Specific Identification of Organic Pollutants in the Catawba

Specific Identification of Organic Pollutants in the Catawba Watershed Wastewater Treatment Plant Effluents John Turner Faculty Mentor: Dr. Pat Owens Faculty Committee Members: Dr. Pat Owens, Dr. Cliff Calloway, Dr. Peter Phillips

Threats posed by organics • Many are toxic (harmful), carcinogenic (cancercausing), or mutagenic (capable of altering genetic material) at very low levels • Many organics have been shown to be endocrine disruptors capable of altering the sex of fishes • May have the capability for bioaccumulation in the fat cells and membranes

From The Charlotte Observer October 4 th, 2004

The Status Quo • Presently there is no testing of organic levels in WWTP effluents within the Catawba watershed • Furthermore, testing of drinking water and river water for organics is not being done. • Some organics such as 1, 4 -dioxane have been known to travel as far as 200 kilometers from its point of entry in a watershed. (Draper et al, 2000)

Research Design and Methods • Collection of WWTP samples – Bottle washing procedure • Glass bottles (approximately 600 m. L) were used with glass stoppers • 2 m. L HPLC grade Methanol added to each bottle • Stoppers returned to bottle and rolled and flipped so that Methanol completely covered the inside of the bottle • This was then repeated using reagent grade Ethyl Acetate • Unstopped bottles were placed in a dry oven at 108 o. C for 24 hours

Research Design and Methods • Collection of WWTP samples – Samples were collected from 2 WWTPs • Samples were collected from Rock Hill (Manchester Creek) and Fort Mill WWTPs. • The samples that were collected were composite samples
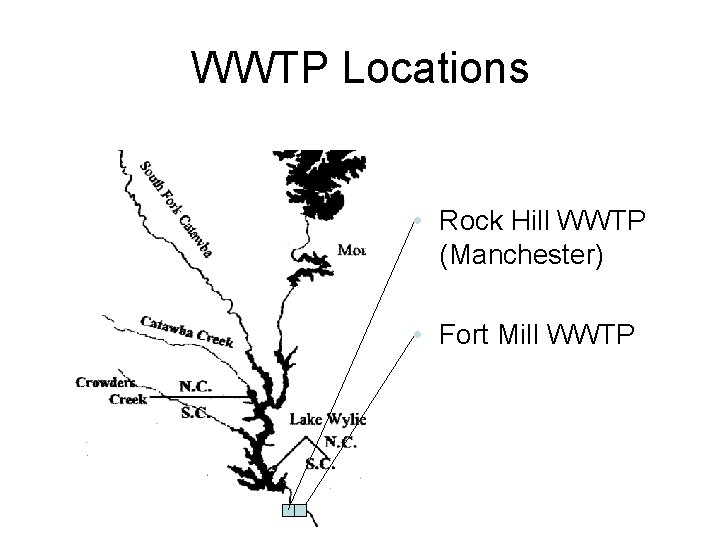
WWTP Locations • Rock Hill WWTP (Manchester) • Fort Mill WWTP

Research Design and Methods • Organic Extraction – The Basic Apparatus Glass Funnel Membrane Filter Vacuum Hose Erlenmeyer Vacuum Flask

Research Design and Methods • Organic Extraction – Empore C 18 Membrane Filter Preparation • An Empore C 18 Membrane Filter was placed at the bottom of the Glass Funnel on the screen. • 25 grams of Empore Filter Aid glass beads were added to the funnel • The disk washed with 20 m. L of elution solvent (methanol or ethyl acetate) and then allowed to dry • 20 m. L of methanol was used to condition the disk

Research Design and Methods • Organic Extraction – Concentration of Organics onto the filter • The sample was decanted into the glass funnel. • The vacuum was then applied and the sample was allowed to run through the disk. • The vacuum was allowed to continue to run for 5 -20 minutes so that residual water was removed.

