Sources of Hydrocarbons Hydrocarbons l Hydrocarbons are compounds

Sources of Hydrocarbons

Hydrocarbons l. Hydrocarbons are compounds consisting of hydrogen and carbon only, bonded together covalently.

Sources Fossil Fuels Crude Oil Natural gas Coal

l Crude oil and natural gas were formed from the remains of marine plants and animals. l Coal was produced from dead wood
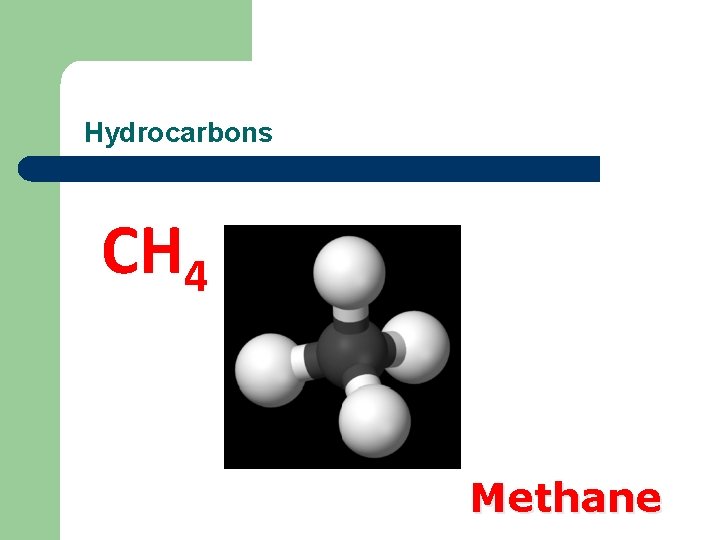
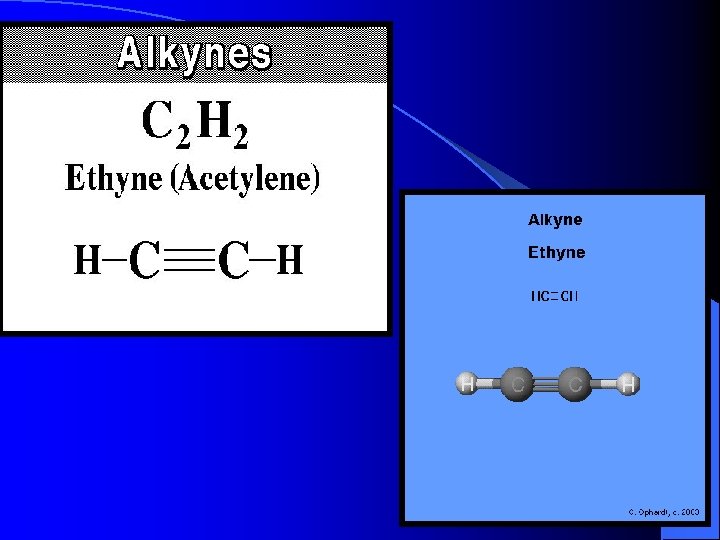
Hydrocarbons CH 4 Methane

Methane Production Methane can be produced in: § Coalmines § Slurry pits § Waste dumps § Digestive tracts of animals

Hazards of Methane Production l l l Fires and explosions Death by suffocation Contributes to the greenhouse effect

When the chain is between 5 and 9 carbons, the hydrocarbon is gasoline. About a dozen carbons and it is diesel. Around 20 carbons is motor oil. A chain of hundreds to thousands of carbon and hydrogens make plastic. This particular plastic is polyethylene.
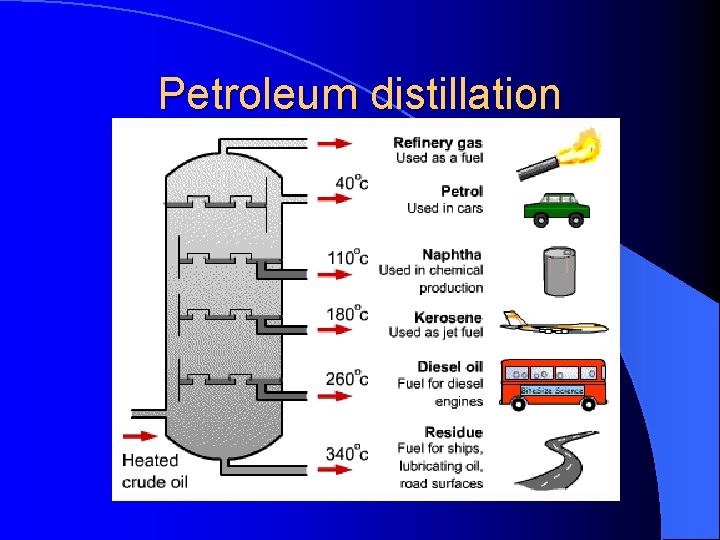
Petroleum distillation



