Specimen collection Clean tube Na FKoxalate tube SST
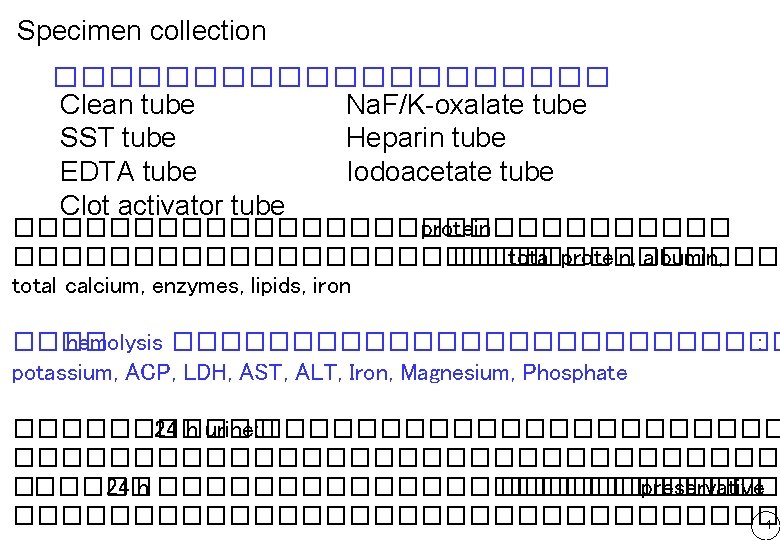
Specimen collection ���������� Clean tube Na. F/K-oxalate tube SST tube Heparin tube EDTA tube Iodoacetate tube Clot activator tube ��������������� protein ����������������� total protein, albumin, total calcium, enzymes, lipids, iron ���� hemolysis ������������� : potassium, ACP, LDH, AST, ALT, Iron, Magnesium, Phosphate ������ 24 h urine: ���������������������������� 24 h ������������� preservative ����������������� 1
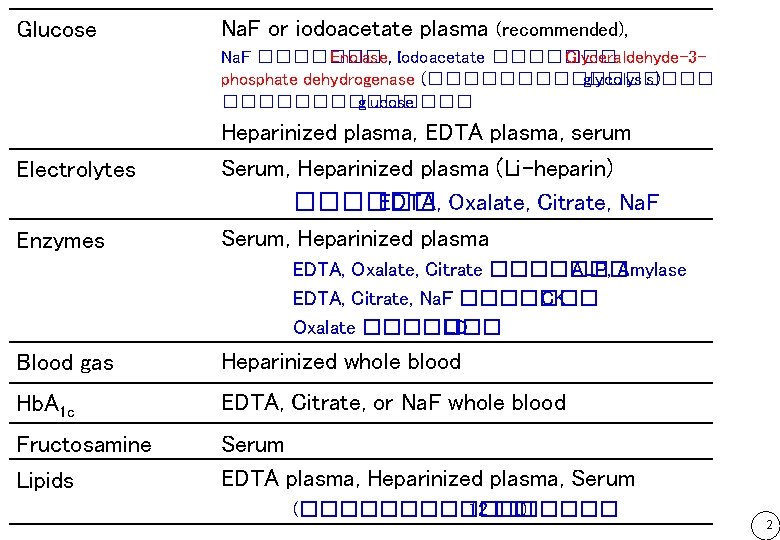
Glucose Na. F or iodoacetate plasma (recommended), Na. F ������� Enolase, Iodoacetate ������� Glyceraldehyde-3 phosphate dehydrogenase (�������� glycolysis) ������� glucose Heparinized plasma, EDTA plasma, serum Electrolytes Enzymes Serum, Heparinized plasma (Li-heparin) ������ EDTA, Oxalate, Citrate, Na. F Serum, Heparinized plasma EDTA, Oxalate, Citrate ������� ALP, Amylase EDTA, Citrate, Na. F ������� CK Oxalate ������� LD Blood gas Heparinized whole blood Hb. A 1 c EDTA, Citrate, or Na. F whole blood Fructosamine Lipids Serum EDTA plasma, Heparinized plasma, Serum (�������� 12 ��. ) 2
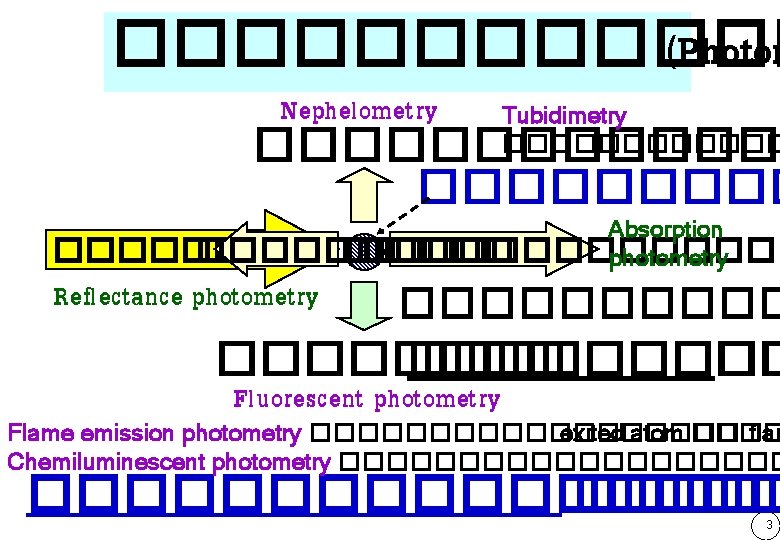
������ (Photom Nephelometry Tubidimetry ������������ Absorption �������������� photometry Reflectance photometry ��������� �� Fluorescent photometry Flame emission photometry ��������� exited atom ���� flam Chemiluminescent photometry ���������� ����� 3
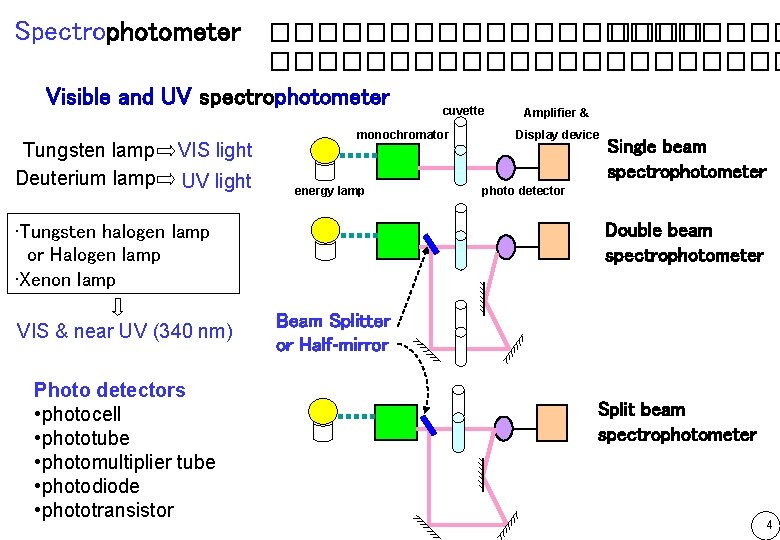
Spectrophotometer ������������� Visible and UV spectrophotometer cuvette Amplifier & Tungsten lamp Deuterium lamp VIS light UV light monochromator energy lamp Photo detectors • photocell • phototube • photomultiplier tube • photodiode • phototransistor Single beam spectrophotometer photo detector Double beam spectrophotometer • Tungsten halogen lamp or Halogen lamp • Xenon lamp VIS & near UV (340 nm) Display device Beam Splitter or Half-mirror Split beam spectrophotometer 4
