SOUTHERN BLOTTING Invented by Edwin Southern in the
SOUTHERN BLOTTING

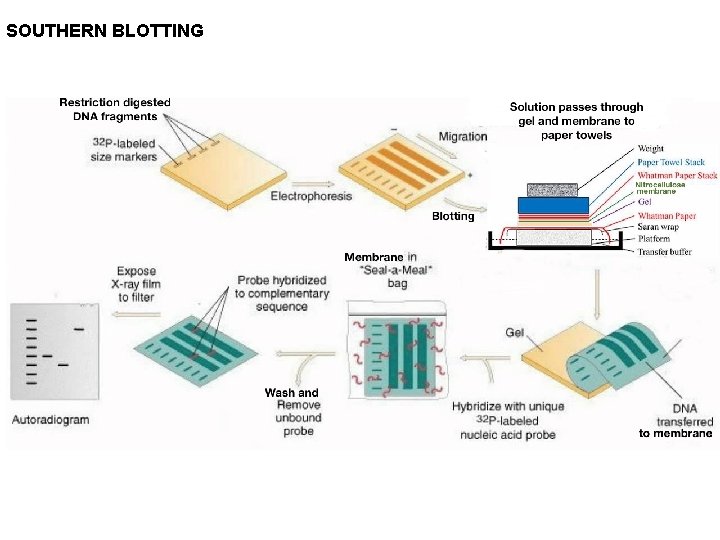
Invented by Edwin Southern in the year 1975, Southern blot is a method used in molecular biology for detection of a specific DNA sequence in DNA samples. The technique combines transfer of electrophoresis-separated DNA fragments to a filter membrane (BLOTTING) and subsequent fragment detection by probe hybridization (on the basis of base complementarity). The details of Southern Blotting are as follows. (A) The mixture of double-stranded DNA fragments generated by restriction endonuclease treatment of DNA is separated according to length by agarose gel electrophoresis. The gel is then exposed to UV for nicking of DNA so as to facilitate denaturation and separation of ds. DNA. This is followed by incubating the gel in denaturation buffer (containing Na. OH) for rendering the ds. DNA in a ss. DNA state. The gel is then renatured for bringing the p. H back to 8. 0 for transfer of DNA to occur.

(B) A sheet of either nitrocellulose paper or nylon paper is laid over the gel, and the separated DNA fragments are transferred to the sheet by blotting. As the transfer buffer is sucked through, it transfers the single stranded fragments from the gel to the surface of the nitrocellulose sheet, where they adhere firmly. This transfer is necessary to keep the DNA firmly in place while the hybridization procedure is carried out. The nitrocellulose sheet is carefully peeled off the gel. The Nitrocellulose membrane is then air dried, incubated in an Oven at 80 o. C for 2 hours, and crosslinked using UV transilluminator for permamnet binding of the ss. DNA to the membrane. The membrane is also treated with 20 M solution of Salmon Sperm DNA for a process called as Blocking in which the membrane gets ds. DNA all over the empty areas for maximum attachment of the probe with the ss. DNA and not to the membrane empty areas. (D) The sheet containing the bound single-stranded DNA fragments is placed in a sealed plastic bag together with buffer containing a radioactively labeled DNA probe specific for the required DNA sequence. The sheet is exposed for a prolonged period to the probe under conditions favoring hybridization. The sheet is removed from the bag and washed thoroughly, so that only probe molecules that have hybridized to the DNA on the paper remain attached. After autoradiography, the DNA that has hybridized to the labeled probe will show up as bands on the autoradiograph.
NORTHERN BLOTTING

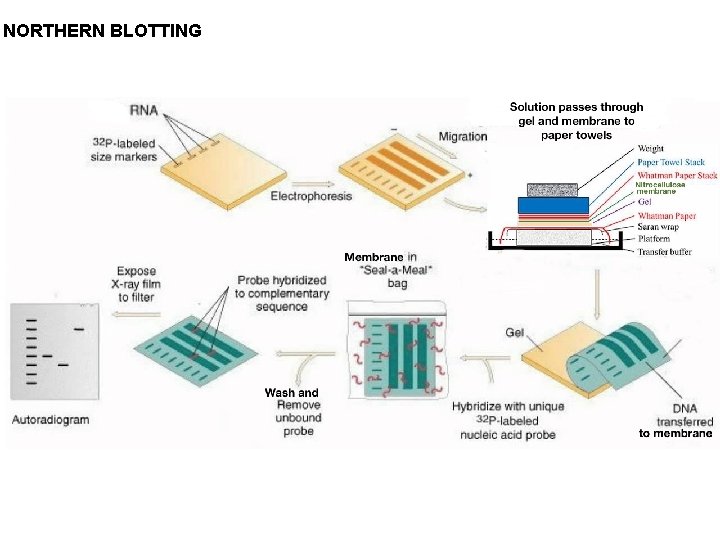
Similar and analogous to Southern Blot analysis, a method in molecular biology which is used to detect RNA transcripts in a sample or across sample usingle stranded DNA or RNA probes is called Northern Blotting. RNA transcripts being single stranded does not require certain steps such as UV-Nicking, Denaturation-Renaturation. However, during the Agarose gel electrophoresis based separation, one has to keep in mind that RNA can fold on its own causing secondary structure formation which may inhibit or obstruct probe binding with the RNA molecule and for that, the AGE used in northern blotting is a denaturing AGE wherein Formalin-Formaldehyde are added to the AGE gel for keeping the RNA linear, unfolded. it is difficult to get bands like profile in a northern blot as the gel and eventually the membrane end up having the RNA as a blob like structure and so the probes normally show binding to form a dense black spot on a autoradiogram. One can assess presenceabsence and to some extent quantification of gene expression can also be done.
WESTERN BLOTTING

