SPECIALTY PHARMACY MARKET OVERVIEW By John Sugrue What

SPECIALTY PHARMACY MARKET OVERVIEW By: John Sugrue

What is a specialty drug? ■ May have special handling and/or storage requirements ■ Usually not oral medications ■ Could need healthcare provider administration ■ May require regular monitoring (electrolytes, drug levels, etc…) ■ Often requires a prior authorization through patient’s insurance

Common Specialty Conditions ■ Cancer ■ Anemia ■ Multiple Sclerosis ■ Hepatitis ■ Cystic Fibrosis ■ Psoriasis ■ Rheumatoid Arthritis ■ Hemophilia

Orphan Drug Act - 1983 ■ Orphan Drug – meets the needs of a disease in which less than 200, 000 people are afflicted. ■ Provides incentives for drug companies to develop drugs for rare conditions ■ Less than 10 specialty drugs were developed between 1973 and 1983 ■ Since then, over 400 specialty drugs have been FDA approved http: //www. fda. gov/For. Industry/Developing. Productsfor. Rare. Diseases. Conditions/ucm 2005525. htm
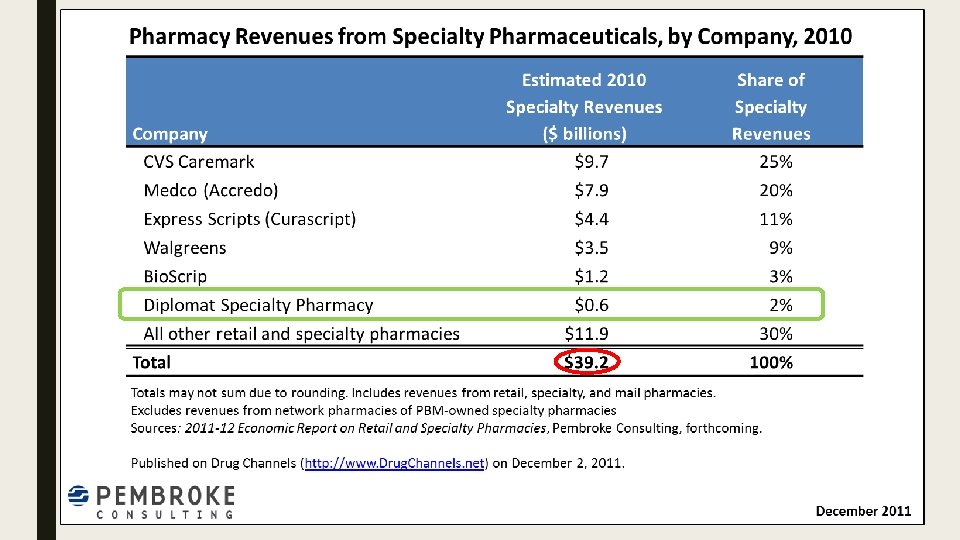
![SPECIALTY PHARMACY MARKET SHARE 2010 Top 3 56% [CATEGORY NAME] [PERCENTAGE] Top 4 65% SPECIALTY PHARMACY MARKET SHARE 2010 Top 3 56% [CATEGORY NAME] [PERCENTAGE] Top 4 65%](http://slidetodoc.com/presentation_image_h2/a110c7976a85e18baf99bc4e48786951/image-6.jpg)