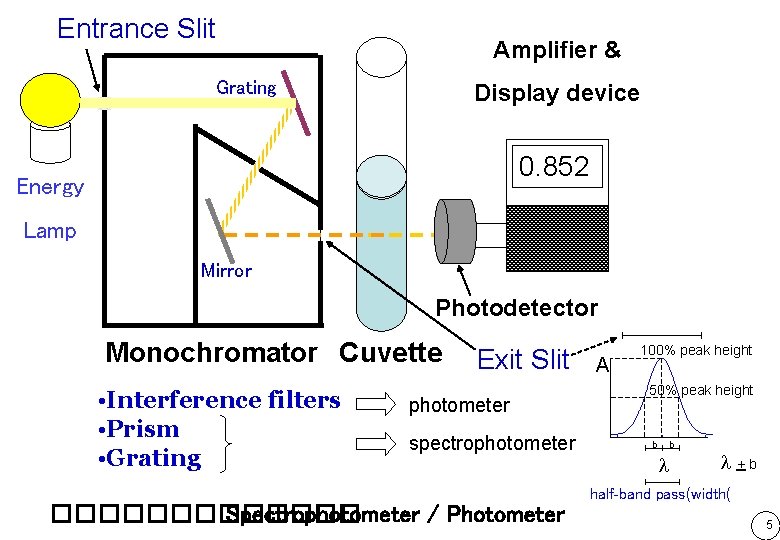
Entrance Slit Amplifier & Grating Display device 0. 852 Energy Lamp Mirror Photodetector Monochromator Cuvette • Interference filters • Prism • Grating Exit Slit photometer spectrophotometer A 100% peak height 50% peak height b b +b half-band pass(width( ������� Spectrophotometer / Photometer 5
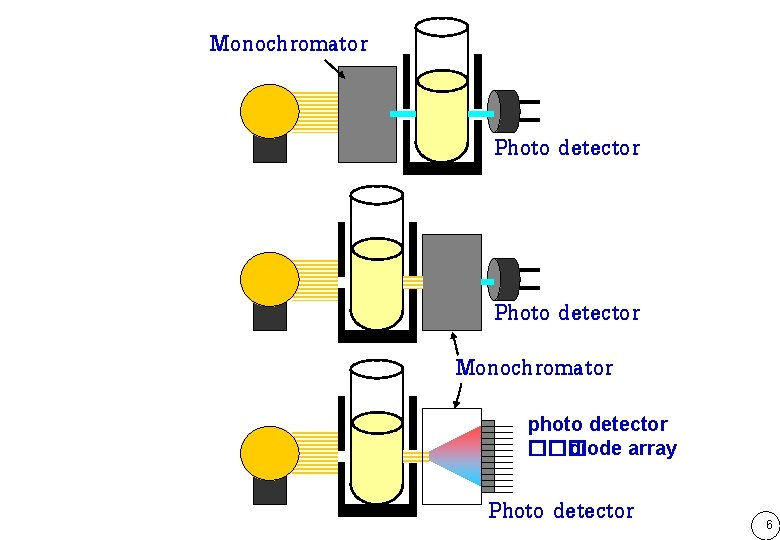
Monochromator Photo detector Monochromator photo detector ��� diode array Photo detector 6
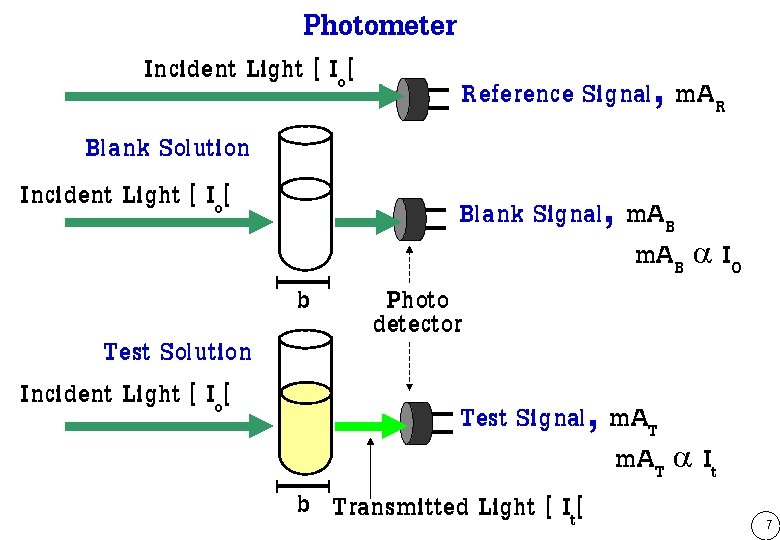
Photometer Incident Light [ Io[ Blank Solution Incident Light [ Io[ Test Solution Incident Light [ Io[ b Reference Signal, m. AR Blank Signal, m. AB a IO Photo detector Test Signal, m. AT a It b Transmitted Light [ It[ 7
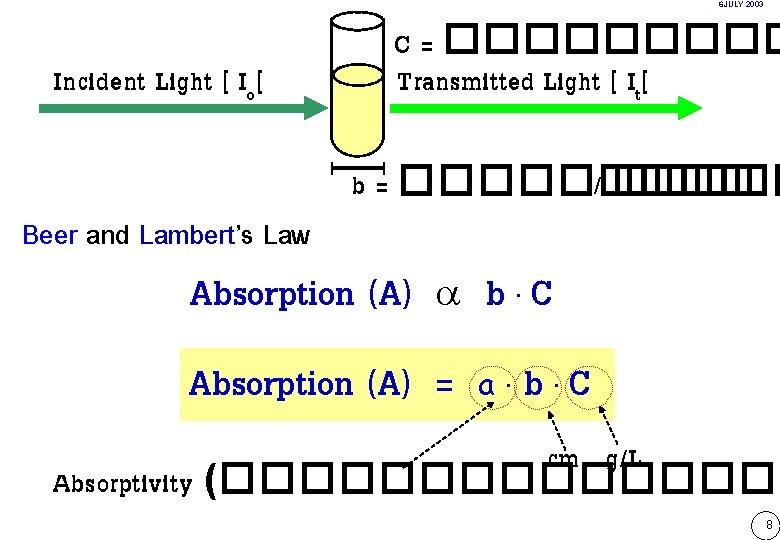
6 JULY 2003 Incident Light [ Io[ Beer and Lambert’s Law C = ����� Transmitted Light [ It[ b = ����� / ����� Absorption (A) a Absorption (A) = b. C a. b. C cm g/L Absorptivity (�������� ( 8
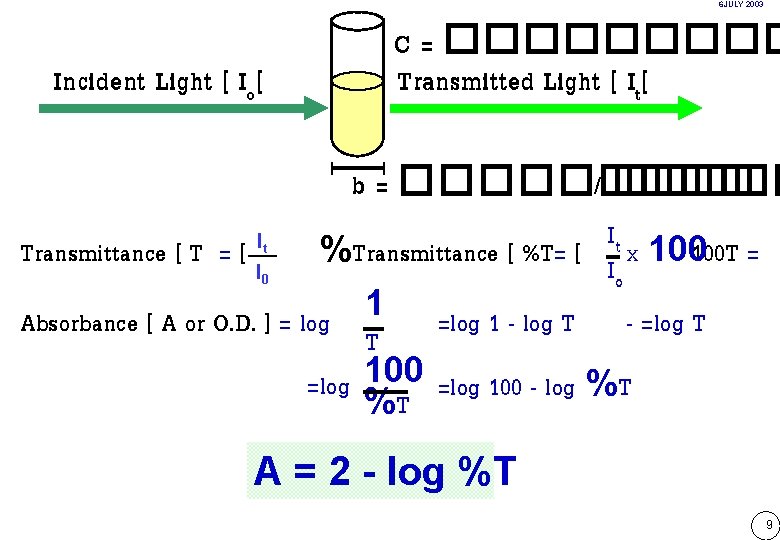
6 JULY 2003 Incident Light [ Io[ C = ����� Transmitted Light [ It[ b = ����� / ����� It Transmittance [ T = [ I %Transmittance [ %T= [ I x 100100 T = o 1 Absorbance [ A or O. D. ] = log =log 1 - log T - =log T T =log 100 - log %T %T It 0 A = 2 - log %T 9
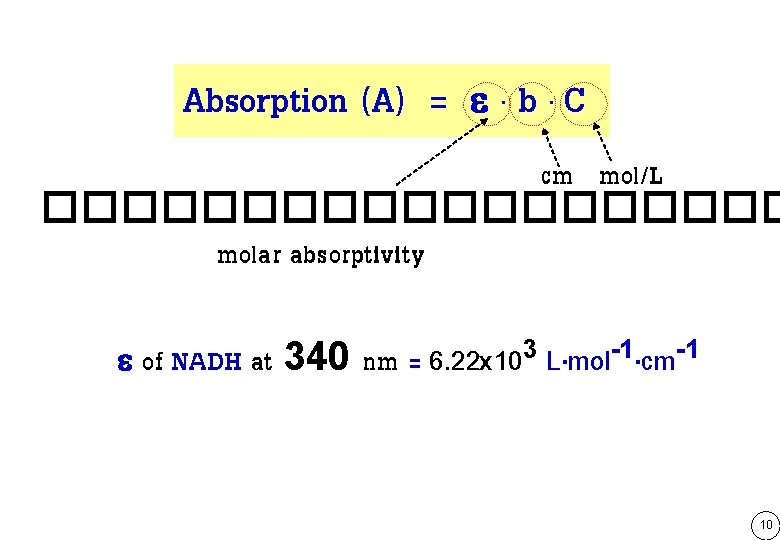
Absorption (A) = e. b. C cm mol/L ���������� molar absorptivity e of NADH at 340 nm = 6. 22 x 103 L. mol-1. cm-1 10
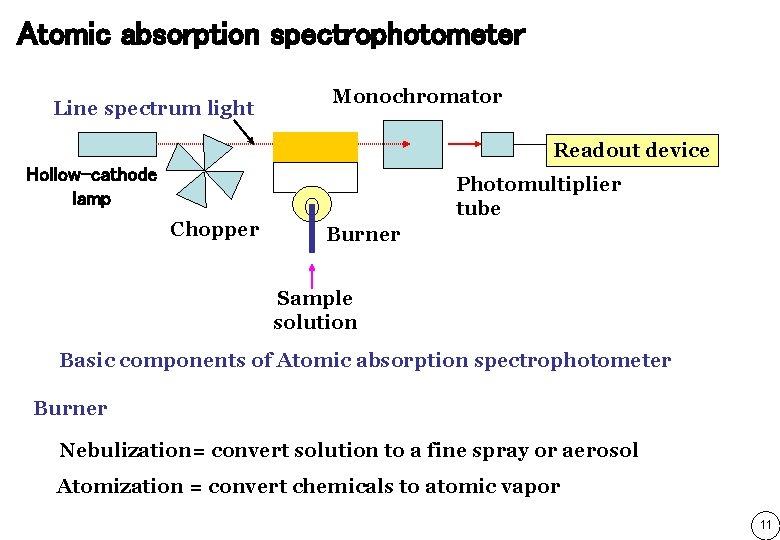
Atomic absorption spectrophotometer Line spectrum light Monochromator Readout device Hollow-cathode lamp Chopper Photomultiplier tube Burner Sample solution Basic components of Atomic absorption spectrophotometer Burner Nebulization= convert solution to a fine spray or aerosol Atomization = convert chemicals to atomic vapor 11
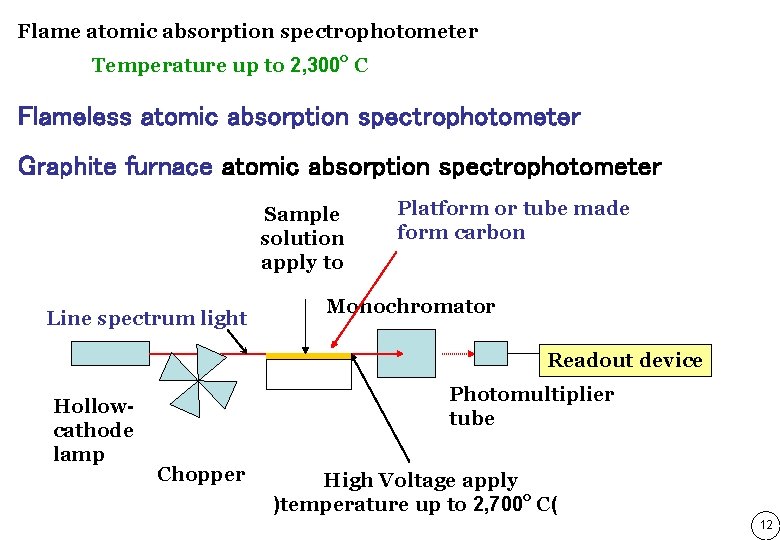
