SPCG15 Johan Stranne MD Ph D FEBU Department

SPCG-15 Johan Stranne MD, Ph. D, FEBU Department of Urology Institute of clinical sciences, University of Gothenburg

3
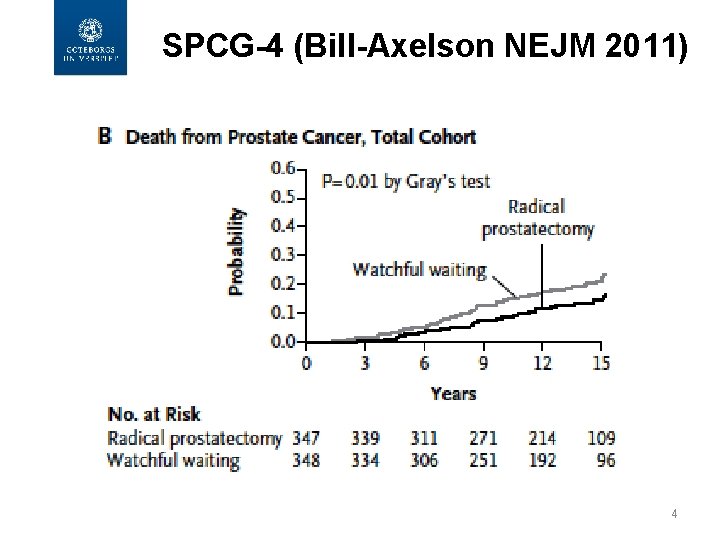
SPCG-4 (Bill-Axelson NEJM 2011) 4
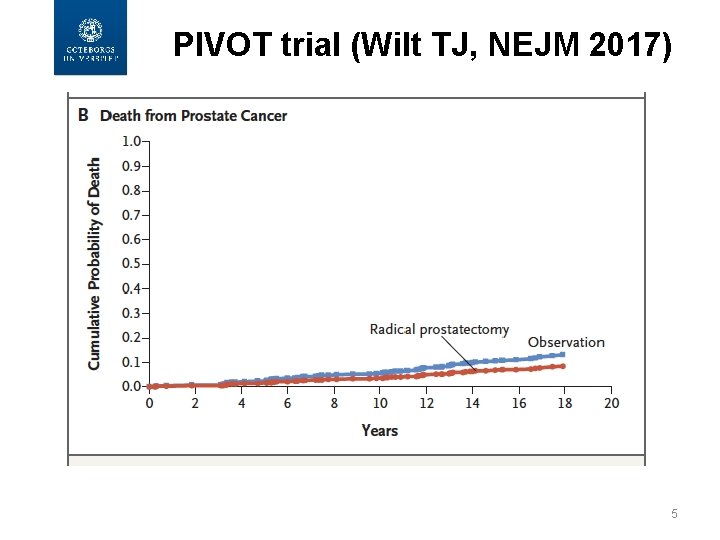
PIVOT trial (Wilt TJ, NEJM 2017) 5
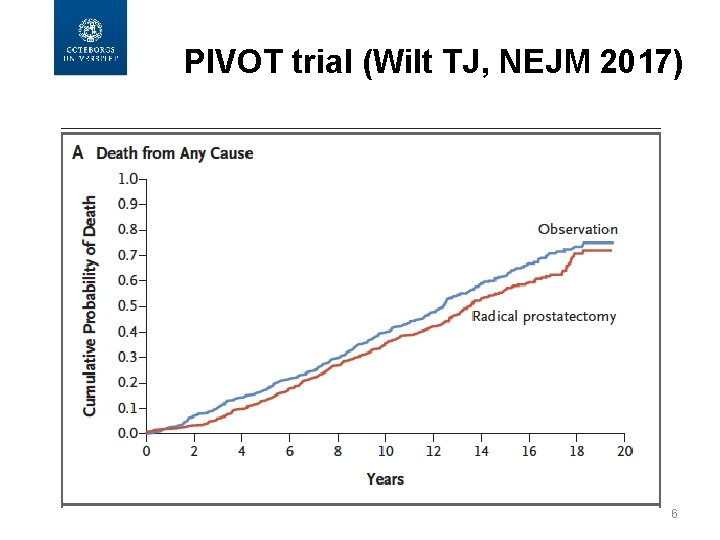
PIVOT trial (Wilt TJ, NEJM 2017) 6
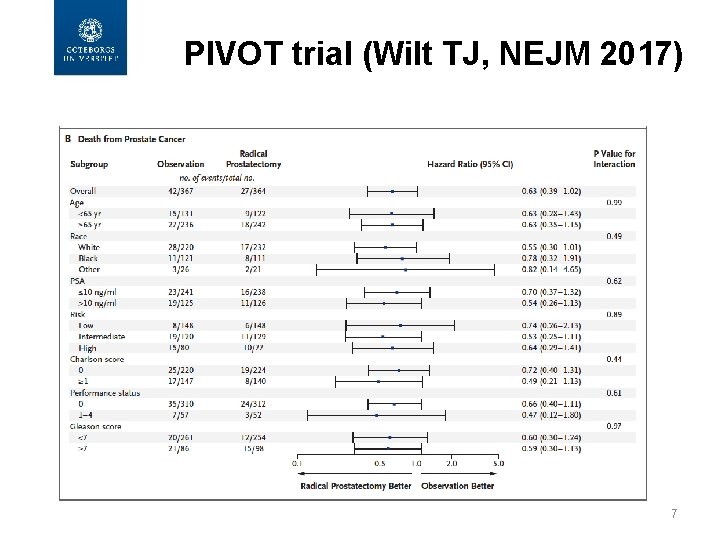
PIVOT trial (Wilt TJ, NEJM 2017) 7