SPECIALTY PHARMACY MARKET SHARE 2010 Top 3 56% [CATEGORY NAME] [PERCENTAGE] Top 4 65% [CATEGORY NAME] [PERCENTAGE] [CATEGORY NAME] [PERCENTAGE] Diplomat 2%
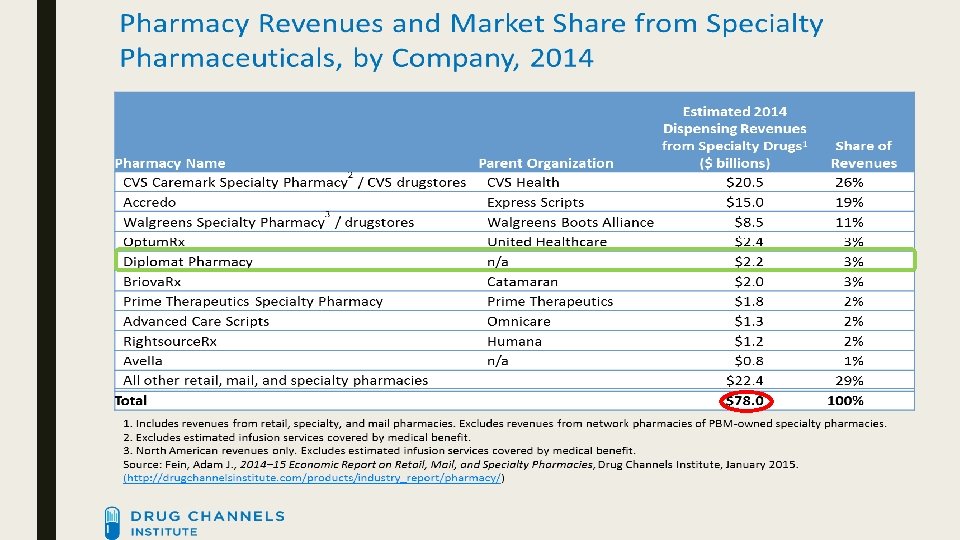
![SPECIALTY PHARMACY MARKET SHARE 2014 [CATEGORY NAME] [PERCENTAGE] Top 3 56% Top 4 59% SPECIALTY PHARMACY MARKET SHARE 2014 [CATEGORY NAME] [PERCENTAGE] Top 3 56% Top 4 59%](http://slidetodoc.com/presentation_image_h2/a110c7976a85e18baf99bc4e48786951/image-8.jpg)
SPECIALTY PHARMACY MARKET SHARE 2014 [CATEGORY NAME] [PERCENTAGE] Top 3 56% Top 4 59% [CATEGO RY NAME] [CATEGORY NAME] [PERCENTAGE] [CATEGORY NAME] [PERCENTAGE] [CATEGORY NAME] [PERCENTAGE] Diplomat 3%
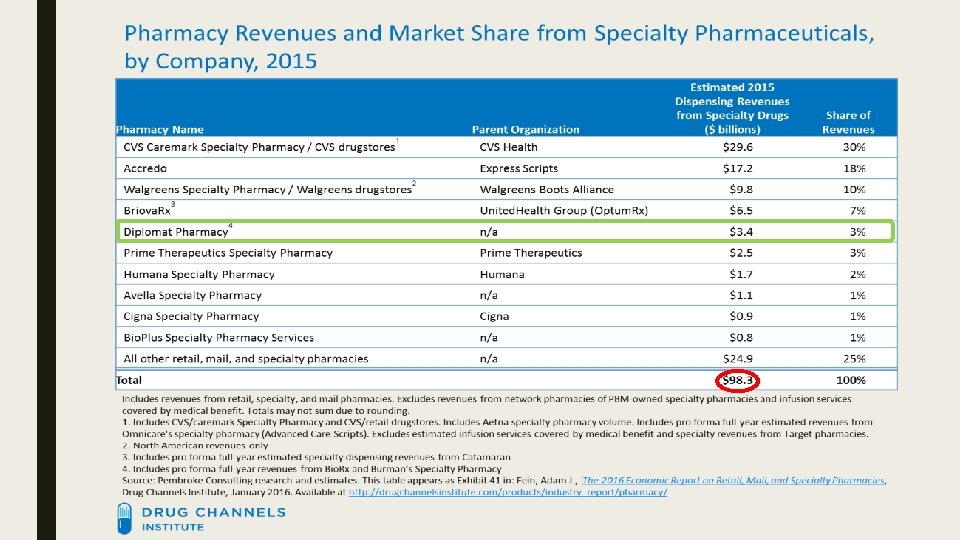
![SPECIALTY PHARMACY MARKET SHARE 2015 [CATEGORY NAME] [PERCENTAGE] Top 3 57% [CATEGORY NAME] [PERCENTAGE] SPECIALTY PHARMACY MARKET SHARE 2015 [CATEGORY NAME] [PERCENTAGE] Top 3 57% [CATEGORY NAME] [PERCENTAGE]](http://slidetodoc.com/presentation_image_h2/a110c7976a85e18baf99bc4e48786951/image-10.jpg)
SPECIALTY PHARMACY MARKET SHARE 2015 [CATEGORY NAME] [PERCENTAGE] Top 3 57% [CATEGORY NAME] [PERCENTAGE] Top 4 64% [CATEGORY NAME] [PERCENTAGE] [CATEGORY [PERCENTAGE] NAME] [PERCENTAGE] [CATEGORY NAME] [PERCENTAGE] Diplomat 3% [CATEGORY NAME] [PERCENTAGE]