Flame atomic absorption spectrophotometer Temperature up to 2, 300 o C Flameless atomic absorption spectrophotometer Graphite furnace atomic absorption spectrophotometer Sample solution apply to Line spectrum light Platform or tube made form carbon Monochromator Readout device Hollowcathode lamp Photomultiplier tube Chopper High Voltage apply )temperature up to 2, 700 o C( 12
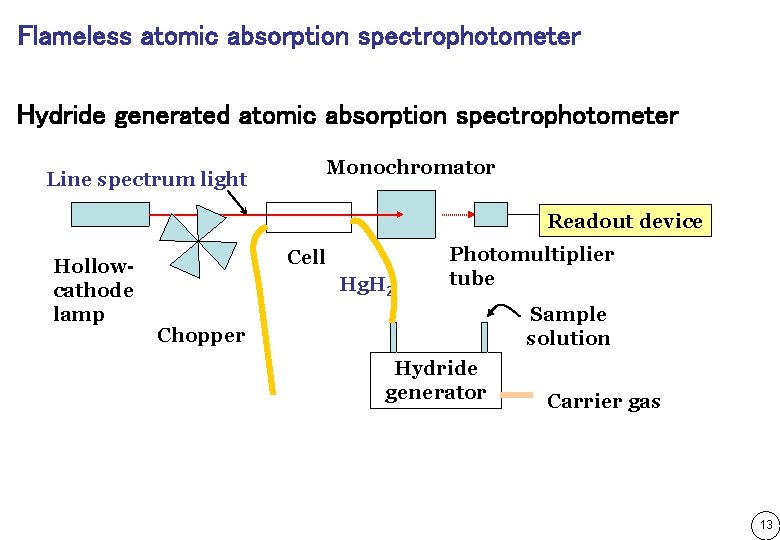
Flameless atomic absorption spectrophotometer Hydride generated atomic absorption spectrophotometer Line spectrum light Monochromator Readout device Hollowcathode lamp Cell Hg. H 2 Photomultiplier tube Sample solution Chopper Hydride generator Carrier gas 13
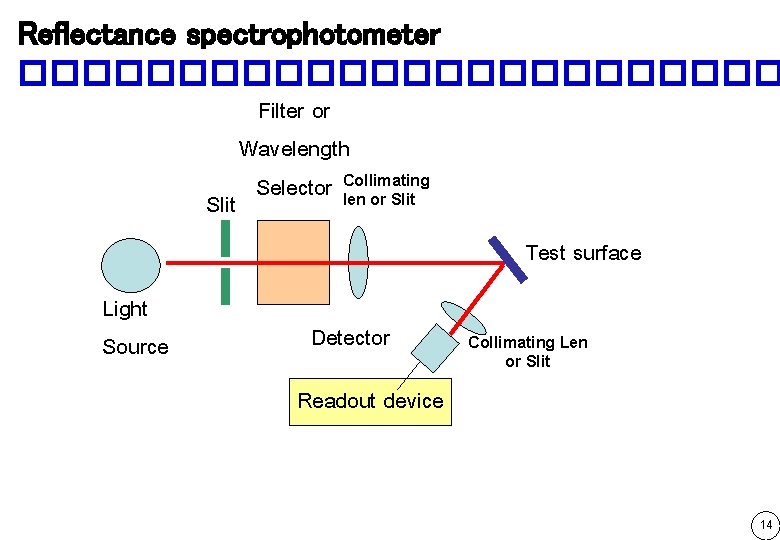
Reflectance spectrophotometer ������������ Filter or Wavelength Selector Collimating len or Slit Test surface Light Source Detector Collimating Len or Slit Readout device 14
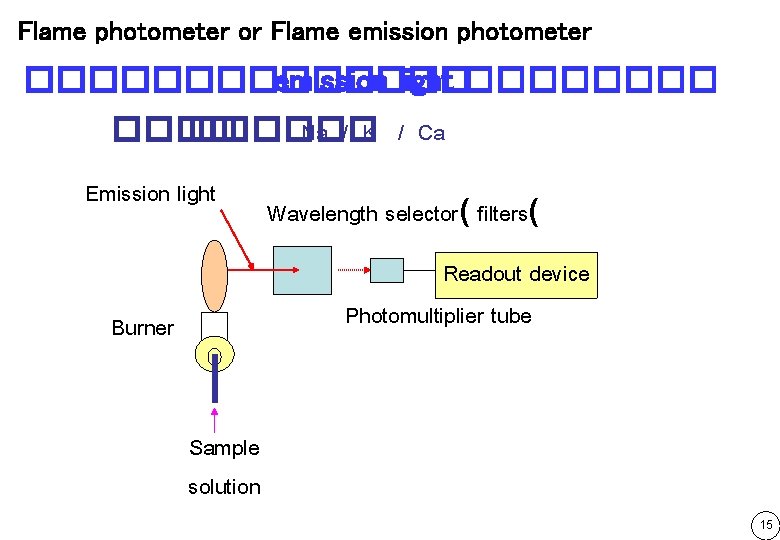
Flame photometer or Flame emission photometer ������� emission light ������ Na / K / Ca Emission light Wavelength selector( filters( Readout device Photomultiplier tube Burner Sample solution 15
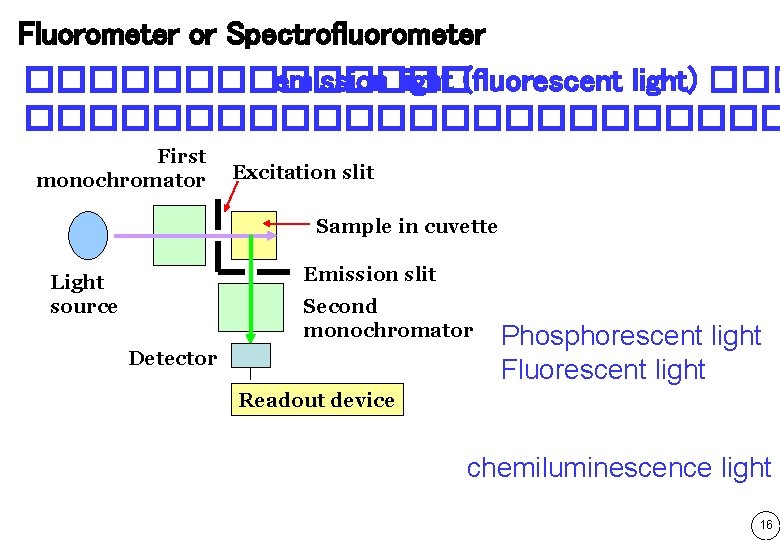
Fluorometer or Spectrofluorometer ������� emission light (fluorescent light) �������������� First monochromator Excitation slit Sample in cuvette Emission slit Light source Second monochromator Detector Phosphorescent light Fluorescent light Readout device chemiluminescence light 16
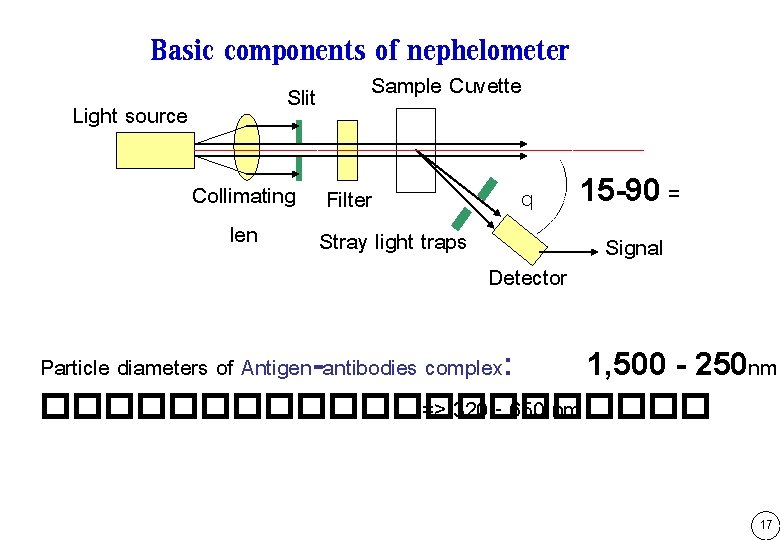
Basic components of nephelometer Light source Slit Sample Cuvette Collimating Filter len Stray light traps q Detector 15 -90 = Signal Particle diameters of Antigen-antibodies complex: 1, 500 - 250 nm ����������� => 320 - 650 nm 17
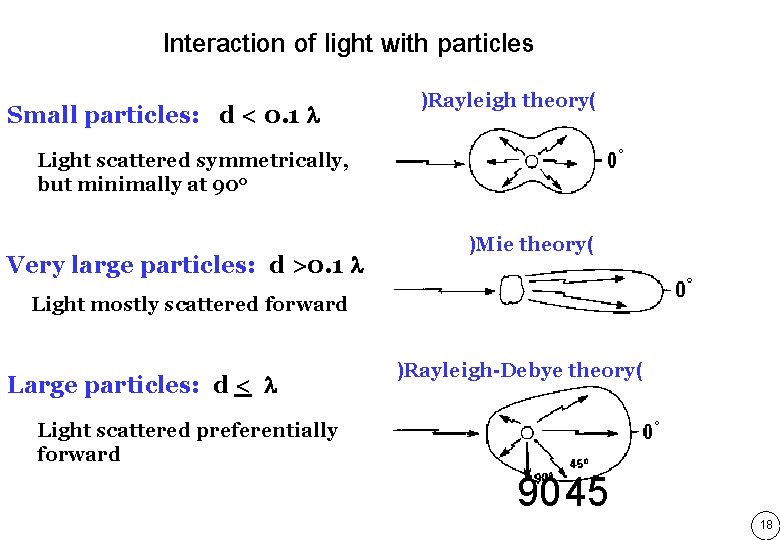