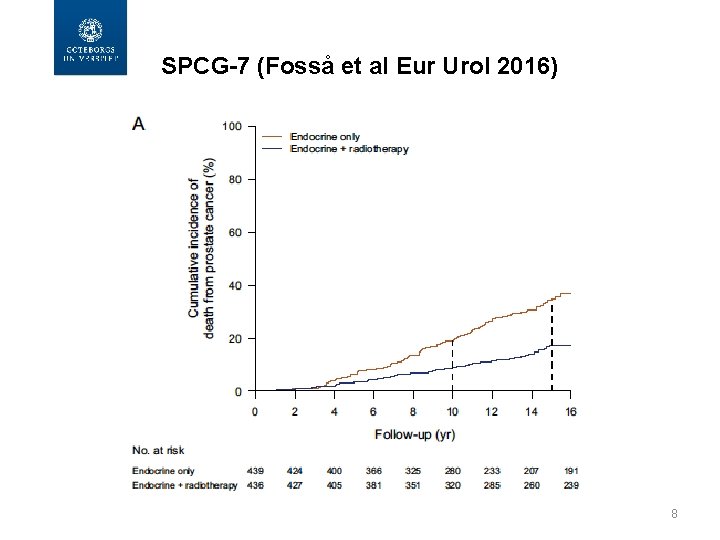
SPCG-7 (Fosså et al Eur Urol 2016) 8
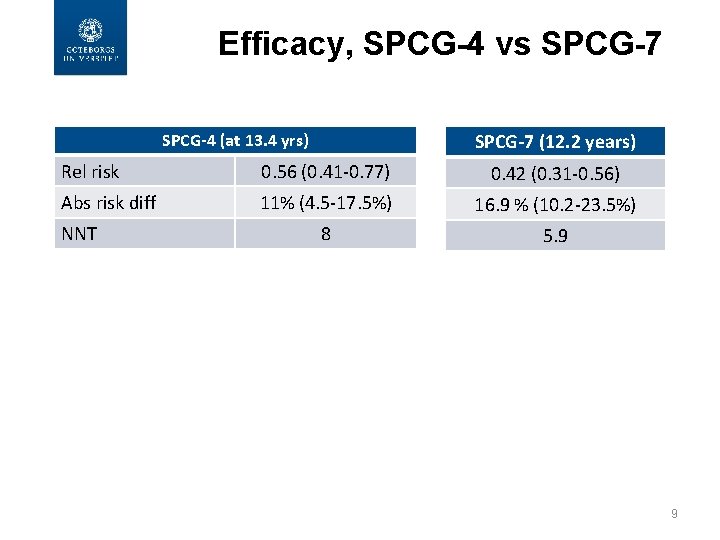
Efficacy, SPCG-4 vs SPCG-7 (12. 2 years) SPCG-4 (at 13. 4 yrs) Rel risk 0. 56 (0. 41 -0. 77) 0. 42 (0. 31 -0. 56) Abs risk diff 11% (4. 5 -17. 5%) 16. 9 % (10. 2 -23. 5%) 8 5. 9 NNT 9
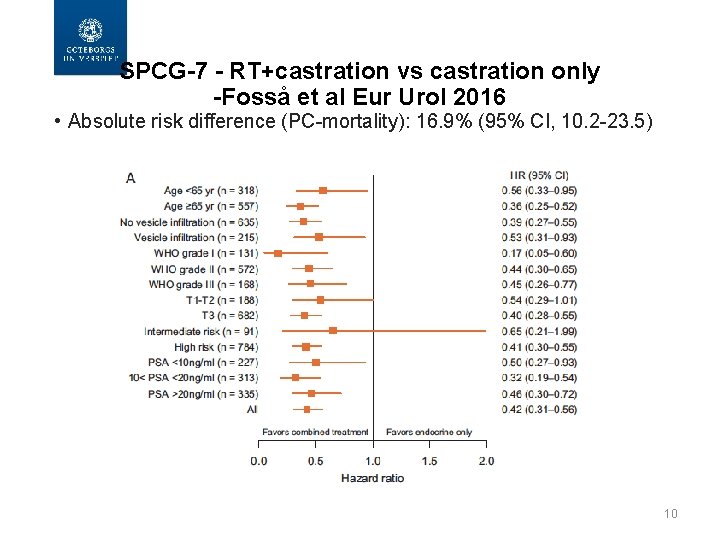
SPCG-7 - RT+castration vs castration only -Fosså et al Eur Urol 2016 • Absolute risk difference (PC-mortality): 16. 9% (95% CI, 10. 2 -23. 5) 10