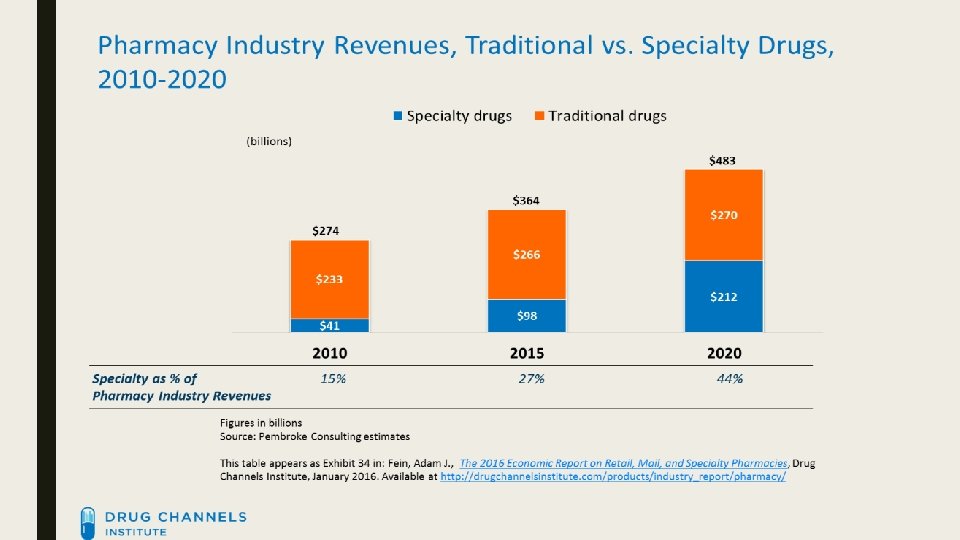
Growth ■ Since 2010, specialty pharmacy estimated revenue has grown from $39. 2 billion to $98. 3 billion (↑ 251%) ■ Diplomat has grown from $600 million to $3. 4 billion (↑ 567%) in the same time frame ■ Report from IMS Health – within the past 10 years, spending has gone from $70 to $425 billion. http: //www. wrbcorp. com/blog/bringing-the-specialty-pharmacy-market-into-the-billions/

Q 2 results for Diplomat ■ Released on 8/9/16 ■ Total revenue was $1. 09 billion (35% increase from last year’s 2 nd quarter) ■ 3% increase in prescriptions dispensed – CEO Phil Hagerman states that this growth is more than it appears. – Moved away from high volume, low revenue compounding at the end of last year. http: //www. drugstorenews. com/article/diplomat-reports-q-results-including--total-revenue-gain? nocache=1

New FDA Drug Approvals - 2015 ■ Most novel new drugs approved since 1992 – 45 new drugs and 9 biologics – 16 of these were considered to be in a new class ■ Of these new approvals, nearly half are considered specialty drugs. – 25 for rare diseases ■ 13 oncology drugs ■ 4 hemophilia drugs ■ 1 for von Willebrand Disease https: //issuu. com/diplomatrx/docs/specialty_drug_approvals_2015_highl_7 a 4 d 31141 f 4 d 2 f? e=15248128/32508071

Hemophilia Drug Approvals - 2015 ■ 4/29/15 – Ixinity (Recombinant Factor IX) – Indicated for Hemophilia B – Other brand names already available: Benef. IX, Alprolix, Rixubis ■ 9/15/15 – Nuwiq (simoctocog alfa) – Indicated for Hemophilia A – First derived from a human cell line – Other brand names: Advate, Recombinate, Xyntha etc. . . https: //issuu. com/diplomatrx/docs/specialty_drug_approvals_2015_highl_7 a 4 d 31141 f 4 d 2 f? e=15248128/32508071

Hemophilia Drug Approvals - 2015 ■ 10/20/15 – Coagadex (Coagulation factor X) – Indicated for factor X deficiency – First and only product indicated for this condition. ■ 11/13/15 – Adynovate (PEGylated Factor VIII) – Indicated for Hemophilia A – Attachment to polyethylene glycol extends the half-life (requires less frequent dosing) ■ 12/8/15 – Vonvendi (recombinant von Willebrand factor) https: //issuu. com/diplomatrx/docs/specialty_drug_approvals_2015_highl_7 a 4 d 31141 f 4 d 2 f? e=15248128/32508071

