Spectroscopy Structure and Ionization Energy of Be OBe

Spectroscopy, Structure, and Ionization Energy of Be. OBe Jeremy M. Merritt, Vladimir E. Bondybey, and Michael C. Heaven

Background Be. O is a strongly bound, closed-shell molecule. Formally the 2 s electrons of Be are donated to the 2 p obitals of O. Why would Be. O bind a second Be atom? Previous experiments show that Be 2 O is stable. L. Theard and D. Hildenbrand, J. Chem. Phys. 41(11), 8689 (1964) Heat of Formation of Be 2 O(g) by Mass Spectrometry C. Thompson and L. Andrews, J. Chem. Phys. 100(12), 8689 (1994) Recorded IR spectra in an Ar matrix; nas = 1412. 4 cm-1 Theoretical calculations show that the ground state is multi-configurational. A. Boldyrev and J. Simons, J. Phys. Chem. 99, 15041 (1995) Multi-Reference Ab initio study
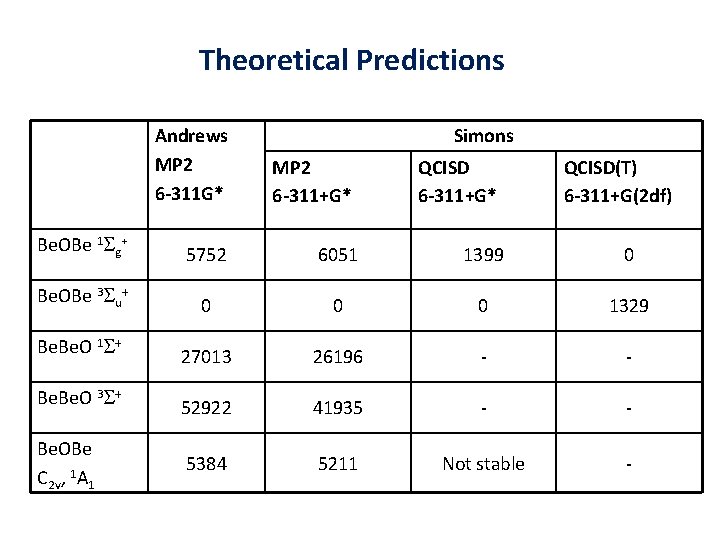
