Spectroscopy of Hybrid InorganicOrganic Interfaces Vibrational Spectroscopy Dietrich

Spectroscopy of Hybrid Inorganic/Organic Interfaces Vibrational Spectroscopy Dietrich RT Zahn
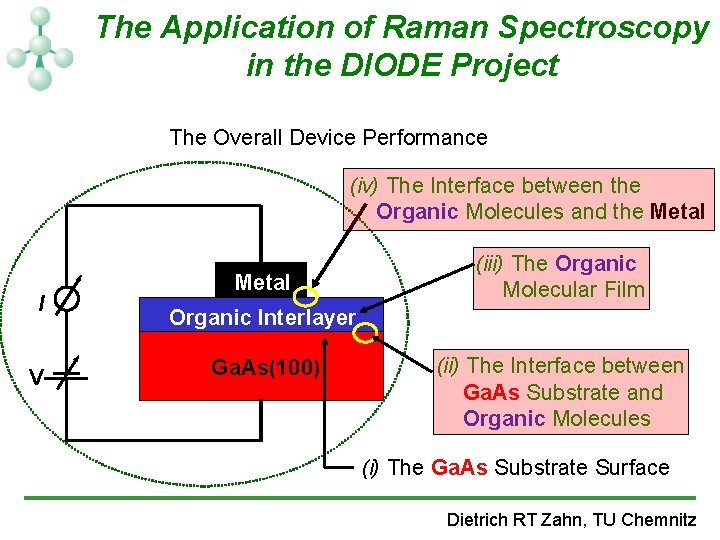
The Application of Raman Spectroscopy in the DIODE Project The Overall Device Performance (iv) The Interface between the Organic Molecules and the Metal I V Metal (iii) The Organic Molecular Film Organic Interlayer Ga. As(100) (ii) The Interface between Ga. As Substrate and Organic Molecules (i) The Ga. As Substrate Surface Dietrich RT Zahn, TU Chemnitz
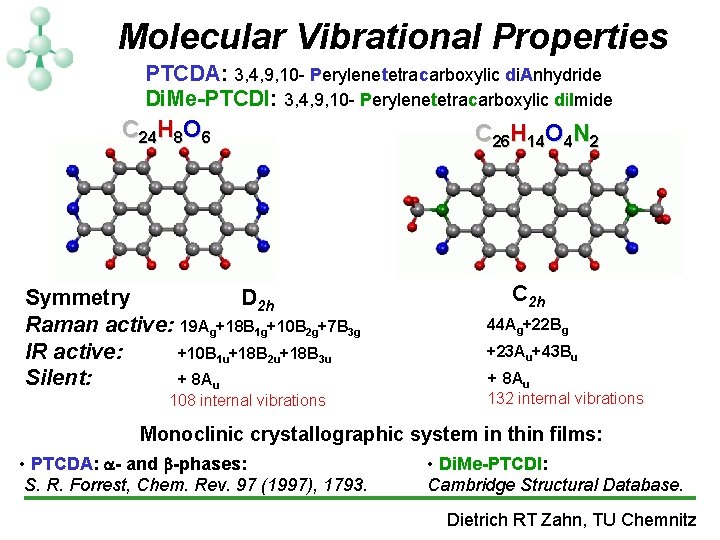
Molecular Vibrational Properties PTCDA: 3, 4, 9, 10 - Perylenetetracarboxylic di. Anhydride Di. Me-PTCDI: 3, 4, 9, 10 - Perylenetetracarboxylic di. Imide C 24 H 8 O 6 Symmetry D 2 h Raman active: 19 Ag+18 B 1 g+10 B 2 g+7 B 3 g IR active: +10 B 1 u+18 B 2 u+18 B 3 u Silent: + 8 Au 108 internal vibrations C 26 H 14 O 4 N 2 C 2 h 44 Ag+22 Bg +23 Au+43 Bu + 8 Au 132 internal vibrations Monoclinic crystallographic system