SPCG-15 Inclusion criteria (n=600+600) • Age ≤ 75 years • T 3 (-T 4) • ISUP ≥ 2 • M 0 • Nodes < 1. 5 cm • PSA < 100 ng/m. L • Eligible for either treatment 11

A: surgical arm • Prostatectomy (without adjuvant or neoadjuvant hormones), center volume >50 per year § Extended PLND (obturator, external and internal iliac, common iliac up to ureteric crossing) § Postoperative RT according to clinical routine 12

B: Radiotherapy arm • Hormonal treatment for 24 months – 3 mo neoadjuvant MAB – 3 mo adjuvant MAB – 18 mo AA (LHRH accepted as alternative) • Radiotherapy – External 2 Gy to 78 Gy, or 2, 5 Gy to 72, 5 Gy (see appendix in protocol) – External + Brachy (2*10 Gy or 1*14, 5 -15 Gy)) 13

Endpoints • Primary: PC-specific survival • (Main) secondary: – Metastasis-free survival – Functional outcome (Qo. L) 14
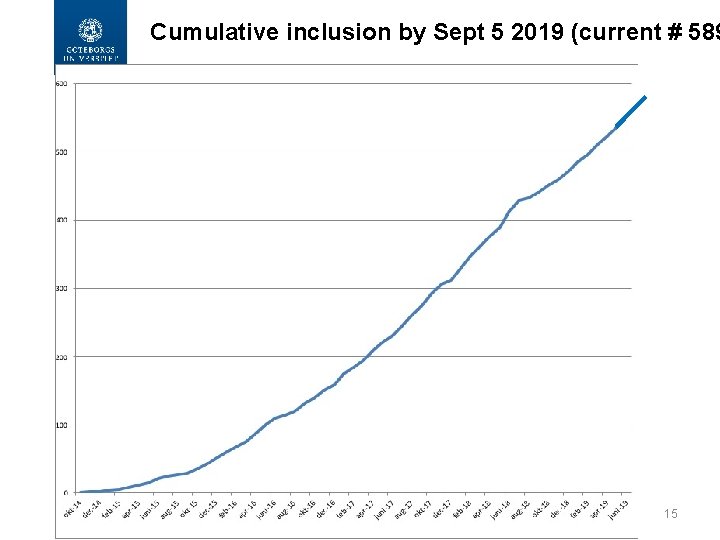
Cumulative inclusion by Sept 5 2019 (current # 589 15
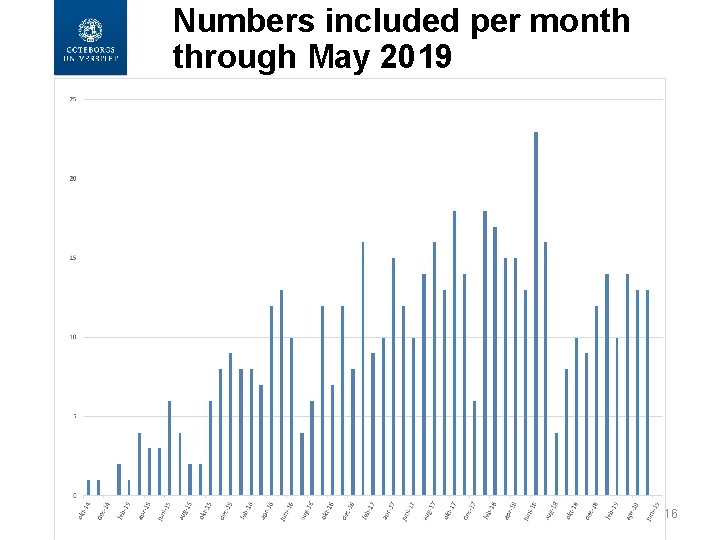
Numbers included per month through May 2019 16
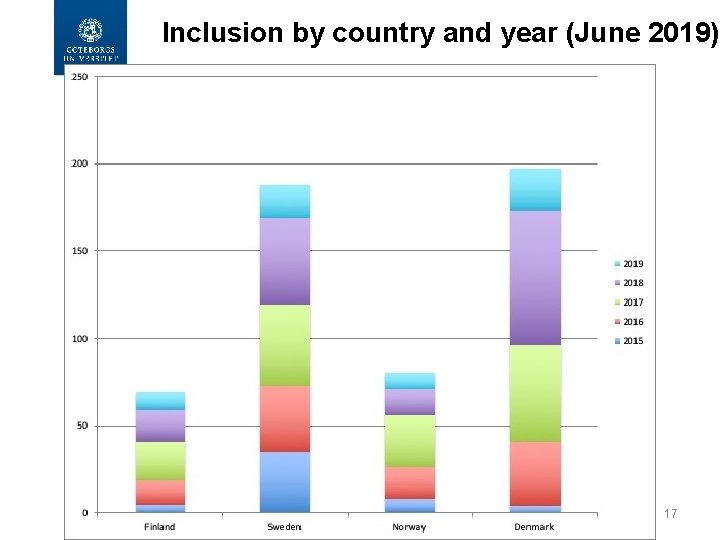
Inclusion by country and year (June 2019) 17
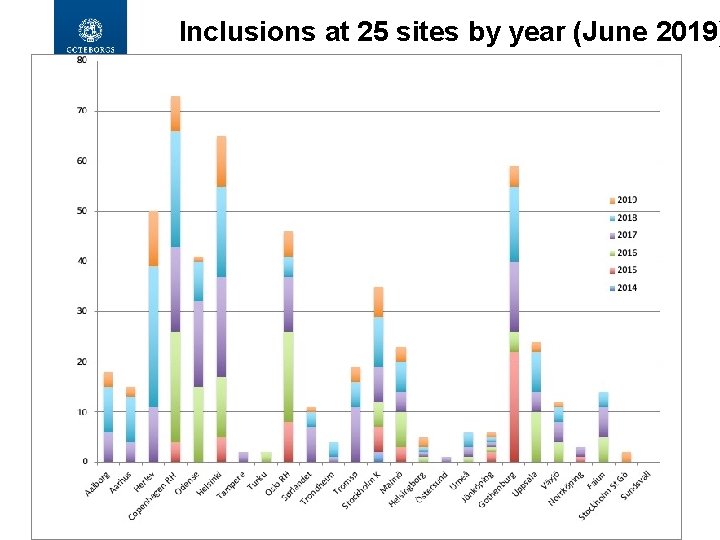
Inclusions at 25 sites by year (June 2019) 18
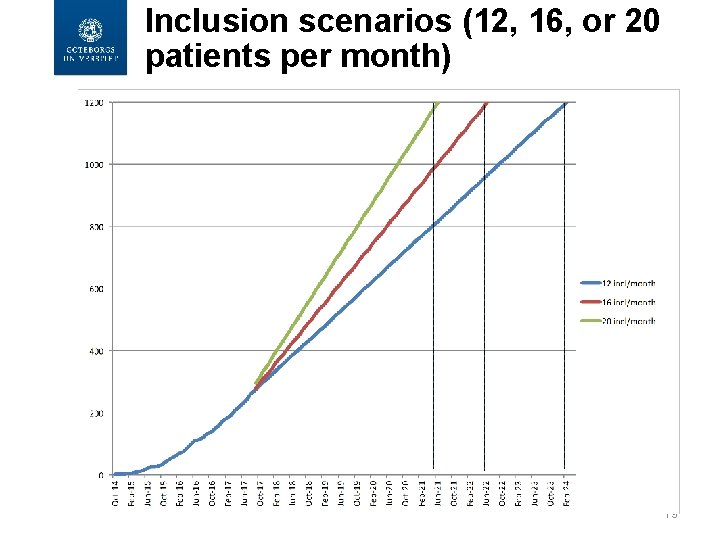
Inclusion scenarios (12, 16, or 20 patients per month) 19