Theoretical Predictions Andrews MP 2 6 -311 G* Simons MP 2 6 -311+G* QCISD(T) 6 -311+G(2 df) Be. OBe 1 Sg+ 5752 6051 1399 0 Be. OBe 3 Su+ 0 0 0 1329 Be. O 1 S+ 27013 26196 - - Be. O 3 S+ 52922 41935 - - 5384 5211 Not stable - Be. OBe C 2 v, 1 A 1
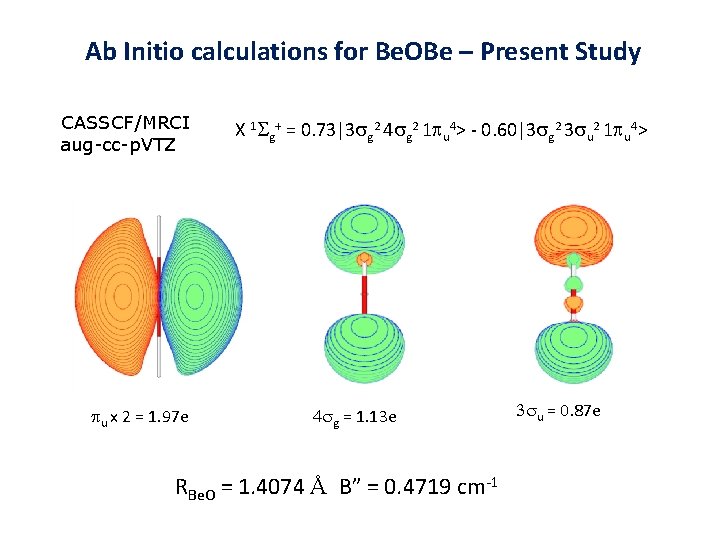
New Multi-Reference Calculations Ab Initio calculations for Be. OBe – Present Study CASSCF/MRCI aug-cc-p. VTZ pu x 2 = 1. 97 e X 1 Sg+ = 0. 73|3 sg 2 4 sg 2 1 pu 4> - 0. 60|3 sg 2 3 su 2 1 pu 4> 4 sg = 1. 13 e RBe. O = 1. 4074 Å B” = 0. 4719 cm-1 3 su = 0. 87 e
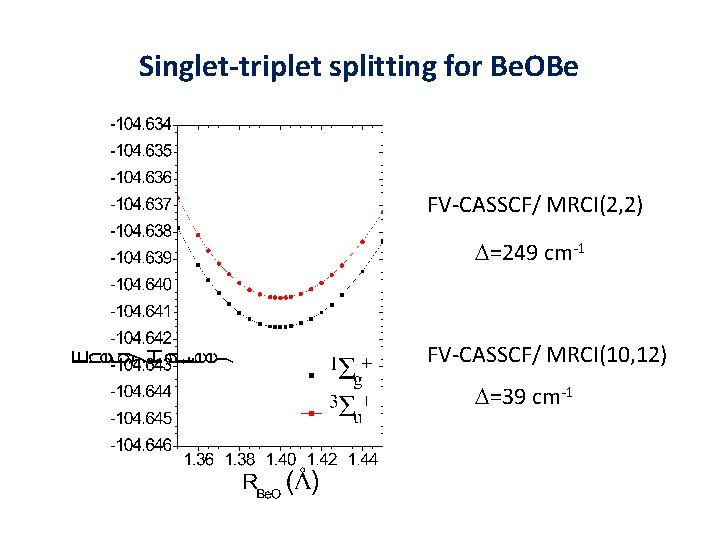
Singlet-triplet splitting for Be. OBe FV-CASSCF/ MRCI(2, 2) D=249 cm-1 FV-CASSCF/ MRCI(10, 12) D=39 cm-1
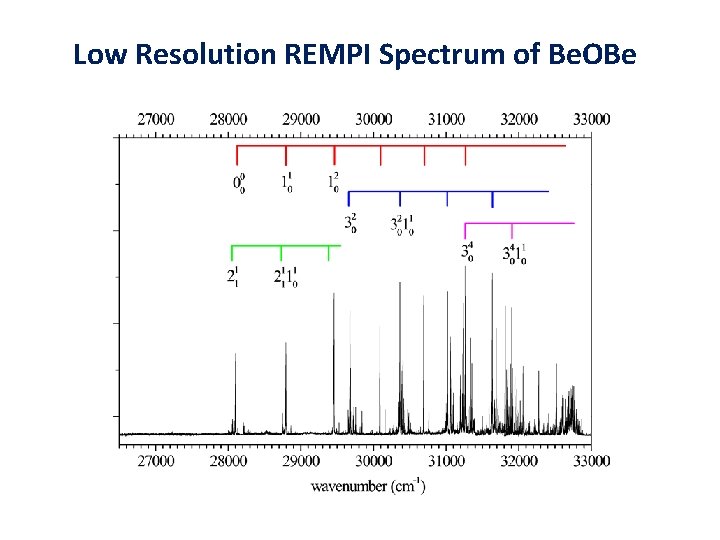
Low Resolution REMPI Spectrum of Be. OBe
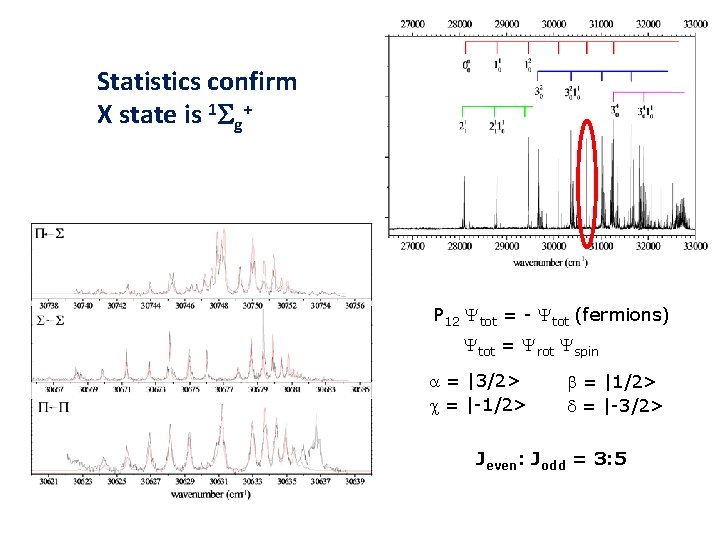
Statistics confirm X state is 1 Sg+ P 12 Ytot = - Ytot (fermions) Ytot = Yrot Yspin a = |3/2> c = |-1/2> b = |1/2> d = |-3/2> Jeven: Jodd = 3: 5
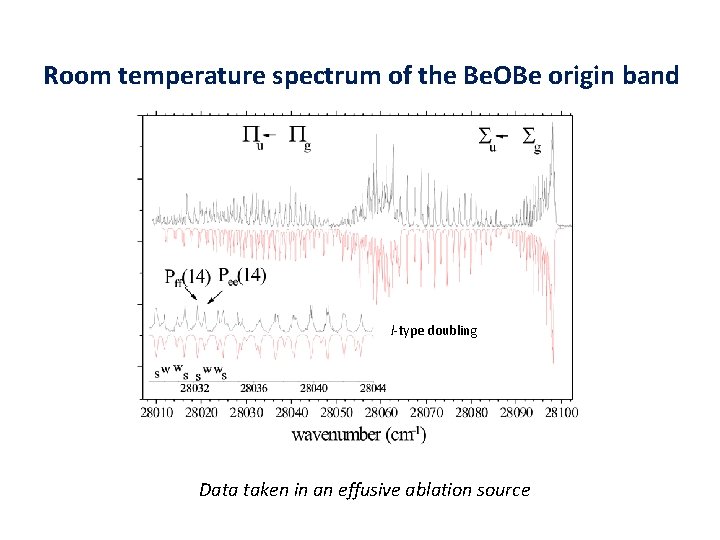