in thin films: • PTCDA: - and -phases: S. R. Forrest, Chem. Rev. 97 (1997), 1793. • Di. Me-PTCDI: Cambridge Structural Database. Dietrich RT Zahn, TU Chemnitz
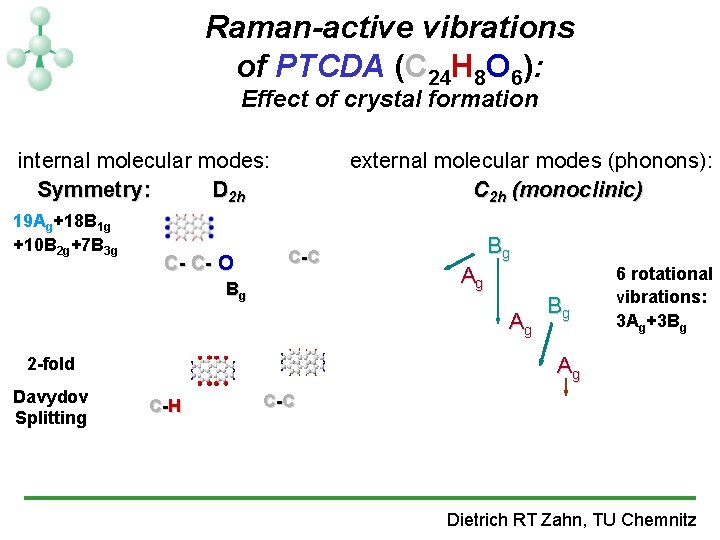
Raman-active vibrations of PTCDA (C 24 H 8 O 6): Effect of crystal formation internal molecular modes: Symmetry: D 2 h 19 Ag+18 B 1 g +10 B 2 g+7 B 3 g C- C- O external molecular modes (phonons): C 2 h (monoclinic) C -C Bg Bg Ag Ag Ag 2 -fold Davydov Splitting Bg 6 rotational vibrations: 3 Ag+3 Bg C -H C -C Dietrich RT Zahn, TU Chemnitz
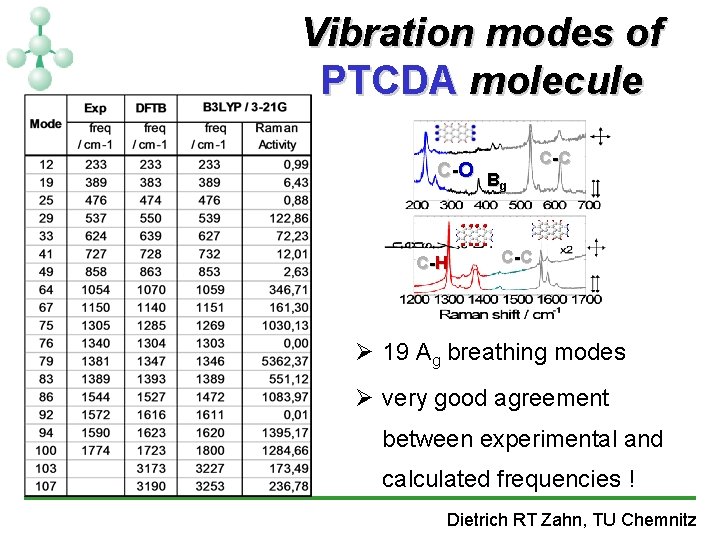
Vibration modes of PTCDA molecule C -O B g C -H C -C Ø 19 Ag breathing modes Ø very good agreement between experimental and calculated frequencies ! Dietrich RT Zahn, TU Chemnitz
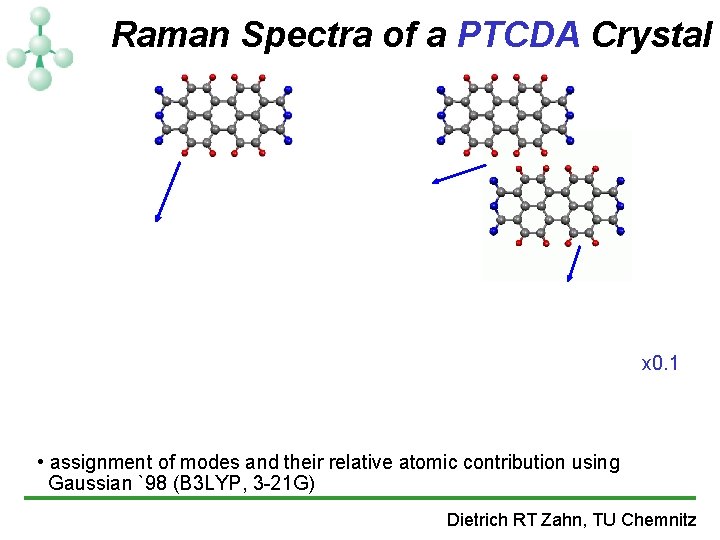
Raman Spectra of a PTCDA Crystal x 0. 1 • assignment of modes and their relative atomic contribution using Gaussian `98 (B 3 LYP, 3 -21 G) Dietrich RT Zahn, TU Chemnitz
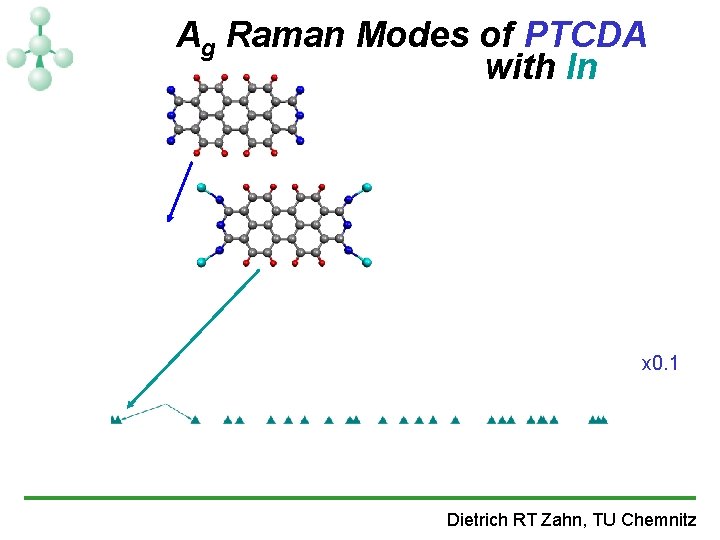