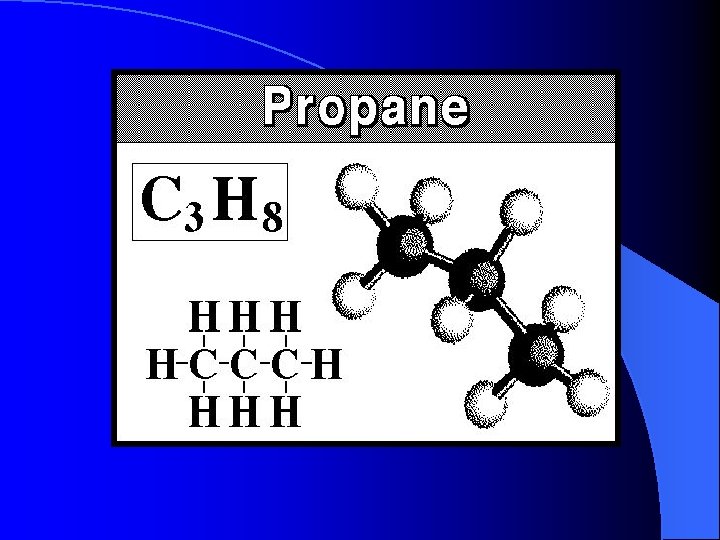

Carbon groups • • Alkanes Methane Ethane Propane Butane Pentane Hexane Cn. H 2 n+2 CH 4 GAS C 2 H 6 GAS C 3 H 8 GAS C 4 H 10 GAS C 5 H 12 LIQUID C 6 H 14 LIQUID


Naming carbon groups • • • Methyl Ethyl Propyl Butyl Pentyl CH 3 C 2 H 5 C 3 H 7 C 4 H 9 C 5 H 11
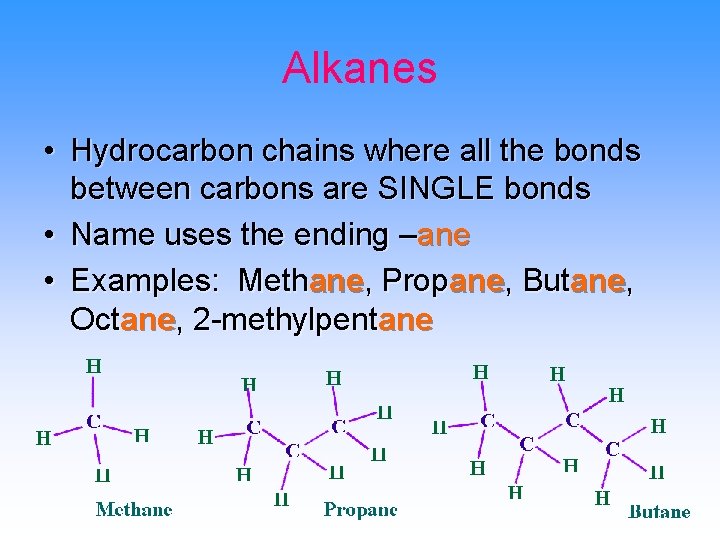
Alkanes • Hydrocarbon chains where all the bonds between carbons are SINGLE bonds • Name uses the ending –ane • Examples: Methane, Propane, Butane, Octane, 2 -methylpentane

Step 1. Find the parent chain. • Where is the longest continuous chain of carbons?

Step 2. Number the parent chain. • Number the parent chain so that the attached groups are on the lowest numbers Methyl is on carbon #2 of the parent chain Methyl is on carbon #4 of the parent chain 1 5 1 8 2 4 3 3 4 2 3 6 7 2 8 1 4 5 5 4 6 3 5 1 GREEN is the right way for this one! 27 1 7 2 6 3 5 4 4 Groups on 2 and 5 Groups on 4, 6, and 7 Groups on 2, 3, and 5 5 3 6 72 1 Groups on 3 and 6

Step 3. Name the attached groups. • Carbon (alkyl) groups – Methyl CH 3 – Ethyl CH 3 CH 2– Propyl CH 3 CH 2 – • Halogens – Fluoro (F-) – Chloro (Cl-) – Bromo (Br-) – Iodo (I-)