Research Design and Methods • Organic Extraction – Organic Elution • The organics were eluted from the disk into an organic phase, and they were collected by placing a Kuderna-Danish flask under the membrane filter. • 10 m. L of elution solvent was then added (either methanol or ethyl acetate) so that it covered the disk completely • The vacuum was used to pull the solvent that now contains the organics into the Kuderna-Danish flask.

Research Design and Methods • Organic Extraction – Concentration of Elution • Once the organics were concentrated into 10 m. L of solvent they were further concentrated for analysis. • Two m. L of elution solvent was removed. • Then the remaining 8 m. L of elution solvent was evaporated down to approximately 1 m. L. • This was done by blowing a gentle stream of nitrogen that passed through an organic carbon filter over the solvent.
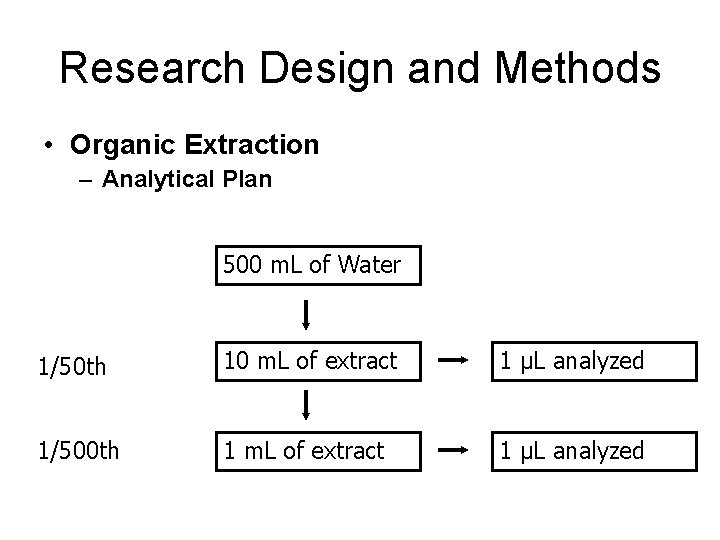
Research Design and Methods • Organic Extraction – Analytical Plan 500 m. L of Water 1/50 th 10 m. L of extract 1 μL analyzed 1/500 th 1 m. L of extract 1 μL analyzed

Research Design and Methods • Run Gas Chromatography-Mass Spectrometry • 1 μl of extract was injected into the GC column • The extract contains a mixture of the organics that were originally found in the wastewater but have now been concentrated into an organic phase. • The GC column separated the analytes based upon their polarity. • The MS measured the molecular weight of the various ions and a chromatogram was produced.