Ag Raman Modes of PTCDA with In x 0. 1 Dietrich RT Zahn, TU Chemnitz
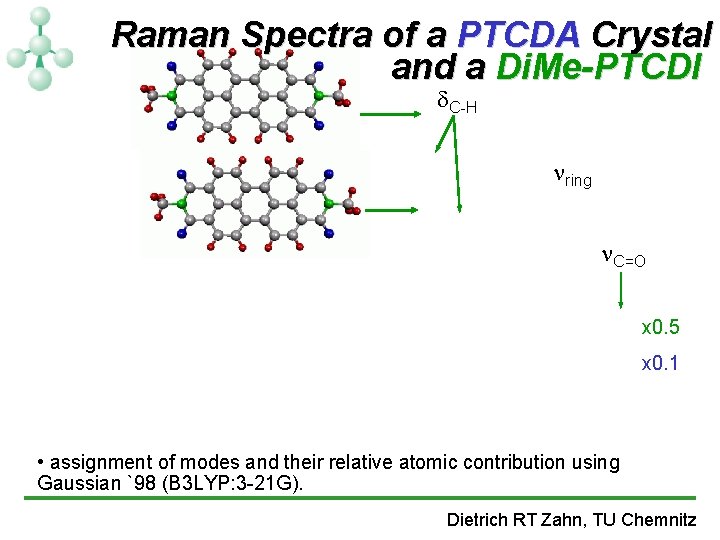
Raman Spectra of a PTCDA Crystal and a Di. Me-PTCDI C-H ring C=O x 0. 5 x 0. 1 • assignment of modes and their relative atomic contribution using Gaussian `98 (B 3 LYP: 3 -21 G). Dietrich RT Zahn, TU Chemnitz
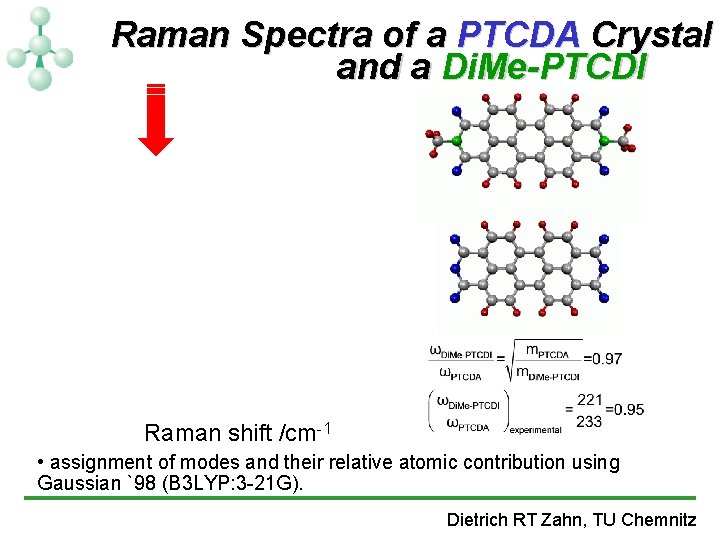
Raman Spectra of a PTCDA Crystal and a Di. Me-PTCDI Raman shift /cm-1 • assignment of modes and their relative atomic contribution using Gaussian `98 (B 3 LYP: 3 -21 G). Dietrich RT Zahn, TU Chemnitz
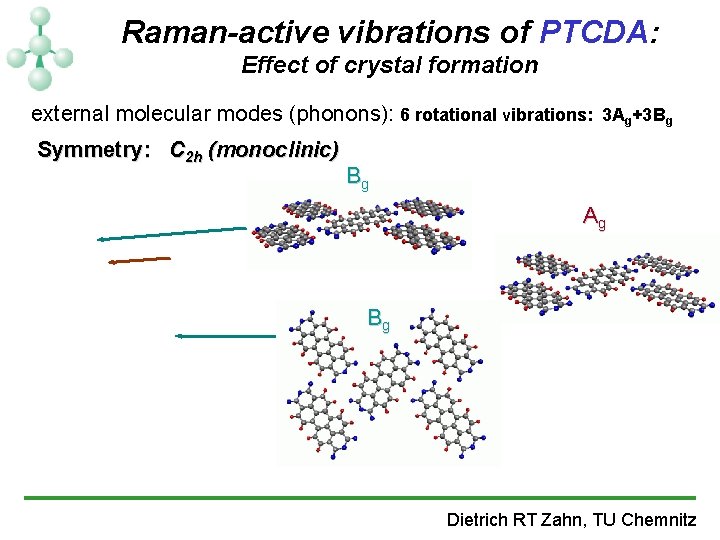
Raman-active vibrations of PTCDA: Effect of crystal formation external molecular modes (phonons): 6 rotational vibrations: 3 Ag+3 Bg Symmetry: C 2 h (monoclinic) Bg Ag Bg Dietrich RT Zahn, TU Chemnitz

Infrared Modes in Films on S-Ga. As Reflection, s-polarized light. C-O+ C-C C-H (oop) C=O ring C-H+ C-N-C C-O-C • Assignment of modes using Gaussian `98 (B 3 LYP, 3 -21 G). Dietrich RT Zahn, TU Chemnitz
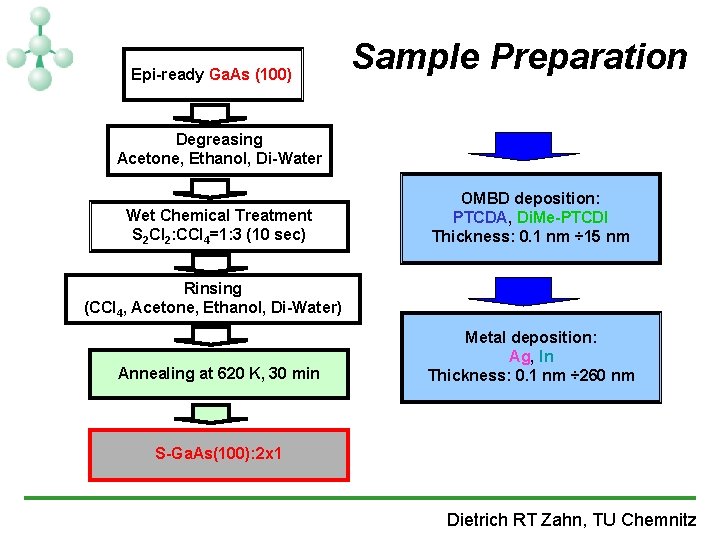
