SPECIFIC TEST Formic acid Acetic acid Turbidity or

SPECIFIC TEST
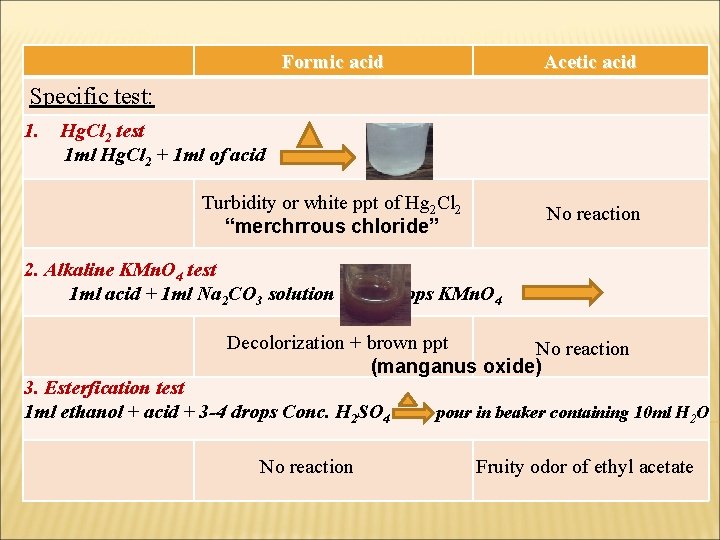
Formic acid Acetic acid Turbidity or white ppt of Hg 2 Cl 2 “merchrrous chloride” No reaction Specific test: 1. Hg. Cl 2 test 1 ml Hg. Cl 2 + 1 ml of acid 2. Alkaline KMn. O 4 test 1 ml acid + 1 ml Na 2 CO 3 solution + 2 -3 drops KMn. O 4 Decolorization + brown ppt No reaction (manganus oxide) 3. Esterfication test 1 ml ethanol + acid + 3 -4 drops Conc. H 2 SO 4 No reaction pour in beaker containing 10 ml H 2 O Fruity odor of ethyl acetate

Tartaric acid Citric acid Specific test: 1. Conc. H 2 SO 4 test Acid + 4 drops Conc. H 2 SO 4 Effervescence + heavy charring 2. Dengi’s test Acid + H 2 O + Dengi’s reagent 2 drops No reaction boil Effervescence + yellow color add KMn. O 4 [WHILE IT’S HOT] Decolorization + white ppt
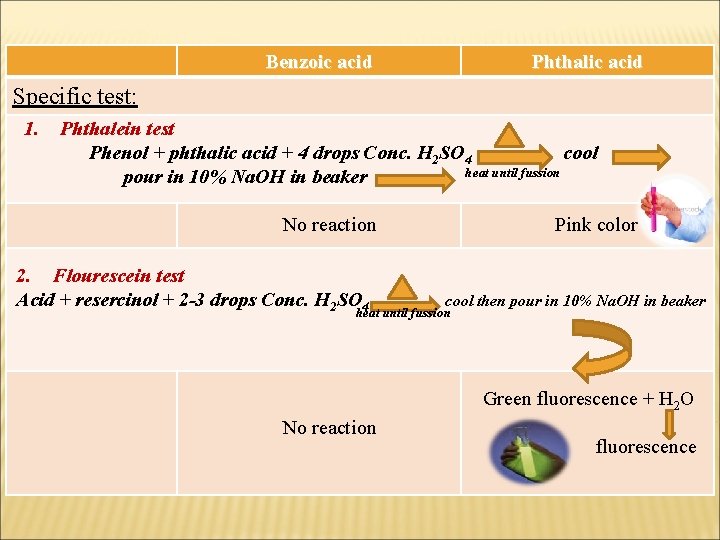
Benzoic acid Phthalic acid Specific test: 1. Phthalein test Phenol + phthalic acid + 4 drops Conc. H 2 SO 4 cool heat until fussion pour in 10% Na. OH in beaker No reaction 2. Flourescein test Acid + resercinol + 2 -3 drops Conc. H 2 SO 4 Pink color cool then pour in 10% Na. OH in beaker heat until fussion Green fluorescence + H 2 O No reaction fluorescence

Specific test of Salicylic acid: 1. Formaldehyde test 1 ml formaldehyde + few gm salicylic acid + 4 drops Conc. H 2 SO 4 (on the wall) Crimson red color 2. Esterfication test 1 ml ethanol + few gm salicylic acid + 3 -4 drops Conc. H 2 SO 4 pour in beaker containing 10 ml H 2 O Oil of winter green “ Vicks odor due to methyl salicylate” 2. Phthalein test Few gm Salicylic acid + few gm phthalic anhydride + 4 drops Conc. H 2 SO 4 cool pour in 10 ml 10% Na. OH in beaker Pink color
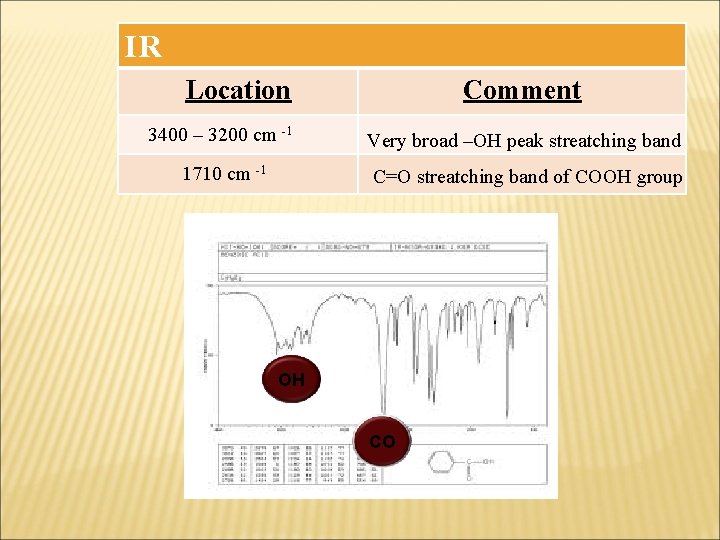
IR Location Comment 3400 – 3200 cm -1 Very broad –OH peak streatching band 1710 cm -1 C=O streatching band of COOH group OH CO
- Slides: 6