Interaction of light with particles Small particles: d < 0. 1 )Rayleigh theory( Light scattered symmetrically, but minimally at 90 o Very large particles: d >0. 1 )Mie theory( Light mostly scattered forward Large particles: d < )Rayleigh-Debye theory( Light scattered preferentially forward 9045 18
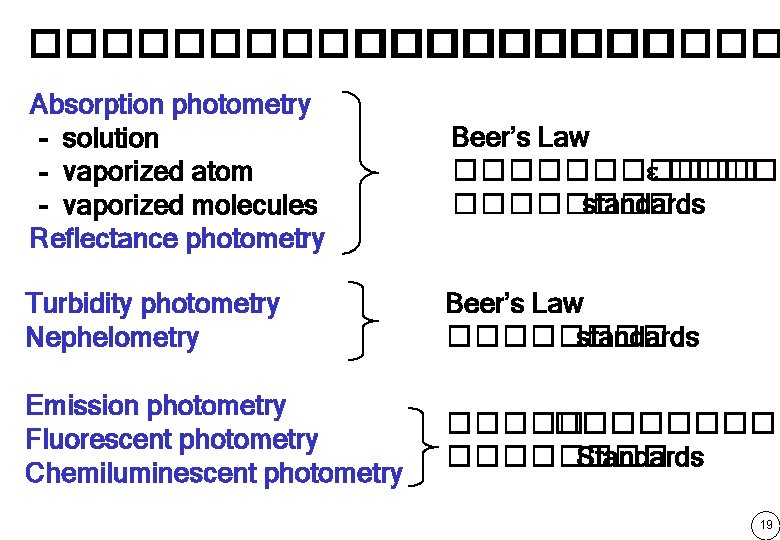
��������� Absorption photometry - solution - vaporized atom - vaporized molecules Reflectance photometry Beer’s Law ������ e �������� standards Turbidity photometry Nephelometry Beer’s Law ���� standards Emission photometry Fluorescent photometry Chemiluminescent photometry ��������� Standards 19
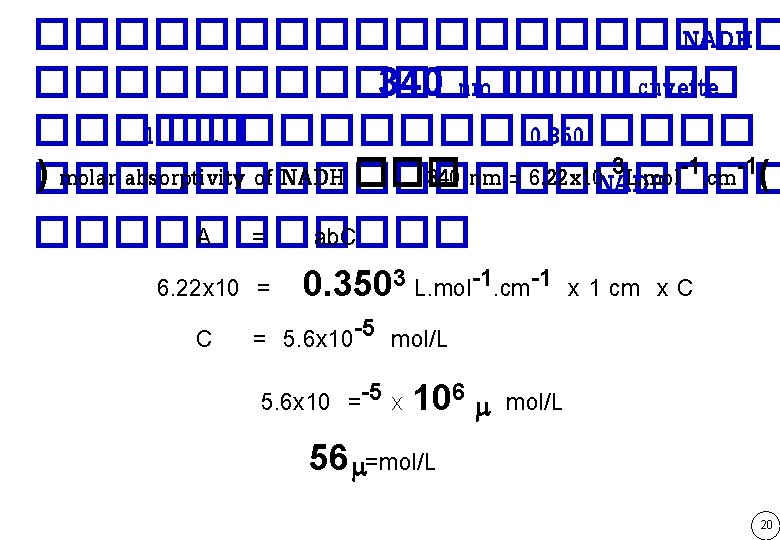
���������� NADH �������� 340 nm ������ cuvette ����� 1 ��. ������� 0. 350 3 L. mol-1. cm-1( )���������� molar absorptivity of NADH ��� 340 nm = 6. 22 x 10 NADH A = ab. C ������ 6. 22 x 10 = 0. 3503 L. mol-1. cm-1 x 1 cm x C C = 5. 6 x 10 -5 mol/L 5. 6 x 10 =-5 x 106 m mol/L 56 m=mol/L 20
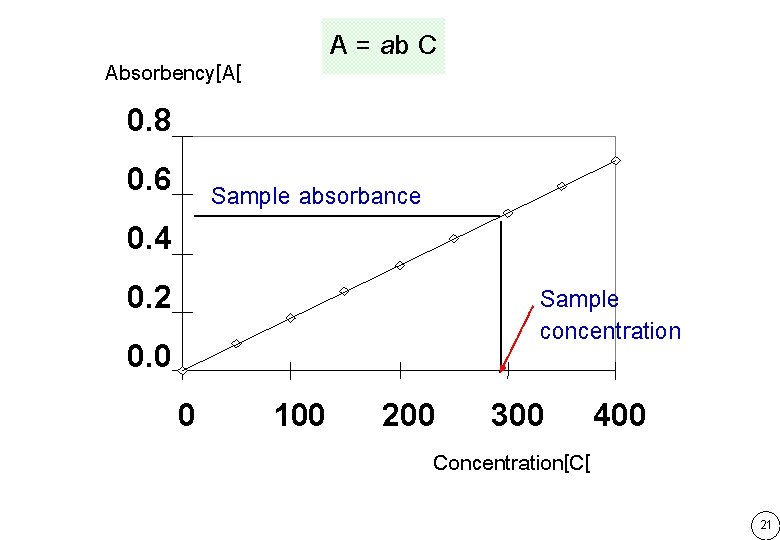
A = ab C Absorbency[A[ 0. 8 0. 6 Sample absorbance 0. 4 Sample concentration 0. 2 0. 0 0 100 200 300 400 Concentration[C[ 21
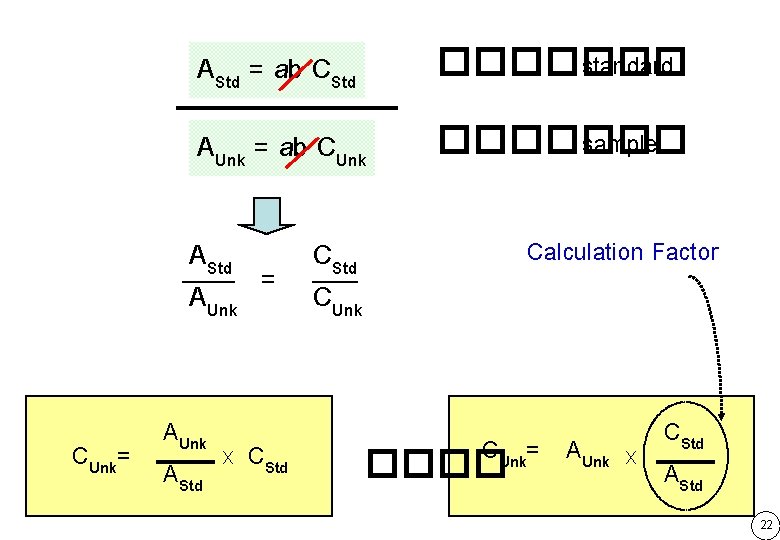
AStd = ab CStd ������� standard AUnk = ab CUnk ������� sample AStd CStd = AUnk CUnk= A x CStd Calculation Factor CUnk= AUnk ���� CStd x AStd 22

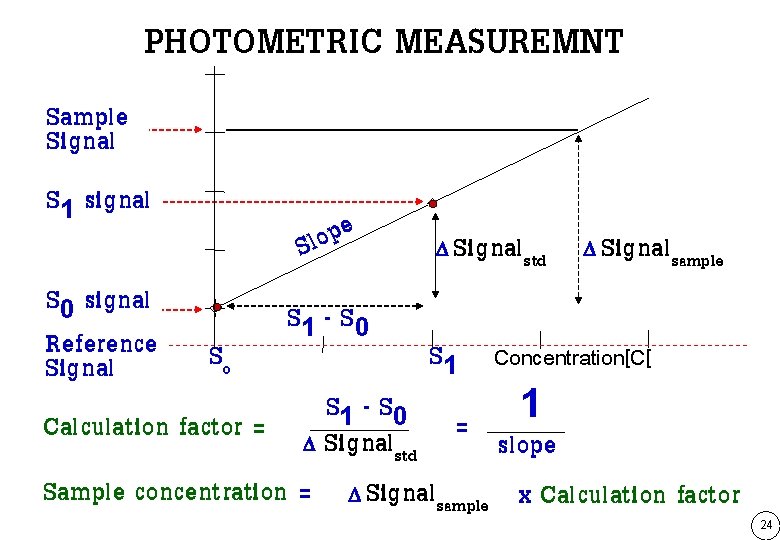
PHOTOMETRIC MEASUREMNT Sample Signal S 1 signal e p o Sl Signalstd Signalsample S 0 signal S 1 - S 0 Reference S S o 1 Concentration[C[ Signal S 1 - S 0 = 1 Calculation factor = Signal slope std Sample concentration = Signalsample x Calculation factor 24
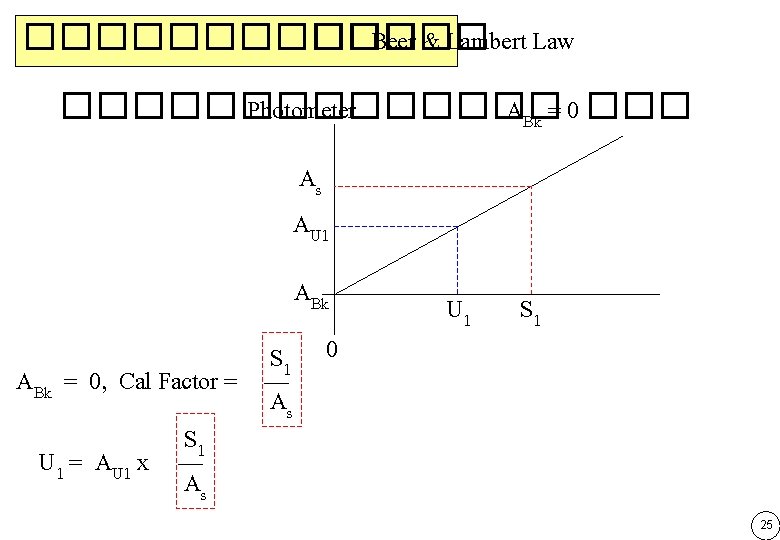
������� ��� Beer & Lambert Law ����� Photometer ���� ABk = 0 ��� As AU 1 ABk S 1 0 ABk = 0, Cal Factor = As S 1 U 1 = AU 1 x As U 1 S 1 25
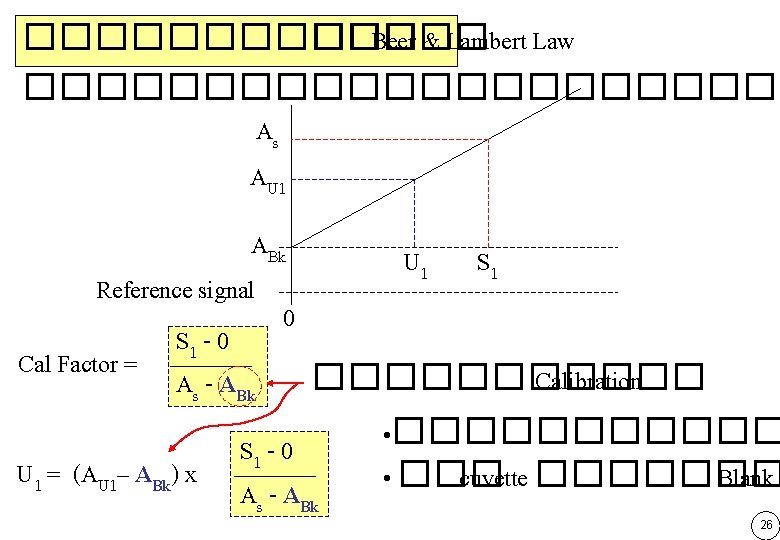
������� ��� Beer & Lambert Law ����������� As AU 1 ABk U 1 S 1 Reference signal 0 S 1 - 0 Cal Factor = ������ Calibration As - ABk • ������ S 1 - 0 U 1 = (AU 1– ABk) x • ��� cuvette ������� Blank � A -A s Bk 26