Epi-ready Ga. As (100) Sample Preparation Degreasing Acetone, Ethanol, Di-Water Wet Chemical Treatment S 2 Cl 2: CCl 4=1: 3 (10 sec) OMBD deposition: PTCDA, Di. Me-PTCDI Thickness: 0. 1 nm ÷ 15 nm Rinsing (CCl 4, Acetone, Ethanol, Di-Water) Annealing at 620 K, 30 min Metal deposition: Ag, In Thickness: 0. 1 nm ÷ 260 nm S-Ga. As(100): 2 x 1 Dietrich RT Zahn, TU Chemnitz

Ex Situ (Micro-) and In Situ (Macro- Configuration) Raman Spectroscopy Ar+ line Dilor XY 800 Spectrometer Monochromatic light source: Ar+ Laser (2. 54 e. V), Detector: CCD • resonance condition with the absorption band of the organic crystalline material. • resolution: 1. 2 cm-1 to 3. 5 cm-1. Dietrich RT Zahn, TU Chemnitz
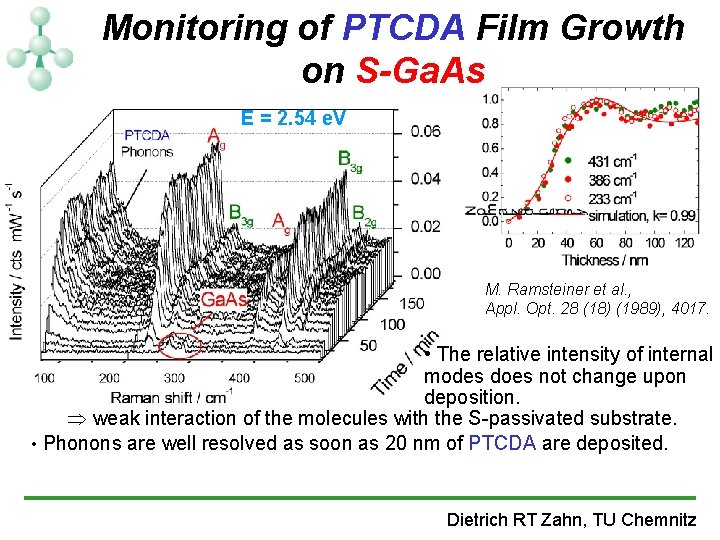
Monitoring of PTCDA Film Growth on S-Ga. As E = 2. 54 e. V M. Ramsteiner et al. , Appl. Opt. 28 (18) (1989), 4017. • The relative intensity of internal modes does not change upon deposition. Þ weak interaction of the molecules with the S-passivated substrate. • Phonons are well resolved as soon as 20 nm of PTCDA are deposited. Dietrich RT Zahn, TU Chemnitz