Results • Siloxanes • Phenol • Phthalates
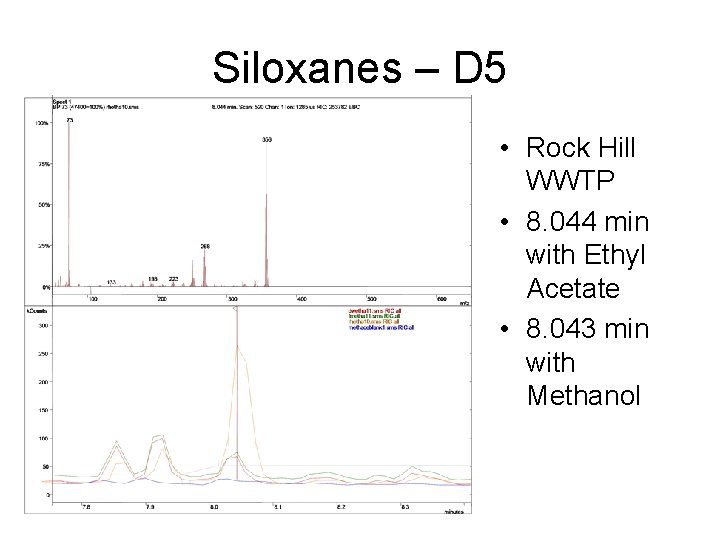
Siloxanes – D 5 • Rock Hill WWTP • 8. 044 min with Ethyl Acetate • 8. 043 min with Methanol
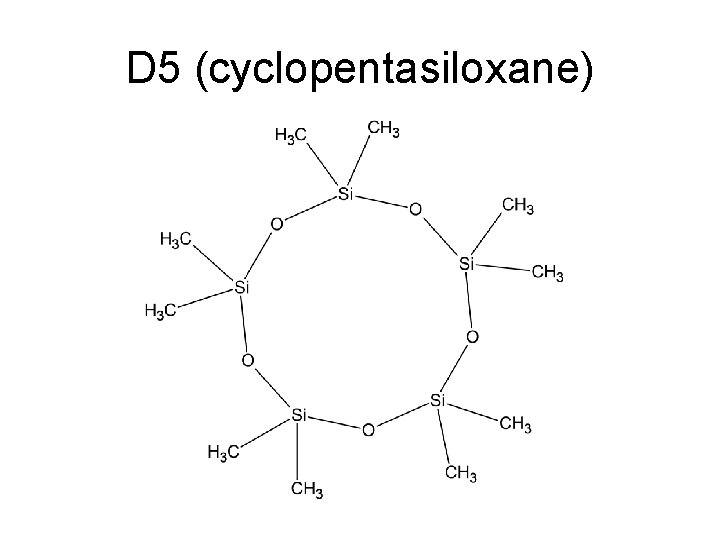
D 5 (cyclopentasiloxane)
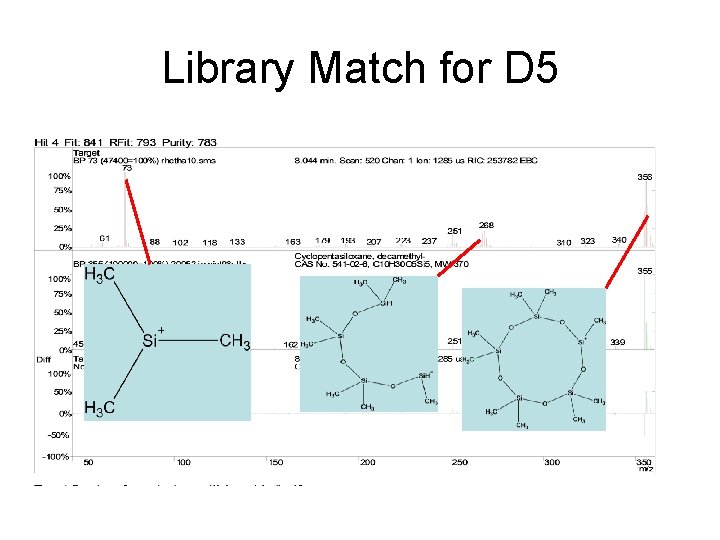
Library Match for D 5
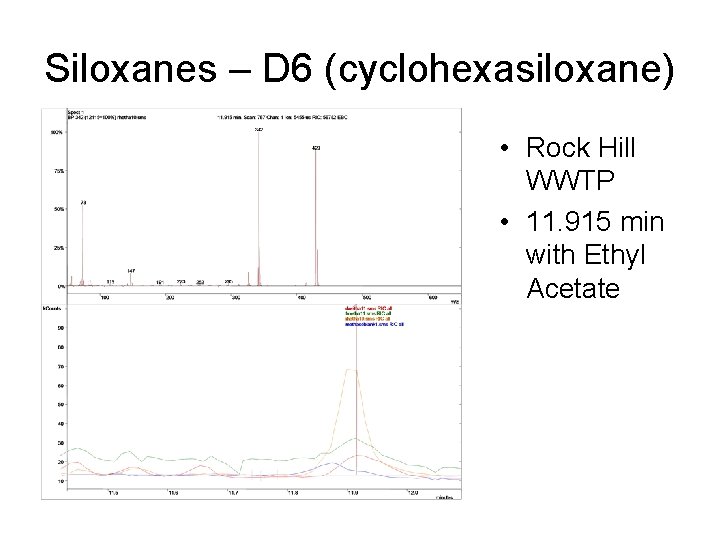
Siloxanes – D 6 (cyclohexasiloxane) • Rock Hill WWTP • 11. 915 min with Ethyl Acetate