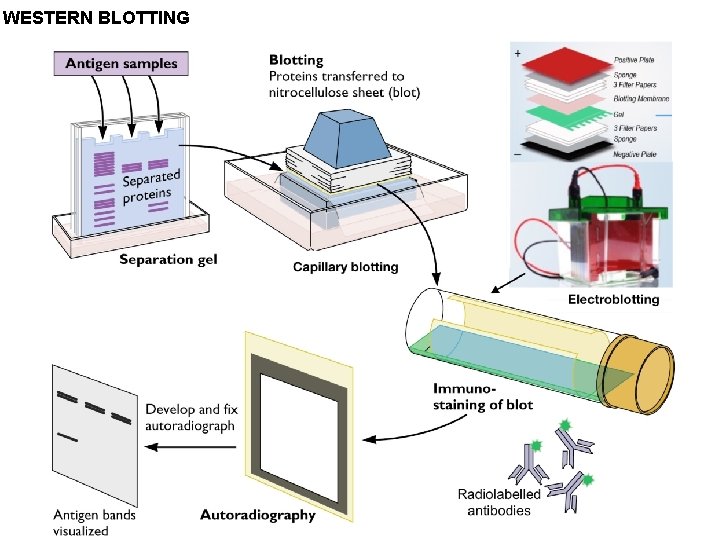
• The western blot or protein immunoblot is a widely used analytical technique in molecular biology, to detect specific proteins in a sample. • Analogous to Southern and Northern Blotting techniques, this blotting method is a Protein-Protein binding and interaction which helps in detecting the absence or presence of a protein of interest. • The term "western blot" was given by W. Neal Burnette, although the method originated in the laboratory of Harry Towbin at the Friedrich Miescher Institute. Western blotting technique comprises of three major steps: Step 1. PAGE separation of protein in a sample: This step is essentially PAGE or SDS PAGE analysis of the protein sample where the sample is fractionated on to a gel matrix on the basis of charge density or molecular mass respectively. Step 2. Transfer of the protein from Acrylamide gel to NC membrane and preparation of the membrane for hybridization: In this step, the Polyacrylamide gel with the protein fractionated on it is properly bound to a NC membrane and the transfer is done under electric field very much like gel electrophoresis. The transfer buffer takes the protein from the gel and blots it on the membrane. This entire blotting has to be done at 4 -15 o. C so that there is no damage to proteins. Once the transfer is over (approximately 30 minutes to 2 hours), the membrane is taken and air dried.

The membrane is blocked with Bovine Serum Albumin or with Milk Powder solution for increasing the probe binding efficiency so that there is no empty area on the membrane (this is called Blocking) Step 3. Hybridization of the Primary and Secondary Antibodies to the protein sample and visualization: For the purpose of hybridization, primary and secondary antibodies are required. Primary antibodies are specific to the protein of interest and are developed in a host animal e. g Rabbit or Mouse. These Primary antibodies are able to bind to the protein (of interest) on the gel. For the purpose of visualization, another type of antibody called secondary antibody are used which are developed against the host animal system from which the primary was developed and thus secondary are reffered to as anti-goat or anti-rabbit and so on. The secondary antibody is tagged with a flurorecsent or chemiluminiscent or enzyme system which can develop a chromatic or light reaction e. g. biotin or a reporter enzyme such as Alkaline phosphatase or Horseradish peroxidase. The NC membrane is treated with primary antibody, washed for removing extra primary antibody and then treated with secondary, again subject to proper washing and then the membrane is subjected to a treatment as per the chemistry of the Probe tagged with secondary antibody. In this manner, the Membrane is developed to show the presence and position of the desired protein on the gel.
- Slides: 8