Chemistry at Organic/S-Ga. As(100): 2 x 1 Vibrational Properties: PTCDA Annealing at 623 K for 30 40 nm x 0. 01 0. 45 nm (x 0. 6) 0. 18 nm ann. x 4. 4 min: • Molecules remaining at the surface: NPTCDA(0. 04 nm)~1013 cm-2 Nd. Si ~ 1012 cm-2 • Spectrum of annealed film similar to that of an annealed PTCDA film on Si(100). Þ The strongest interaction: between the PTCDA molecules and defects due to Si at the Ga. As surface. Dietrich RT Zahn, TU Chemnitz
Calculated Vibrational Properties: PTCDA Dietrich RT Zahn, TU Chemnitz

Calculated Vibrational Properties: PTCDA Molecular charging with one elementary charge: • significant spectral changes predicted for the C=C modes around 1600 cm-1 Þ fractional charge transfer between the PTCDA and the defects at the Ga. As surface. Dietrich RT Zahn, TU Chemnitz
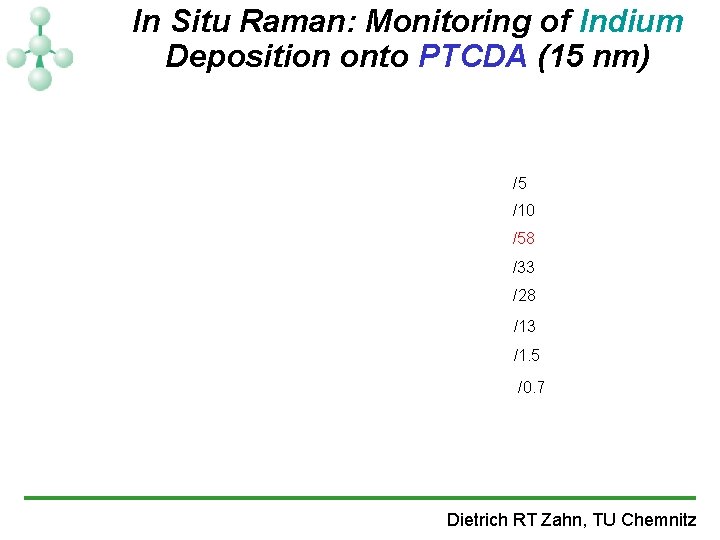
In Situ Raman: Monitoring of Indium Deposition onto PTCDA (15 nm) /5 /10 /58 /33 /28 /13 /1. 5 /0. 7 Dietrich RT Zahn, TU Chemnitz