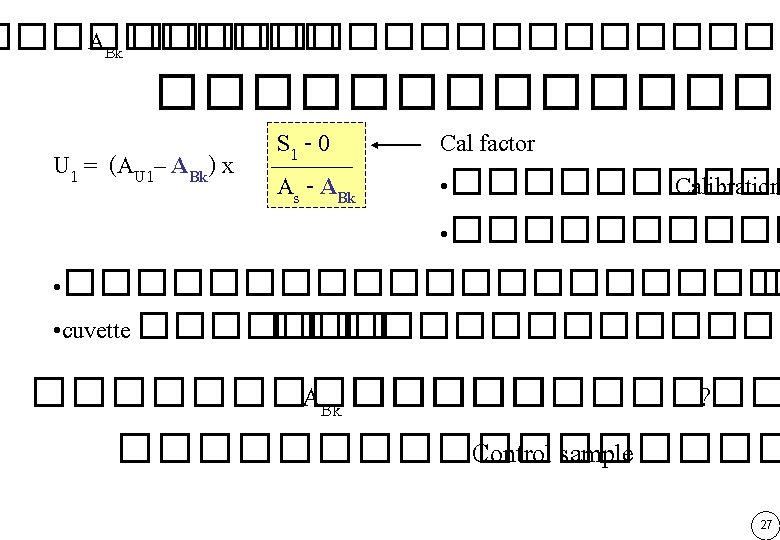
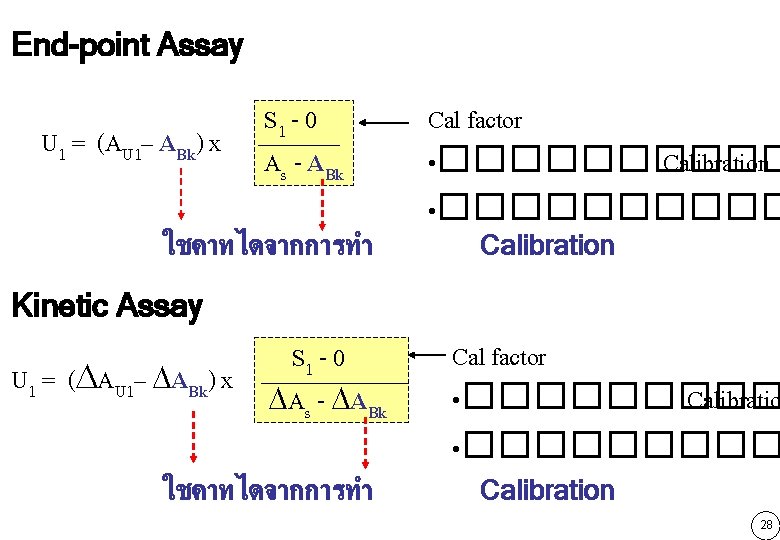
End-point Assay U 1 = (AU 1– ABk) x S 1 - 0 As - ABk ใชคาทไดจากการทำ Cal factor • ���������� Calibration Kinetic Assay S 1 - 0 U 1 = ( AU 1– ABk) x As - ABk ใชคาทไดจากการทำ Cal factor • ��������� Calibration 28
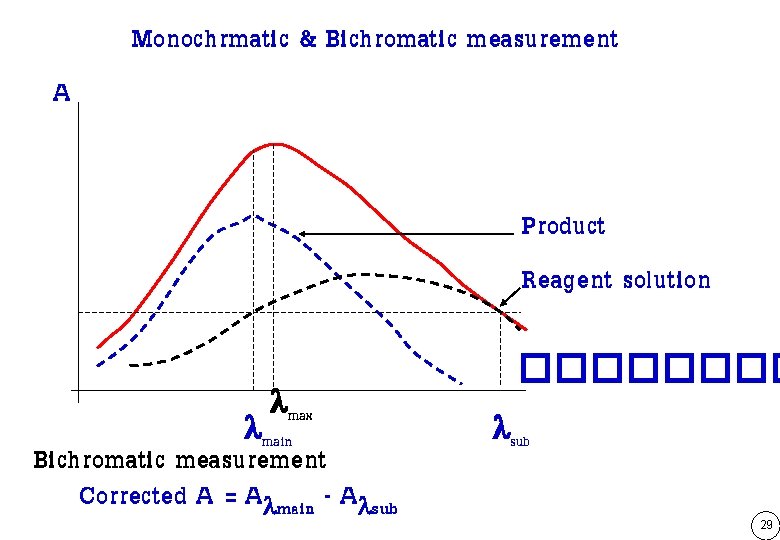
Monochrmatic & Bichromatic measurement A Product Reagent solution max main Bichromatic measurement Corrected A = A main - A sub ���� sub 29
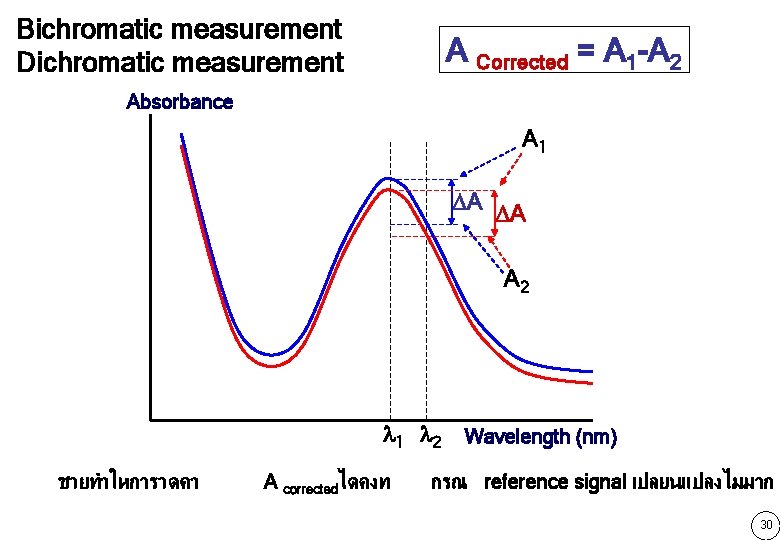
Bichromatic measurement Dichromatic measurement A Corrected = A 1 -A 2 Absorbance A 1 A A A 2 1 2 Wavelength (nm) ชวยทำใหการวดคา A correctedไดคงท กรณ reference signal เปลยนแปลงไมมาก 30
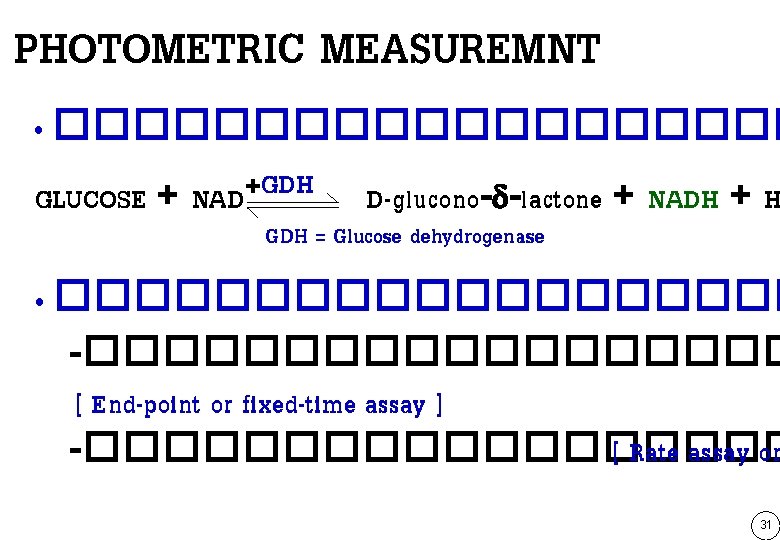
PHOTOMETRIC MEASUREMNT • ���������� GLUCOSE + NAD+GDH D-glucono-d-lactone + NADH + H GDH = Glucose dehydrogenase • ���������� -��������� [ End-point or fixed-time assay ] -��������� [ Rate assay or 31

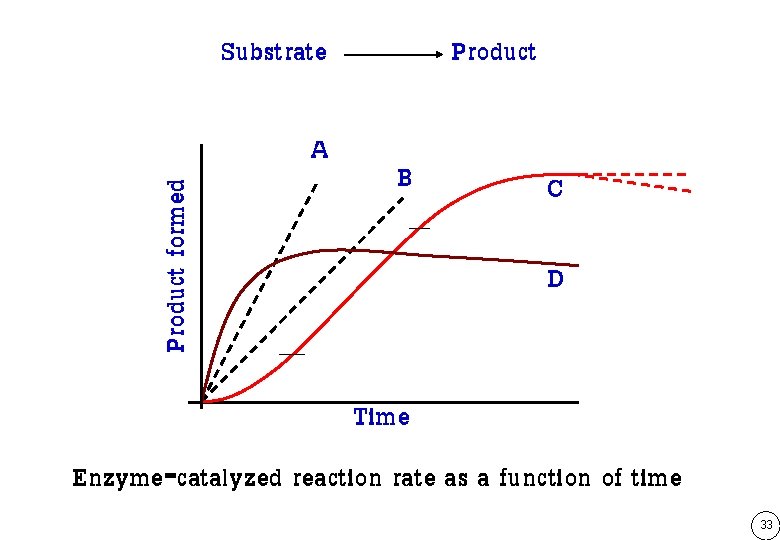
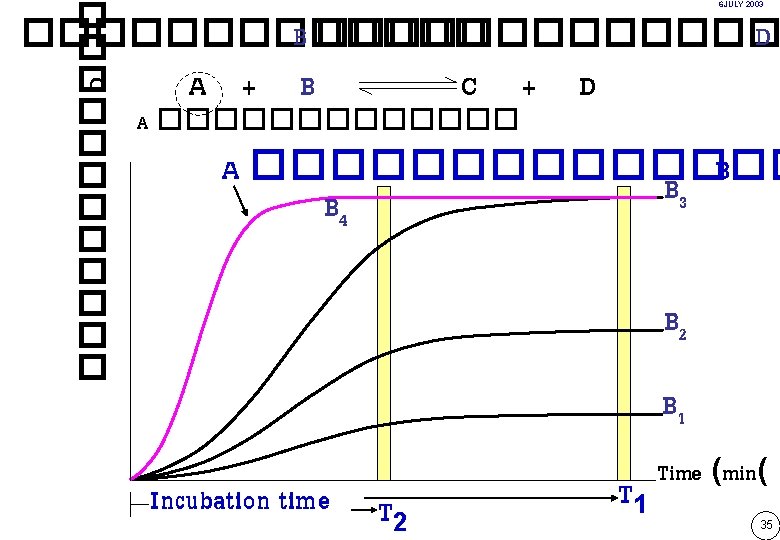
Substrate Product formed A Product B C D Time Enzyme-catalyzed reaction rate as a function of time 33
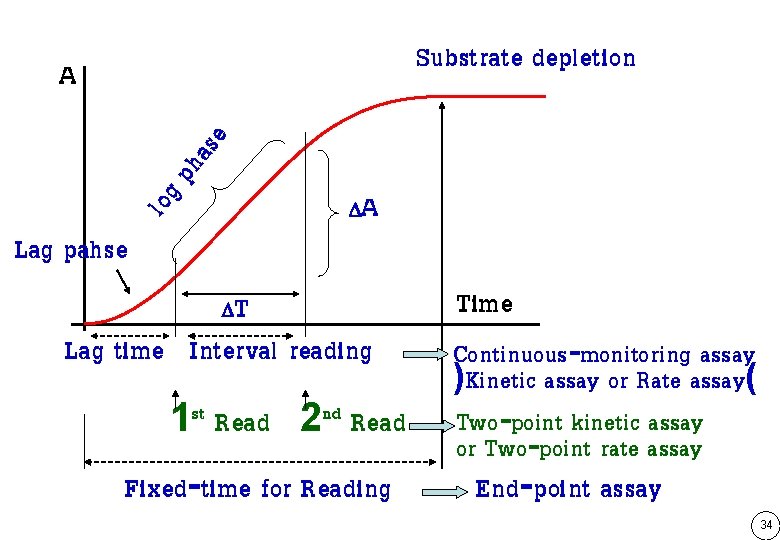
Substrate depletion log pha se A Lag pahse A T Lag time Interval reading 1 st Read 2 nd Read Fixed-time for Reading Time Continuous-monitoring assay )Kinetic assay or Rate assay( Two-point kinetic assay or Two-point rate assay End-point assay 34
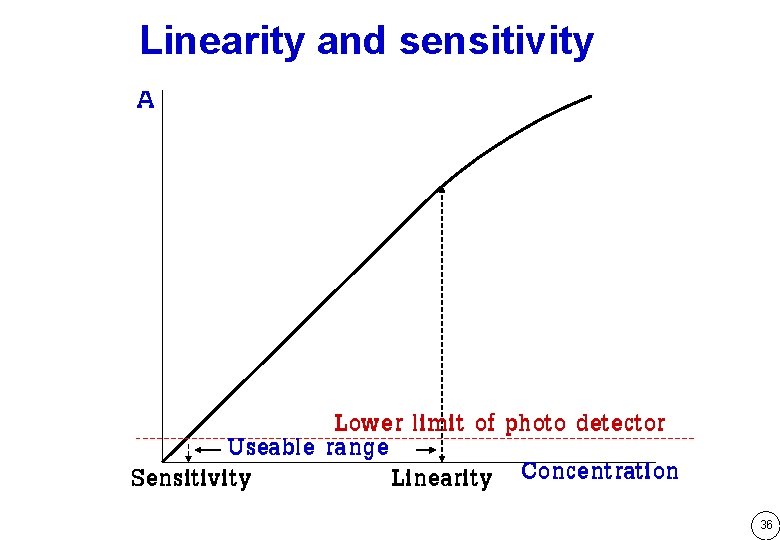
Linearity and sensitivity A Lower limit of photo detector Useable range Sensitivity Linearity Concentration 36
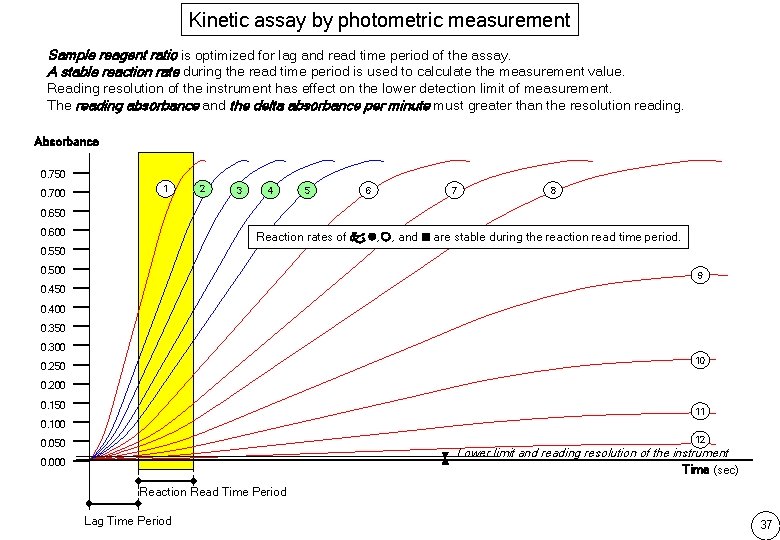
Kinetic assay by photometric measurement Sample reagent ratio is optimized for lag and read time period of the assay. A stable reaction rate during the read time period is used to calculate the measurement value. Reading resolution of the instrument has effect on the lower detection limit of measurement. The reading absorbance and the delta absorbance per minute must greater than the resolution reading. Absorbance 0. 