Library Match for D 6 Loss of 88
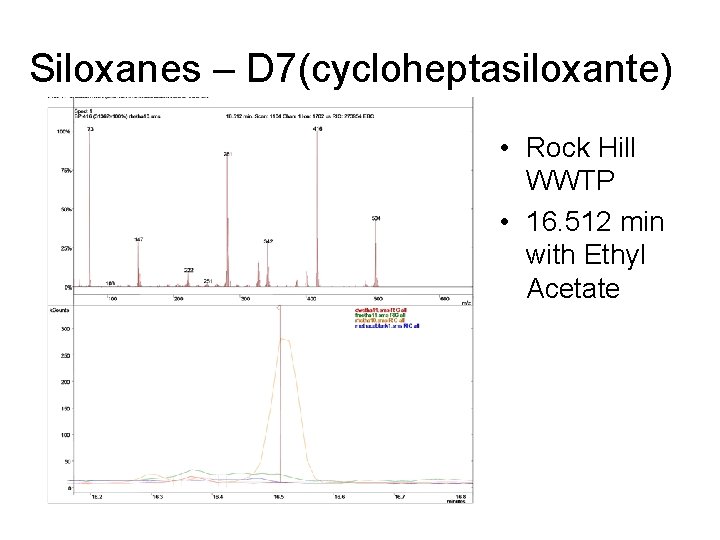
Siloxanes – D 7(cycloheptasiloxante) • Rock Hill WWTP • 16. 512 min with Ethyl Acetate
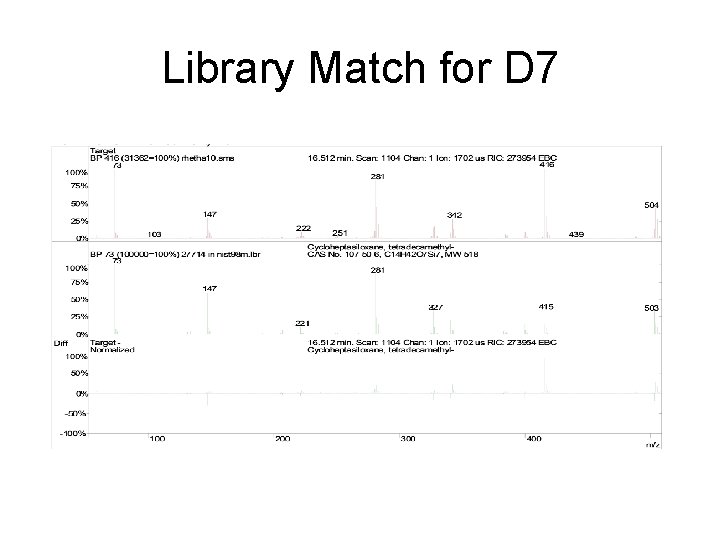
Library Match for D 7
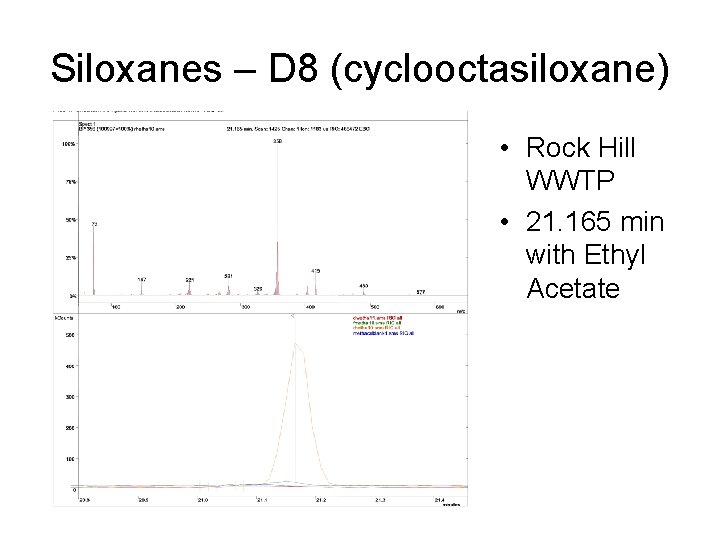
Siloxanes – D 8 (cyclooctasiloxane) • Rock Hill WWTP • 21. 165 min with Ethyl Acetate
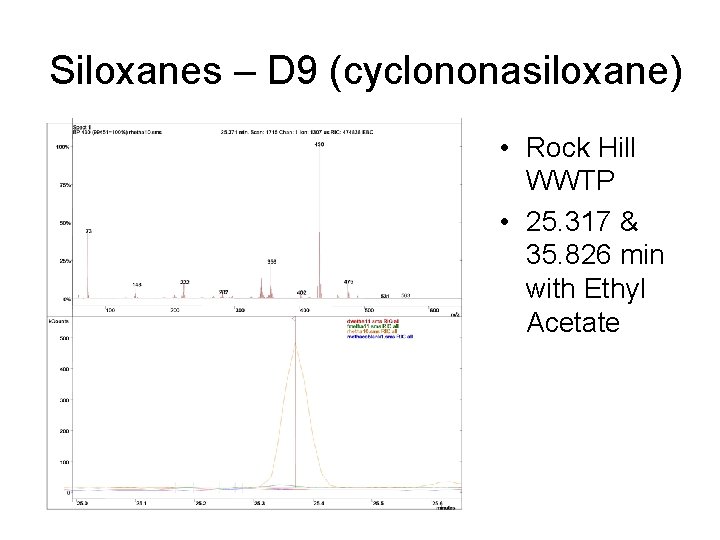