Influence of Indium on Vibrational Spectra of PTCDA Dietrich RT Zahn, TU Chemnitz
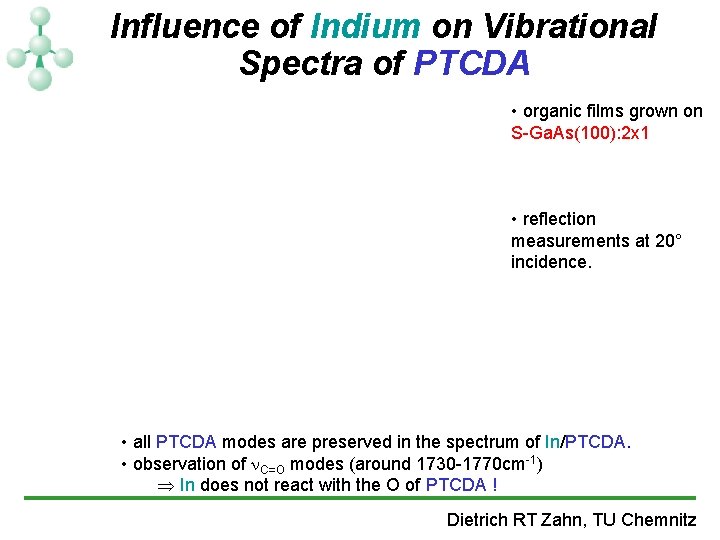
Influence of Indium on Vibrational Spectra of PTCDA • organic films grown on S-Ga. As(100): 2 x 1 • reflection measurements at 20° incidence. • all PTCDA modes are preserved in the spectrum of In/PTCDA. • observation of C=O modes (around 1730 -1770 cm-1) Þ In does not react with the O of PTCDA ! Dietrich RT Zahn, TU Chemnitz
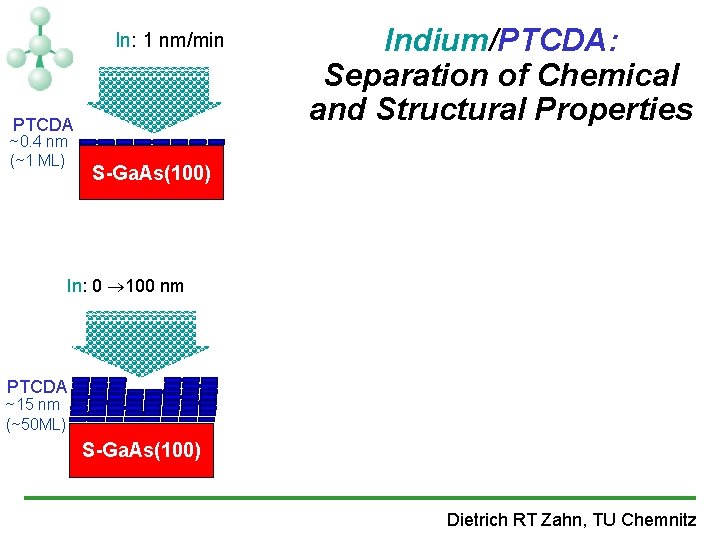
In: 1 nm/min PTCDA ~0. 4 nm (~1 ML) Indium/PTCDA: Separation of Chemical and Structural Properties S-Ga. As(100) In: 0 100 nm PTCDA ~15 nm (~50 ML) S-Ga. As(100) Dietrich RT Zahn, TU Chemnitz
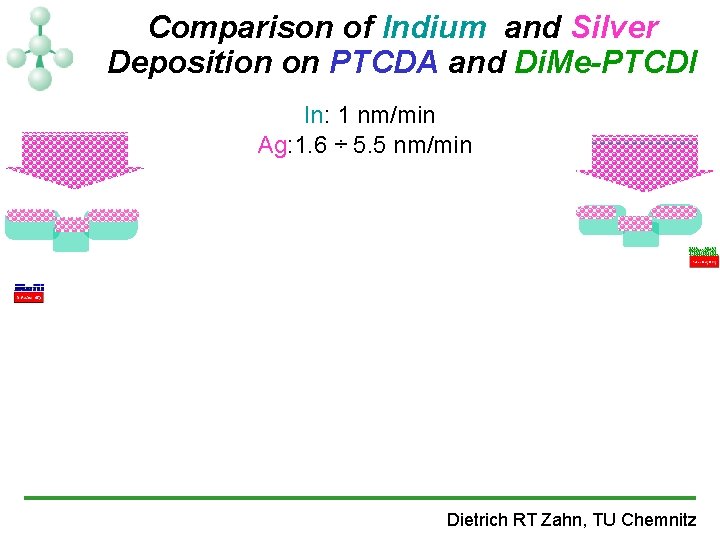
Comparison of Indium and Silver Deposition on PTCDA and Di. Me-PTCDI In: 1 nm/min Ag: 1. 6 ÷ 5. 5 nm/min Dietrich RT Zahn, TU Chemnitz

Comparison of Indium and Silver Deposition on PTCDA and Di. Me-PTCDI • the PTCDA external modes: Ø are preserved broadened after 0. 3 nm Ag deposition. Ø disappear after 0. 4 nm In. • the Di. Me-PTCDI external modes: Ø less affected compared to PTCDA. Ø probably due to less compact crystalline structure. Dietrich RT Zahn, TU Chemnitz