750 0. 700 1 2 3 4 5 6 7 8 0. 650 0. 600 Reaction rates of , , , and are stable during the reaction read time period. 0. 550 0. 500 9 0. 450 0. 400 0. 350 0. 300 10 0. 250 0. 200 0. 150 11 0. 100 12 0. 050 Lower limit and reading resolution of the instrument 0. 000 Time (sec) Reaction Read Time Period Lag Time Period 37
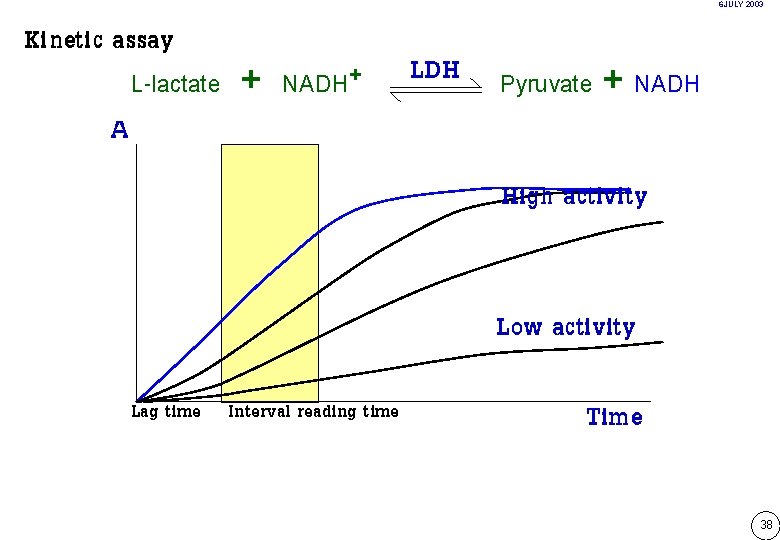
6 JULY 2003 Kinetic assay A L-lactate + NADH+ LDH Pyruvate + NADH High activity Low activity Lag time Interval reading time Time 38

LDH Pyruvate + NADH Time Absorbance A/30 sec. A/min. 00: 00 Lag time 00: 01: 000. 300 00: 01: 300. 316 0. 032 00: 02: 000. 330 0. 014 0. 002 0. 028 00: 02: 300. 346 0. 016 0. 002 0. 032 00: 03: 000. 360 0. 014 0. 002 0. 028 0. 030 ������������� LDH Activity (U/L) = A/min ������� cuve Calculation factor ����� Interval reading L-lactate + NADH+ 39
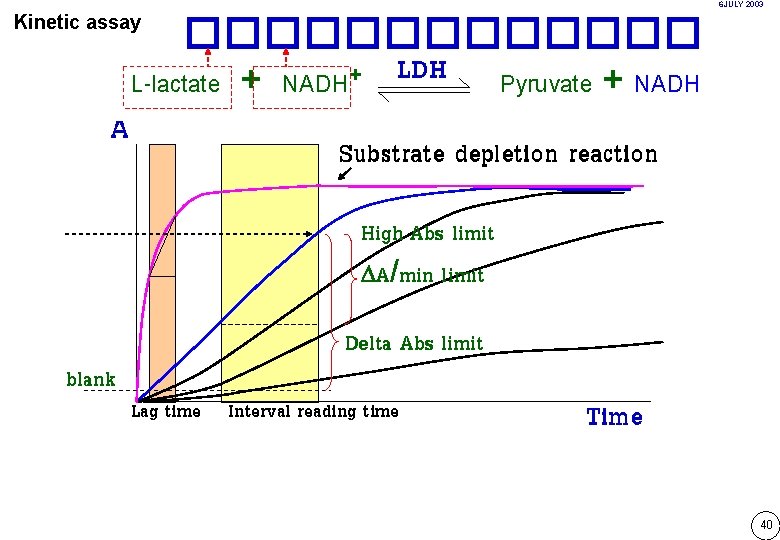
6 JULY 2003 Kinetic assay A ������� LDH + L-lactate + NADH Pyruvate + NADH Substrate depletion reaction High Abs limit A/min limit blank Delta Abs limit Lag time Interval reading time Time 40
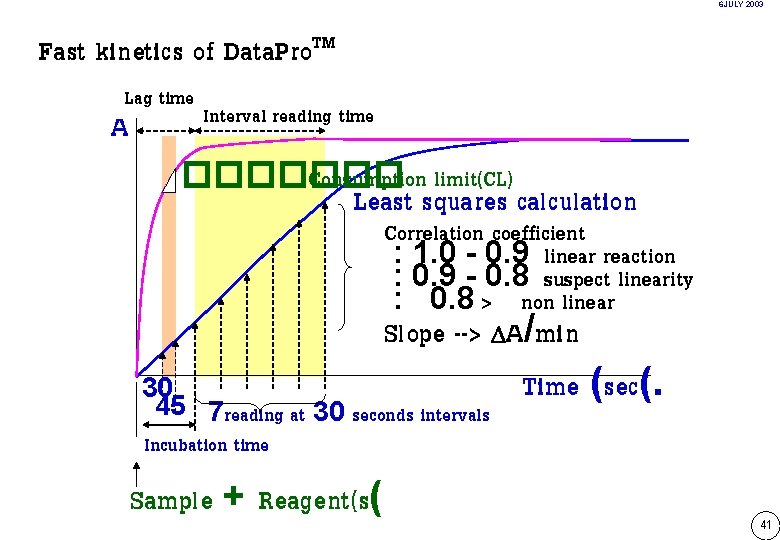
6 JULY 2003 Fast kinetics of Data. Pro. TM Lag time A Interval reading time ������� Consumption limit(CL) Least squares calculation Correlation coefficient : 1. 0 - 0. 9 linear reaction : 0. 9 - 0. 8 suspect linearity : 0. 8 > non linear Slope --> A/min Time (sec(. 30 45 7 reading at 30 seconds intervals Incubation time Sample + Reagent(s( 41
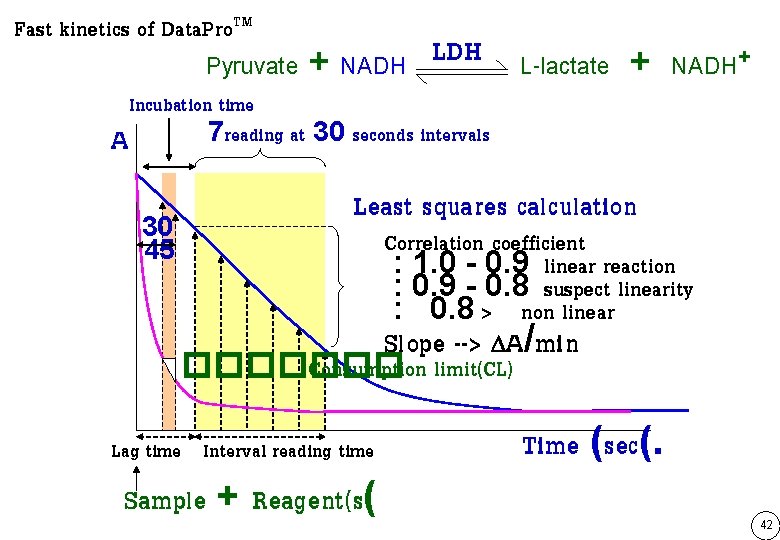
Fast kinetics of Data. Pro. TM Pyruvate + NADH LDH Incubation time 7 reading at 30 seconds intervals A 30 45 L-lactate + NADH+ Least squares calculation Correlation coefficient : 1. 0 - 0. 9 linear reaction : 0. 9 - 0. 8 suspect linearity : 0. 8 > non linear Slope --> A/min ������� Consumption limit(CL) Lag time Interval reading time Sample + Reagent(s( Time (sec(. 42