Siloxanes – D 9 (cyclononasiloxane) • Rock Hill WWTP • 25. 317 & 35. 826 min with Ethyl Acetate
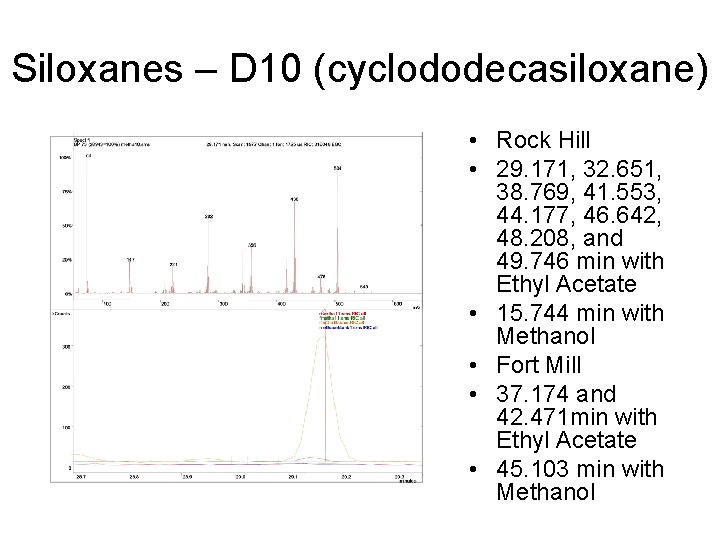
Siloxanes – D 10 (cyclododecasiloxane) • Rock Hill • 29. 171, 32. 651, 38. 769, 41. 553, 44. 177, 46. 642, 48. 208, and 49. 746 min with Ethyl Acetate • 15. 744 min with Methanol • Fort Mill • 37. 174 and 42. 471 min with Ethyl Acetate • 45. 103 min with Methanol
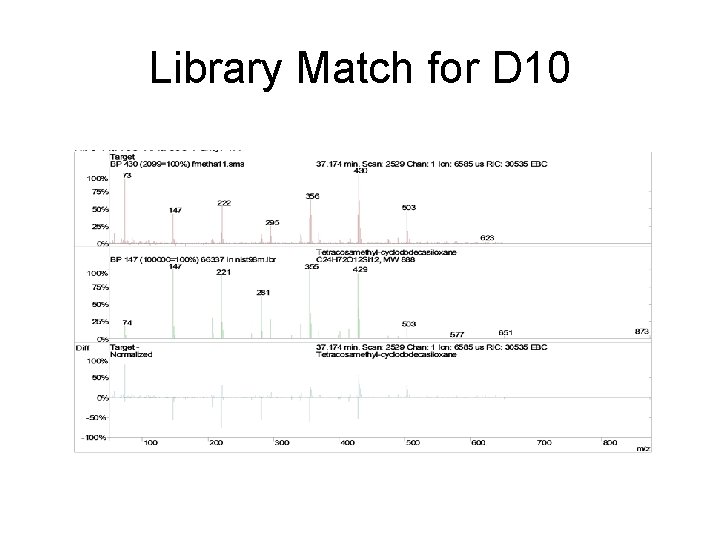
Library Match for D 10