Room temperature spectrum of the Be. OBe origin band l-type doubling Data taken in an effusive ablation source

Trial assignment scheme for band groupings A Pg Su Fails because 1. Combination difference error is too large X Pu Sg 2. Upper state perturbation in the P-S band is not seen in the P-P band.
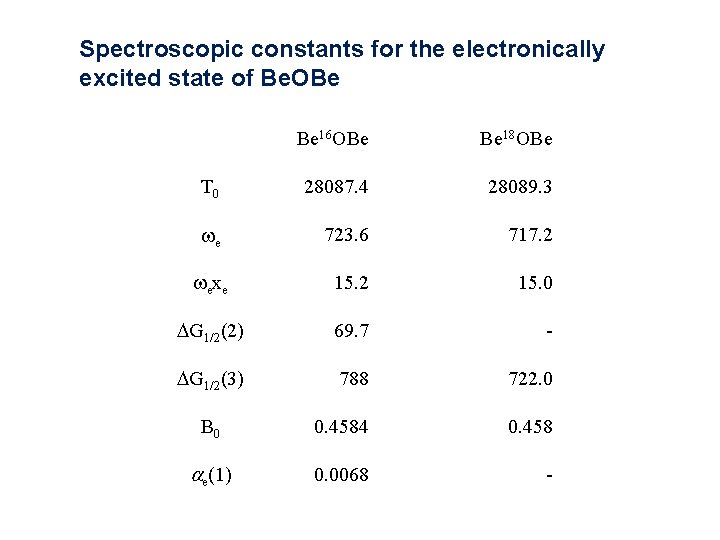
Spectroscopic constants for the electronically excited state of Be. OBe Be 16 OBe Be 18 OBe T 0 28087. 4 28089. 3 we 723. 6 717. 2 wexe 15. 2 15. 0 DG 1/2(2) 69. 7 - DG 1/2(3) 788 722. 0 B 0 0. 4584 0. 458 ae(1) 0. 0068 -

Excited state energies for Be. OBe
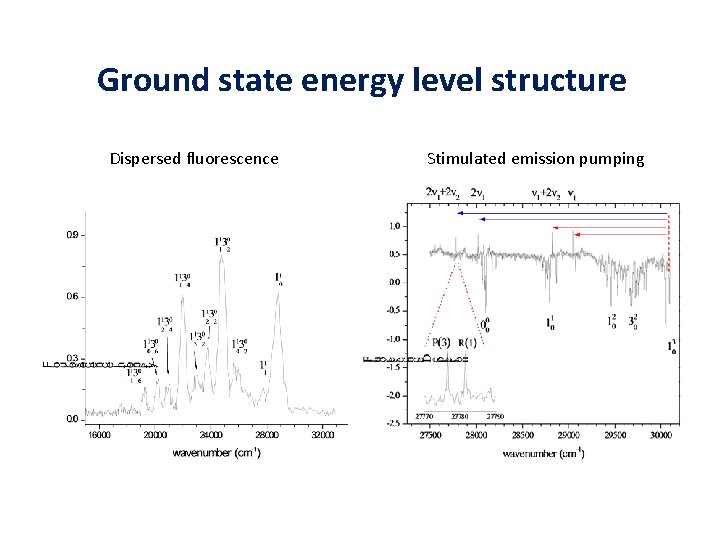
Ground state energy level structure Dispersed fluorescence Stimulated emission pumping
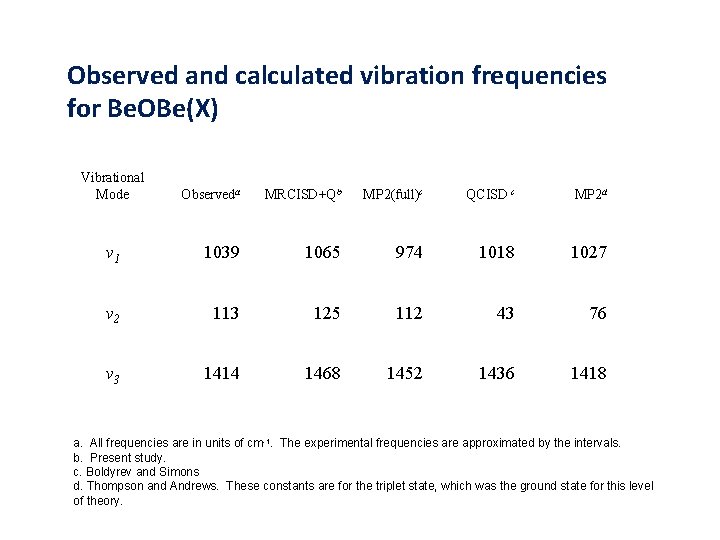
Observed and calculated vibration frequencies for Be. OBe(X) Vibrational Mode Observeda MRCISD+Qb MP 2(full)c QCISD c MP 2 d v 1 1039 1065 974 1018 1027 v 2 113 125 112 43 76 v 3 1414 1468 1452 1436 1418 a. All frequencies