คำอธบายระบบทำงานของเครองวเคราะหอตโนมต Batch analysis A number of specimens are processed in the same analytical session, or run. Sequential analysis Each specimen in the batch enters the analytical process one after another, and each result or set of results emerges in the same order as specimens are entered. Parallel analysis All specimens are subjected to a series of analytical processes at the same time in a parallel fashion. 43
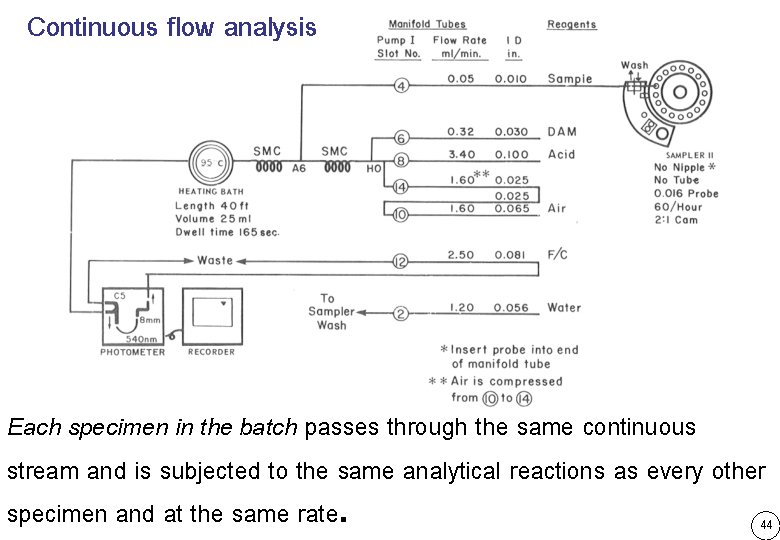
Continuous flow analysis Each specimen in the batch passes through the same continuous stream and is subjected to the same analytical reactions as every other specimen and at the same rate. 44
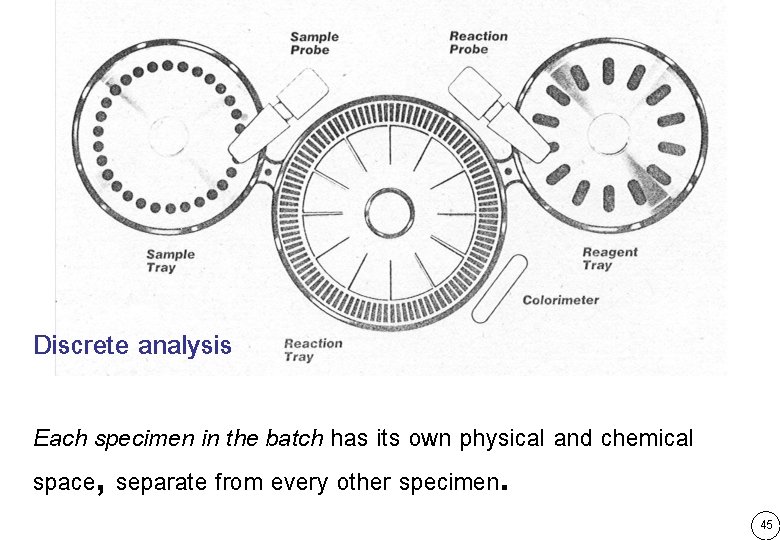
Discrete analysis Each specimen in the batch has its own physical and chemical space, separate from every other specimen. 45

Single-channel analysis ( single test analysis( Each specimen is subjected to a single process so that results for a single analyte are produced. Multi-channel analysis ( multitest analysis( Each specimen is subjected to multiple analytical processes so that a set of test results is obtained. Continuous flow multi-channel analysis Discretionary multi-channel analysis 46

��������� • Reaction cuvette Tested cuvette & Reagent blank cuvette • Pipetting probe Precision Contamination during pipetting • Photometric accuracy Energy lamp Wavelength accuracy Electrical noise 47

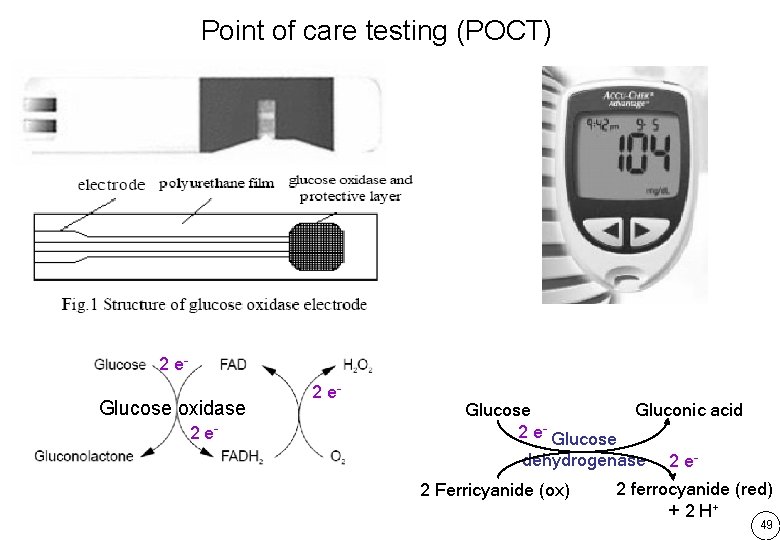
Point of care testing (POCT) 2 e- Glucose oxidase 2 e- Glucose Gluconic acid dehydrogenase 2 Ferricyanide (ox) 2 e- 2 ferrocyanide (red) + 2 H+ 49
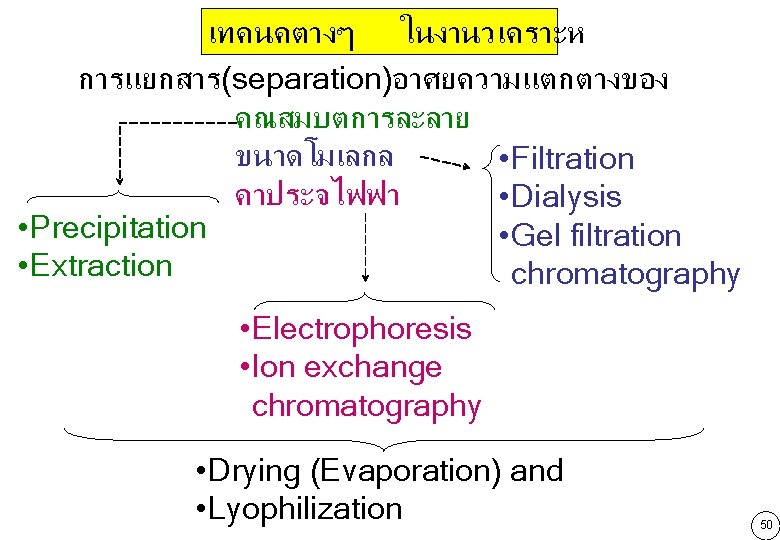
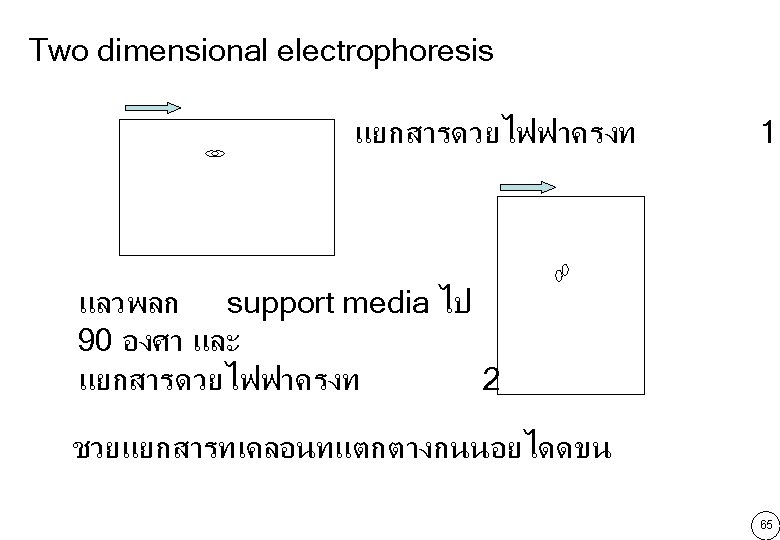
