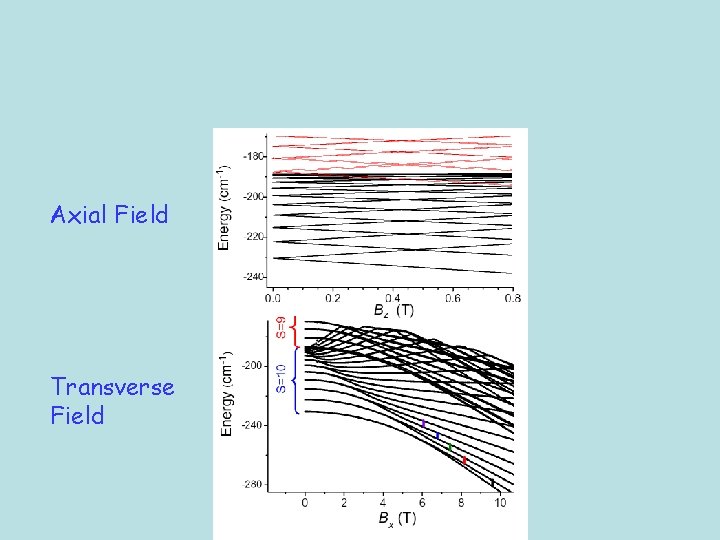
Axial Field Transverse Field

Calculated angular dependence of the transverse resonances -10 -9 -9 -8 -8 -7 -7 -6
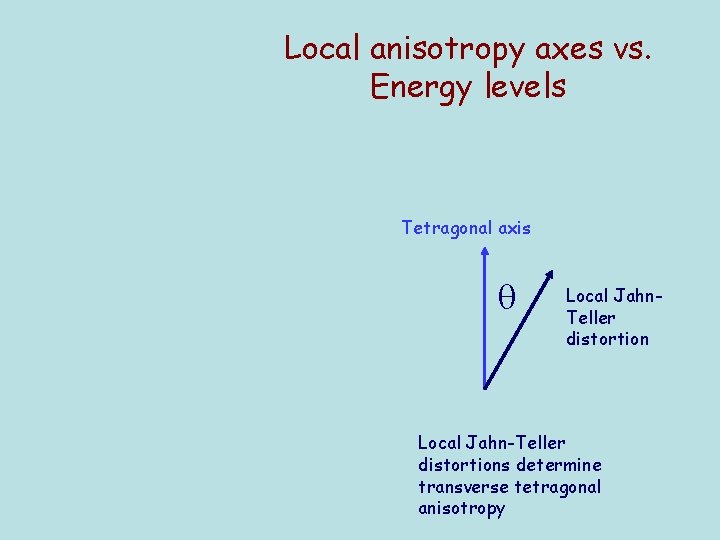
Local anisotropy axes vs. Energy levels Tetragonal axis Local Jahn. Teller distortion Local Jahn-Teller distortions determine transverse tetragonal anisotropy

Angular dependence of the transverse resonances in the molecule
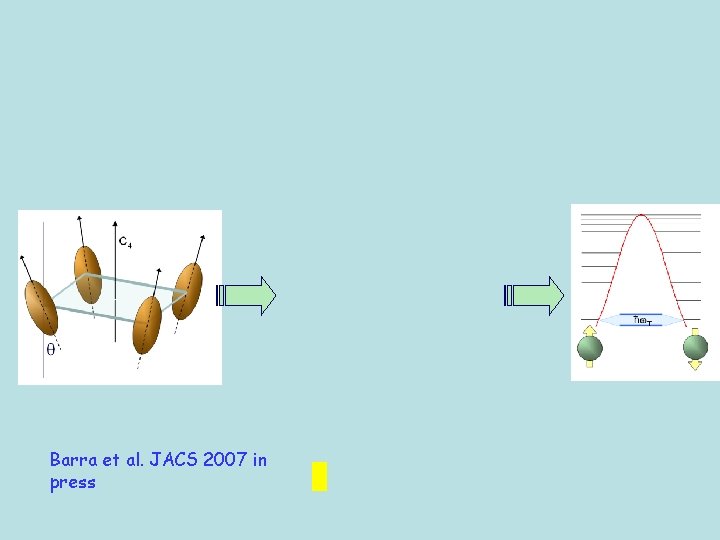
Barra et al. JACS 2007 in press
- Slides: 67