are in units of cm-1. The experimental frequencies are approximated by the intervals. b. Present study. c. Boldyrev and Simons d. Thompson and Andrews. These constants are for the triplet state, which was the ground state for this level of theory.
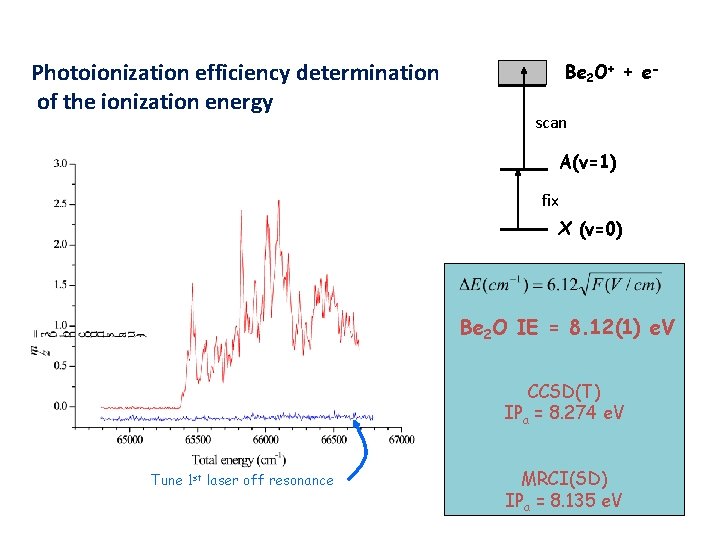
Determining the IE of Be 2 O; Photo-ionization Efficiency (PIE) Curves Photoionization efficiency determination of the ionization energy Be 2 O+ + escan A(v=1) fix X (v=0) Be 2 O IE = 8. 12(1) e. V CCSD(T) IPa = 8. 274 e. V Tune 1 st laser off resonance MRCI(SD) IPa = 8. 135 e. V

Conclusions Spectra for Be. OBe establish a linear X 1 Sg+ ground state. Ab initio calculations indicate a strongly multi-reference ground state. Electronically excited state is A 1 Su+. Vibronic structure indicates that levels from a second excited state are also present in the A-X system. Ionization energy is 8. 12 e. V. MRCI calculations are in good agreement.
- Slides: 15