Mg, In, Ag on PTCDA Dietrich RT Zahn, TU Chemnitz
Mg, In, Ag on Di. Me-PTCDI +Mg Dietrich RT Zahn, TU Chemnitz
Indium and Silver Deposition: Enhancement Factors PTCDA (15 mn) Di. Me-PTCDI (15 nm) Dietrich RT Zahn, TU Chemnitz
![Determination of Molecular Orientation: =0°: x II [011]Ga. As Di. Me-PTCDI =90°: x II Determination of Molecular Orientation: =0°: x II [011]Ga. As Di. Me-PTCDI =90°: x II](http://slidetodoc.com/presentation_image_h/ec74e7f8bb4ddd1a0d6cdc40f4a45084/image-27.jpg)
Determination of Molecular Orientation: =0°: x II [011]Ga. As Di. Me-PTCDI =90°: x II [0 -11] phonons v Azimuthal rotation of a 120 nm thick film; normal incidence. Ø Periodic variation of signal in crossed and parallel polarization. M. Friedrich, G. Salvan, D. Zahn et al. , J. Phys. Cond. Mater. submitted. Dietrich RT Zahn, TU Chemnitz
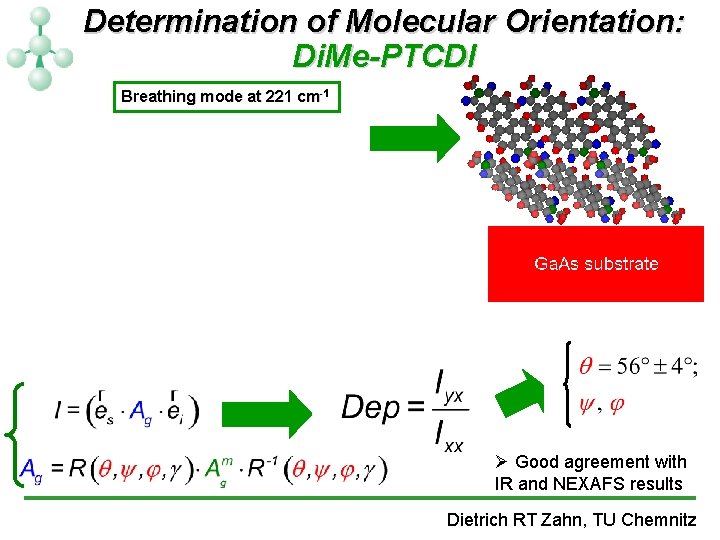