เทคนคตางๆ ในงานวเคราะห การแยกสาร(separation)อาศยความแตกตางของ คณสมบตการละลาย ขนาดโมเลกล • Filtration คาประจไฟฟา • Dialysis • Precipitation • Gel filtration • Extraction chromatography • Electrophoresis • Ion exchange chromatography • Drying (Evaporation) and • Lyophilization 50
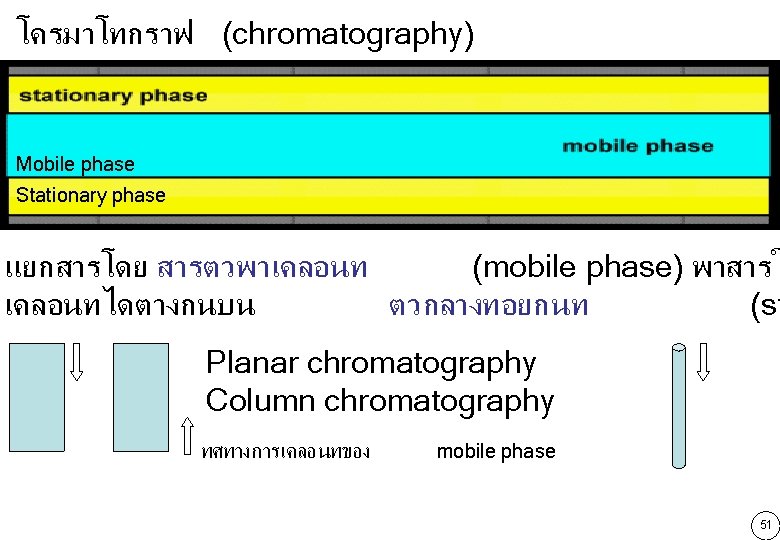
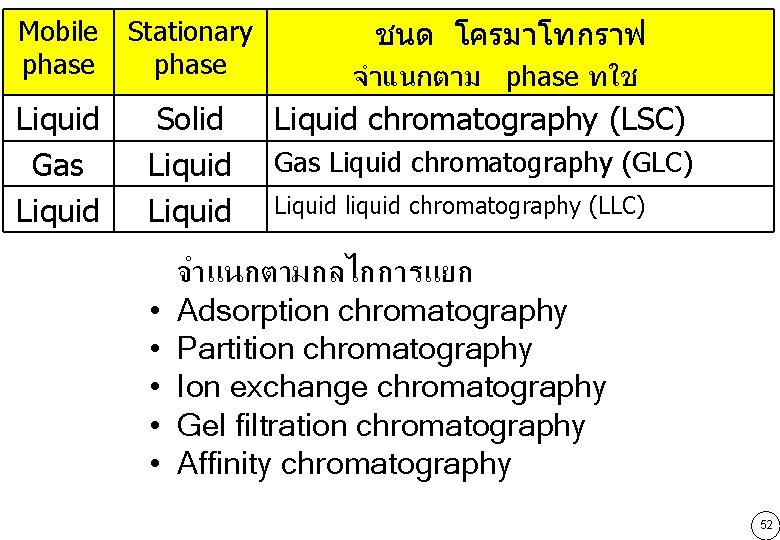
Mobile phase Stationary phase Liquid Gas Liquid Solid Liquid • • • ชนด โครมาโทกราฟ จำแนกตาม phase ทใช Liquid chromatography (LSC) Gas Liquid chromatography (GLC) Liquid liquid chromatography (LLC) จำแนกตามกลไกการแยก Adsorption chromatography Partition chromatography Ion exchange chromatography Gel filtration chromatography Affinity chromatography 52
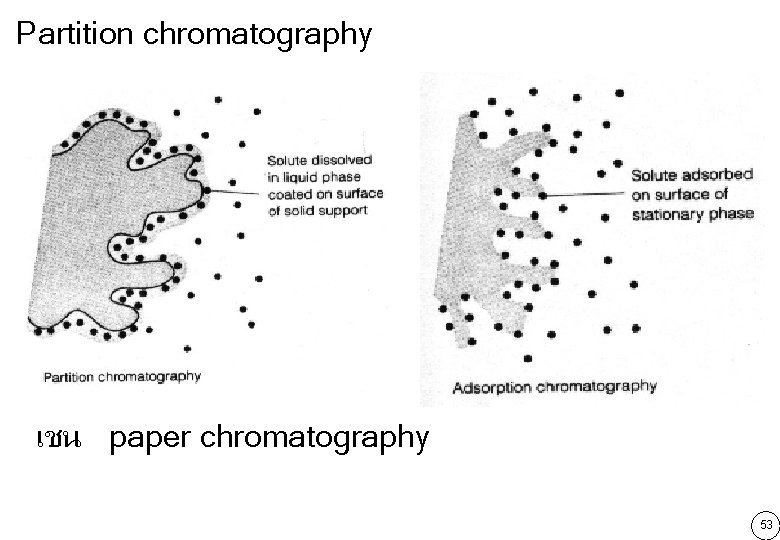
Partition chromatography เชน paper chromatography 53
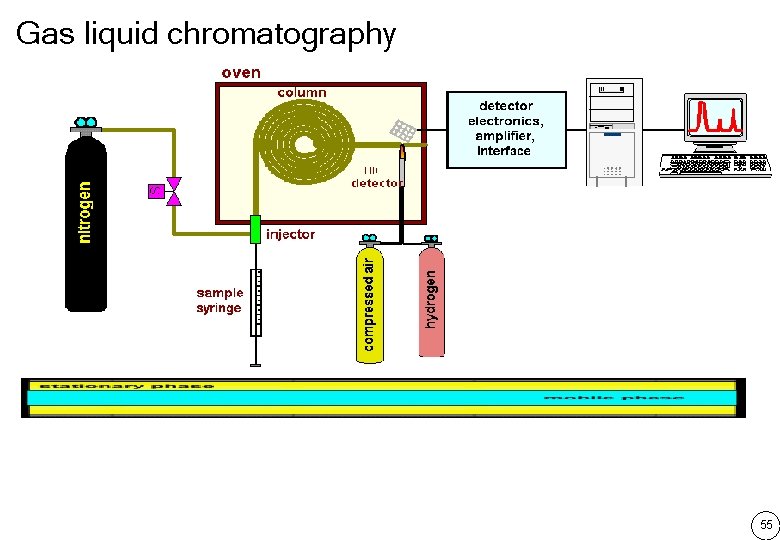
Gas liquid chromatography 55
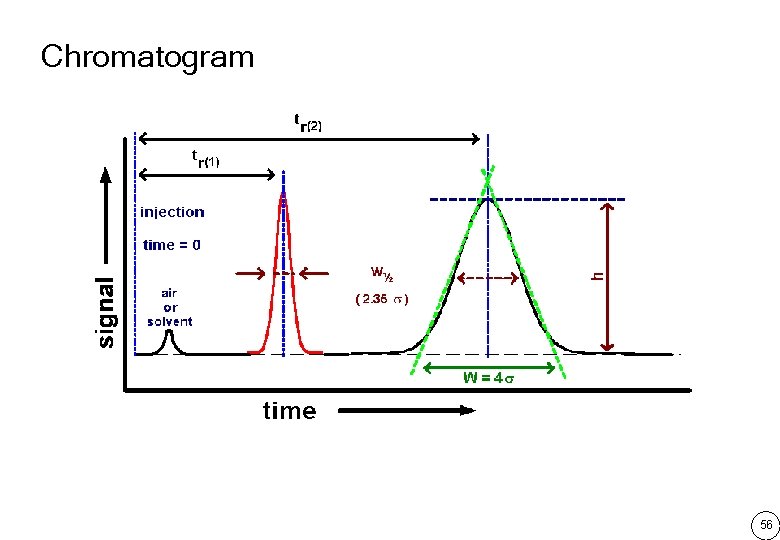
Chromatogram 56

Gel filtration chromatography or Molecular Exclusion Chromatography 57

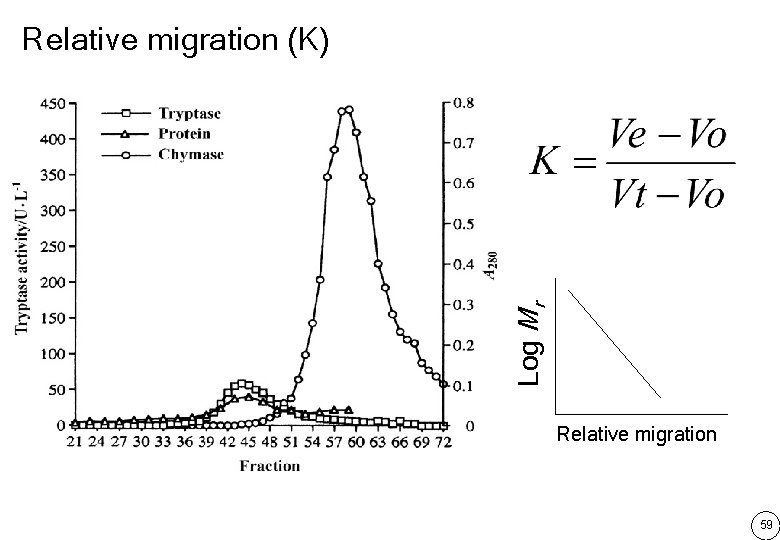
Log Mr Relative migration (K) Relative migration 59

Ion exchange chromatography Matrix Ion exchanger • Anion-exchanger มประจลบ • Cation-exchanger มประจบวก 60
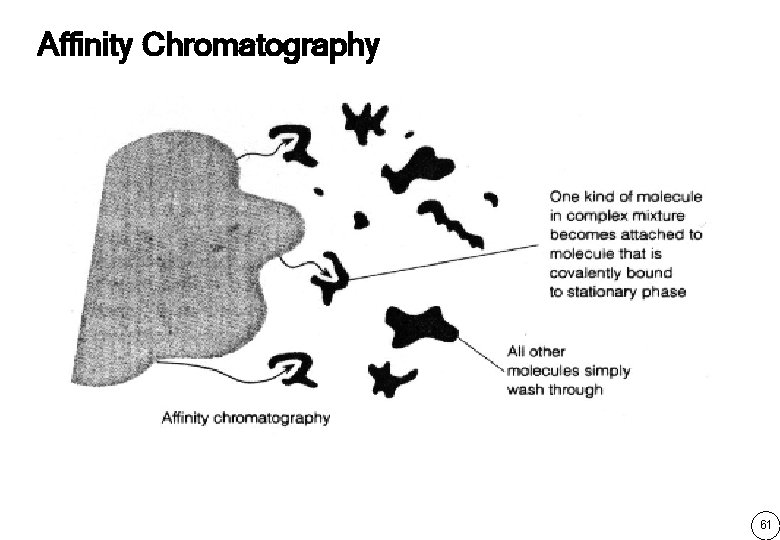
Affinity Chromatography 61

Type of electrophoresis แบงตามชนดของตวกลางคำจน Paper electrophoresis Cellulose electrophoresis Starch gel electrophoresis Agarose gel electrophoresis Polyacrylamide gel electrophoresis Polyacylamide gel เกดจากการ cross-link ของ acrylamide กบ methylene-bis acrylamide โดยม ammonium persulfate เปน ตวเรง และ TEMED เปนตวทำใหเกดอนมลอสระเพอเรมตนปฏกรยา cross-link 62
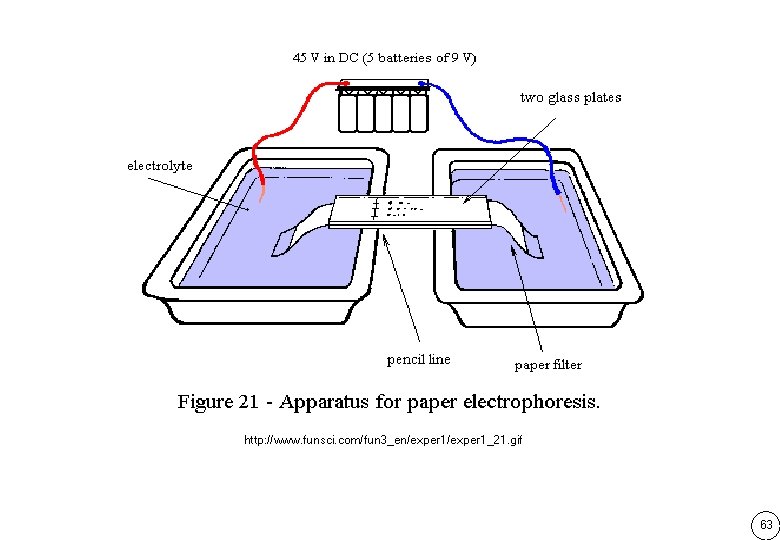
http: //www. funsci. com/fun 3_en/exper 1_21. gif 63
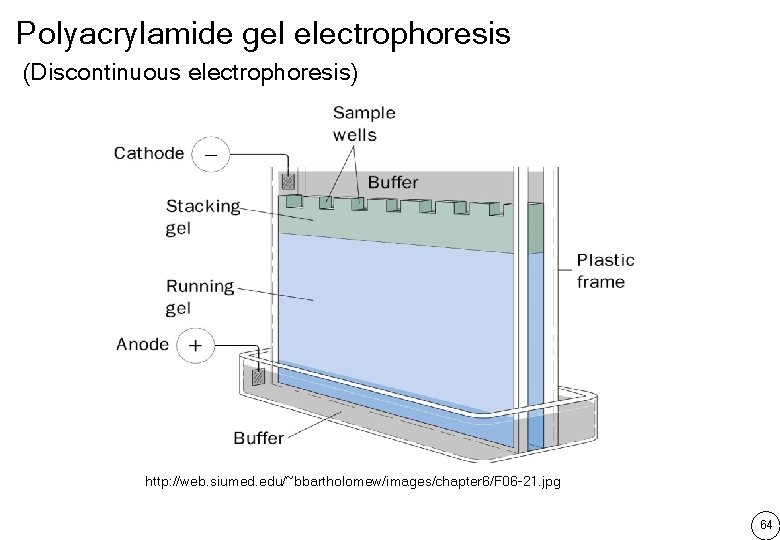
Polyacrylamide gel electrophoresis (Discontinuous electrophoresis) http: //web. siumed. edu/~bbartholomew/images/chapter 6/F 06 -21. jpg 64




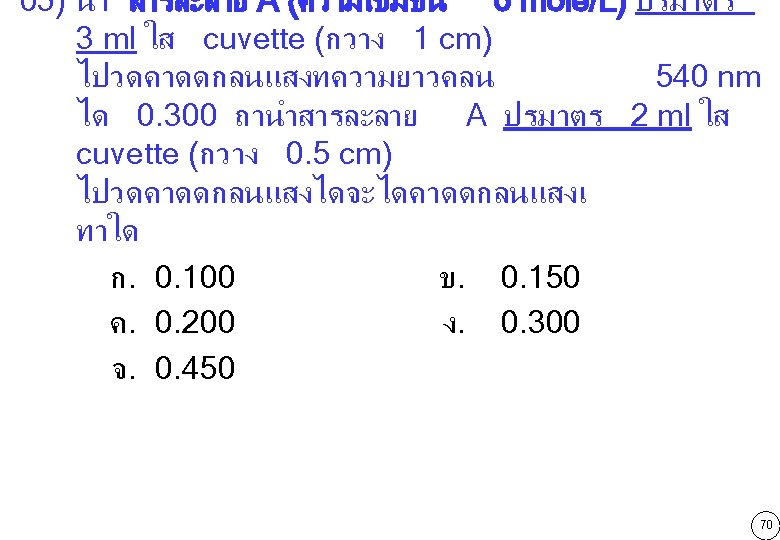

http: //home. kku. ac. th/wiskun 71
- Slides: 71