Hemophilia Drug Approvals - 2016 ■ 3/4/16 – Idelvion (recombinant factor IX linked with albumin fusion protein) – Longer acting factor IX ■ 3/16/16 – Kovaltry (octocog alfa) – Indicated for Hemophilia A – Another option for factor VIII replacement ■ 5/27/16 – Afstyla (recombinant factor VIII single chain) – Longer acting factor VIII https: //www. primetherapeutics. com/en/news/prime-insights/drug-pipeline. html

ACE 910 (emicizumab) ■ Recombinant humanized bispecific antibody ■ Binds to activated factor IX and mimics the action of factor VIII ■ Subcutaneous drug that has a half life of 4 -5 weeks ■ Pharmacokinetic simulation suggested during weekly prophylaxis, peak and trough levels would only differ by ~5%. ■ Out of the 64 patients in dose-escalation study, 2 were positive for anti-ACE 910 antibodies Uchida N, Sambe T, Yoneyama K, et al. Blood. 2016 Mar 31; 127(13): 1633– 1641.

ACE 910 (emicizumab) ■ Results from a study in patients with severe hemophilia A: – 18 Japanese patients with severe hemophilia A enrolled – All received once weekly SQ injection of ACE 910 (12 weeks) – 13/18 patients had no bleeding events ■ 3 of whom had frequent bleeds before trial ■ Less factor replacement was needed to cure a bleeding Shima M, Hanabusa H, Taki M, event et al. N Engl J Med 2016 May 26; 374: 2044 -2053

Questions?
![References ■ Developing Products for Rare Diseases & Conditions [Internet]. Silver Spring, MD. 2016 References ■ Developing Products for Rare Diseases & Conditions [Internet]. Silver Spring, MD. 2016](http://slidetodoc.com/presentation_image_h2/a110c7976a85e18baf99bc4e48786951/image-21.jpg)
References ■ Developing Products for Rare Diseases & Conditions [Internet]. Silver Spring, MD. 2016 [2016 Sep 13]. Available from: http: //www. fda. gov/For. Industry/Developing. Productsfor. Rare. Diseases. Conditions/ucm 2005525. htm. ■ Fein A. Pharmacy Market Share for Specialty Drugs, 2010 [Internet]. Pembroke Consulting. 2011 [2016 Sep 13]. Available from: http: //www. drugchannels. net/2011/12/pharmacy-market-share-for-specialty. html. ■ Fein A. The Top 10 Specialty Pharmacies of 2014 [Internet]. Pembroke Consulting. 2015 [2016 Sep 13]. Available from: http: //www. drugchannels. net/2015/03/the-top-10 -specialty-pharmacies-of-2014. html. ■ Fein A. The Top 10 Specialty Pharmacies of 2015 [Internet]. Pembroke Consulting. 2016 [2016 Sep 13]. Available from: http: //www. drugchannels. net/2016/03/the-top-10 -specialty-pharmacies-of-2015. html. ■ Bringing the Specialty Pharmacy Market into the Billions [Internet]. WRB Communications. 2016 [2016 Sep 13]. Available from: http: //www. wrbcorp. com/blog/bringing-the-specialty-pharmacy-market-into-the-billions/. ■ Fein A. Our 2020 Outlook for Specialty Pharmacy Revenues [Internet]. Pembroke Consulting. 2016 [2016 Sep 13]. Available from: http: //www. drugchannels. net/2016/04/our-2020 -outlook-for-specialty-pharmacy. html. ■ Diplomat reports Q 2 results, including 35% total revenue gain [Internet]. Drug Store News. 2016 [2016 Sep 13]. Available from: www. drugstorenews. com/article/diplomat-reports-q-results-including--total-revenue-gain? nocache=1. ■ Diplomat Publishes New Report on Specialty Drugs Approved in 2015 and Expected for 2016 [Internet]. Flint, MI. PRNewswire. 2016 [2016 Sep 13]. Available from: http: //www. prnewswire. com/news-releases/diplomat-publishes-new-report-on-specialty-drugs-approved-in-2015 -and-expected -for-2016 -300200604. html. ■ Uchida N, Sambe T, Yoneyama K, et al. A first-in-human phase 1 study of ACE 910, a novel factor VIII-mimetic bispecific antibody, in healthy subjects. Blood. 2016 Mar 31; 127(13): 1633– 1641. ■ Shima M, Hanabusa H, Taki M, et al. Factor VIII–Mimetic Function of Humanized Bispecific Antibody in Hemophilia A. N Engl J Med 2016 May 26; 374: 2044 -2053.
- Slides: 21