Determination of Molecular Orientation: Di. Me-PTCDI Breathing mode at 221 cm-1 Ø Good agreement with IR and NEXAFS results Dietrich RT Zahn, TU Chemnitz
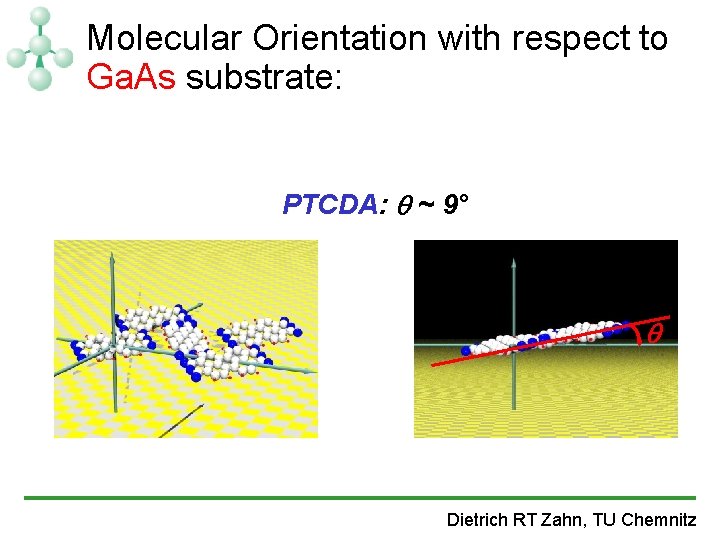
Molecular Orientation with respect to Ga. As substrate: PTCDA: ~ 9° Dietrich RT Zahn, TU Chemnitz
![[-110] Di. Me-PTCDI: ~ 6° ~ 60° Dietrich RT Zahn, TU Chemnitz [-110] Di. Me-PTCDI: ~ 6° ~ 60° Dietrich RT Zahn, TU Chemnitz](http://slidetodoc.com/presentation_image_h/ec74e7f8bb4ddd1a0d6cdc40f4a45084/image-30.jpg)
[-110] Di. Me-PTCDI: ~ 6° ~ 60° Dietrich RT Zahn, TU Chemnitz
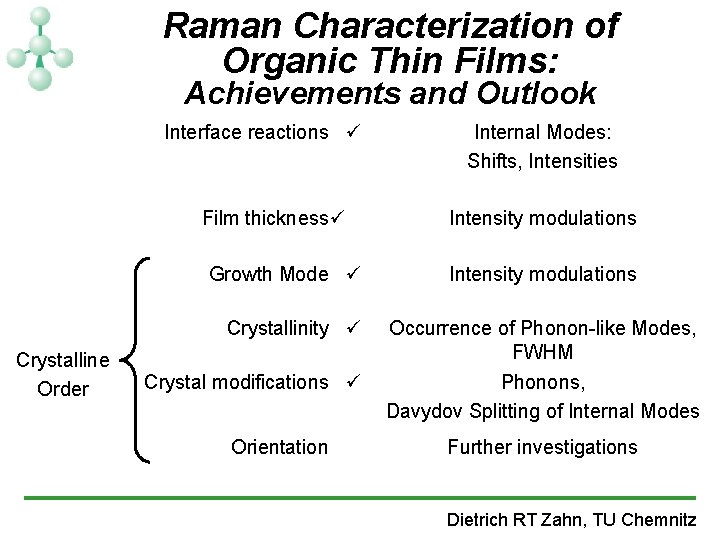
Raman Characterization of Organic Thin Films: Achievements and Outlook Interface reactions ü Film thickness ü Growth Mode ü Crystallinity ü Crystalline Order Crystal modifications ü Orientation Internal Modes: Shifts, Intensities Intensity modulations Occurrence of Phonon-like Modes, FWHM Phonons, Davydov Splitting of Internal Modes Further investigations Dietrich RT Zahn, TU Chemnitz

Raman Spectroscopy Team: Dietrich RT Zahn, TU Chemnitz
- Slides: 32