Risks Associated with Siloxanes • Bioaccumulation in tissues and organs • Capable of inhibiting estrogen, leading to reproductive problems
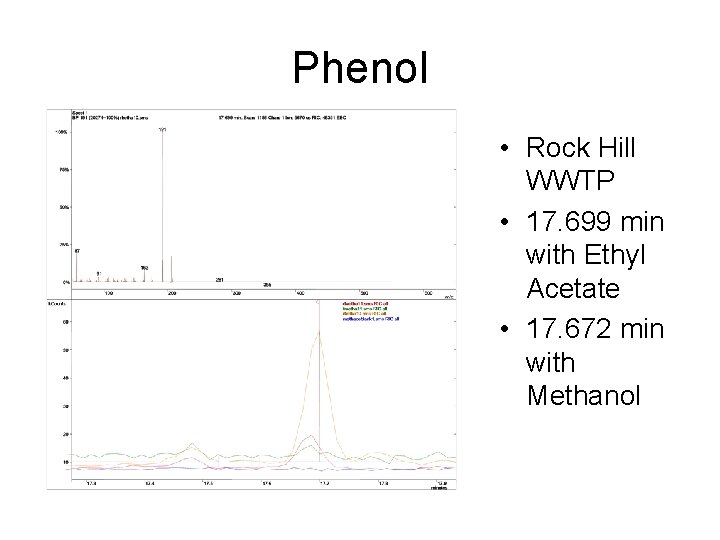
Phenol • Rock Hill WWTP • 17. 699 min with Ethyl Acetate • 17. 672 min with Methanol

Library Match for Phenol

Risks Associated with Phenol • Most affects are related to the exposure to large quantities over time • Probably not a major problem unless found in substantial quantities

Bis (2 -ethylhexyl) phthalate • Rock Hill WWTP • 42. 699 min with Methanol • Fort Mill • 42. 726 min with Methanol

Library Match for a Phthalate

Risks Associated with Bis (2 -ethylhexyl) phthalate • Has been shown to bioaccumulate in tissue • Affects the abilities of males to father babies

Conclusions • GC-MS was shown to be an effective means of identifying organics in wastewater • Within the organics identified, the siloxanes and phthalates have serious health effects at very low levels • This research established a basic understanding of the use of GC-MS in identifying organics in wastewater and has accumulated information to fuel further research in this area in the future at Winthrop
- Slides: 34