What population is under study? Baseline characteristics of the first 477 randomized subjects 20

Age median (IQR) Surgery 68 (64 -72) Radiotherapy 68 (64 -71) PSA median (IQR) 13 (7, 6 -27) 12 (7, 4 -24) Clinical T stage (all subjects) 6, 9% T 1 10, 2% T 2 82, 5% T 3 0, 4% T 4 7% 11% 81, 7% 0% MR T stage (278 cases) T 1 -2 T 3 T 4 2, 8% 92, 3% 4, 9% 4, 4 91, 9% 3, 7%
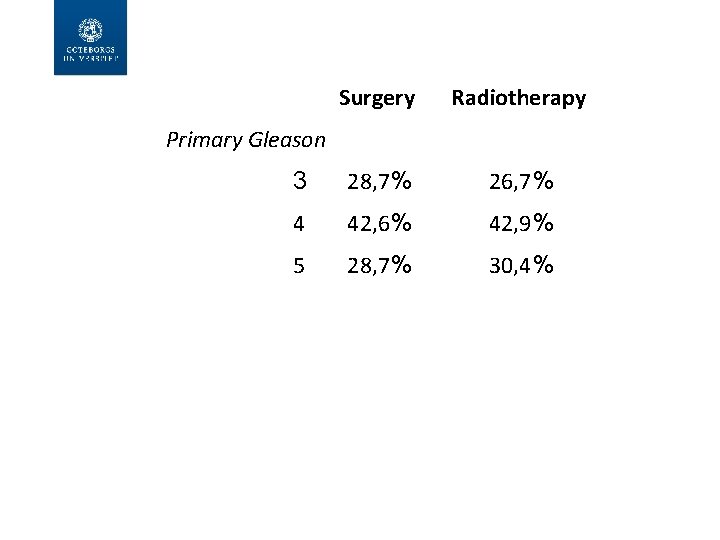
Surgery Radiotherapy 3 28, 7% 26, 7% 4 42, 6% 42, 9% 5 28, 7% 30, 4% Primary Gleason Secondary Gleason 3 30, 0% 4 62, 4% 63, 3% 5 7, 6% 5, 8%

All patients Average prostate volume (ml) 40, 7 Average number of biopsies 10. 4 Average number of biopsies with cancer Average mm cancer 7. 0 54, 6 Average CCI 2. 2 Previous active surveillance 5. 8% Operated patients Postoperative stage p. T 2 p. T 3 a p. T 3 b p. T 4 23% 77% 40% 37% 0. 7%

Organisation • Sweden – Johan Stranne (uro) (dept. Coordinating PI) – Camilla Thellenberg Karlsson (onc) – Eva Johansson (quality-of-life) – Andreas Pettersson (biobanking) – Fredrik Jäderling (radiology) • Norway – Bjørn Brennhovd (uro) – Wolfgang Lilleby (onc) • Finland – Antti Rannikko (uro) – Mauri Kouri (onc) – Tuomas Mirtti (pathol) • Denmark – Klaus Brasso (uro) – Peter Meidahl Petersen (onc) • Coordinating PI: Olof Akre (Swe) • Coordinating project managers: Réka Conrad and Per Henrik Vincent (Swe) 24

- Slides: 24