Step 4. Designate where the group is attached to the parent chain. • Use the numbers of the parent chain from step 2 to designate the location of the attached groups to the parent chain. 2 -methyl 1 2 3 4 5
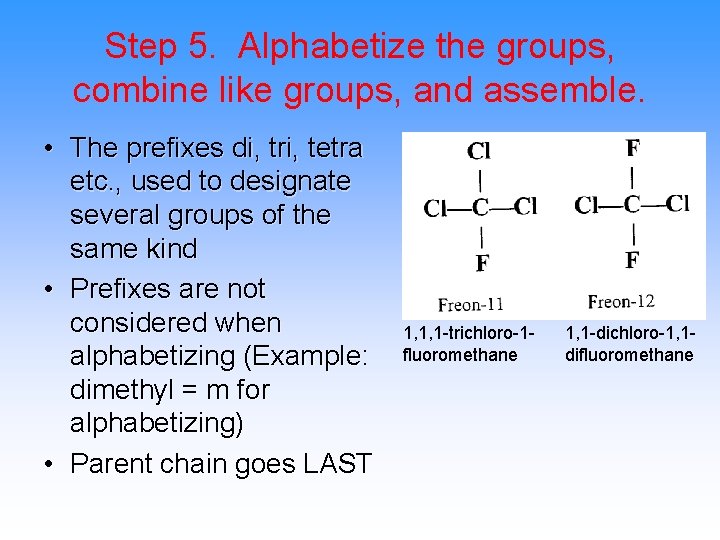
Step 5. Alphabetize the groups, combine like groups, and assemble. • The prefixes di, tri, tetra etc. , used to designate several groups of the same kind • Prefixes are not considered when alphabetizing (Example: dimethyl = m for alphabetizing) • Parent chain goes LAST 1, 1, 1 -trichloro-1 fluoromethane 1, 1 -dichloro-1, 1 difluoromethane

Draw Some Simple Alkanes • 2 -methylpentane • 3 -ethylhexane • 2, 2 -dimethylbutane • 2, 3 -dimethylbutane



Alkenes • • • Cn. H 2 n-2 Ethene Propene Butene Pentene Hexene
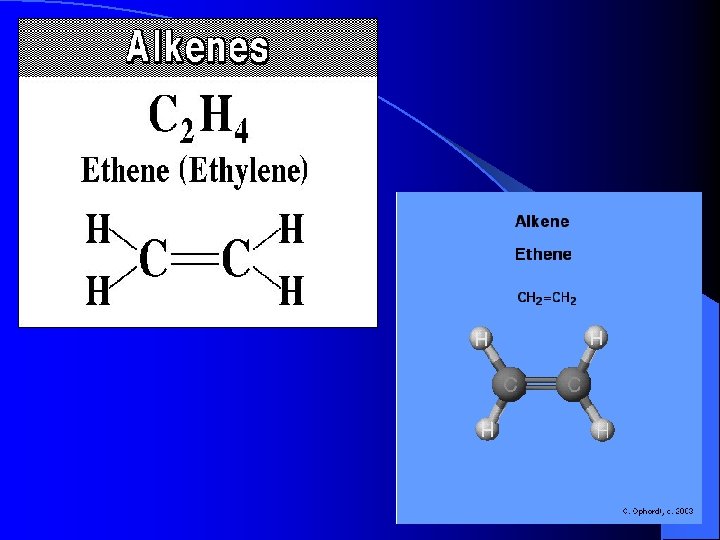


Alkenes



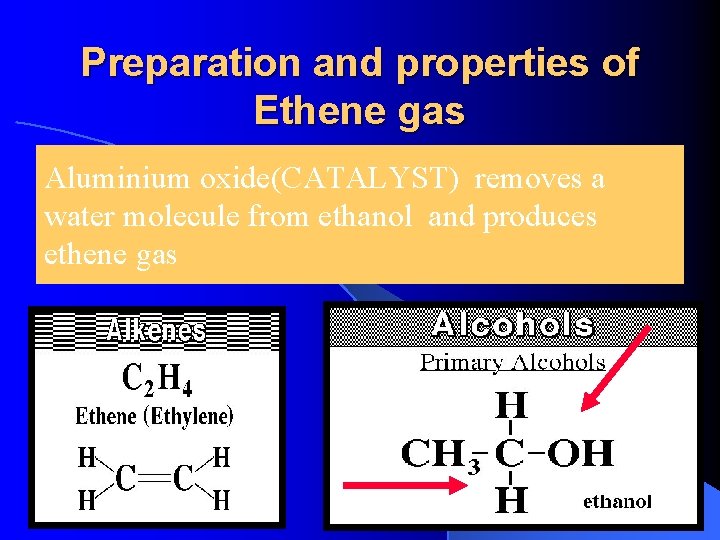
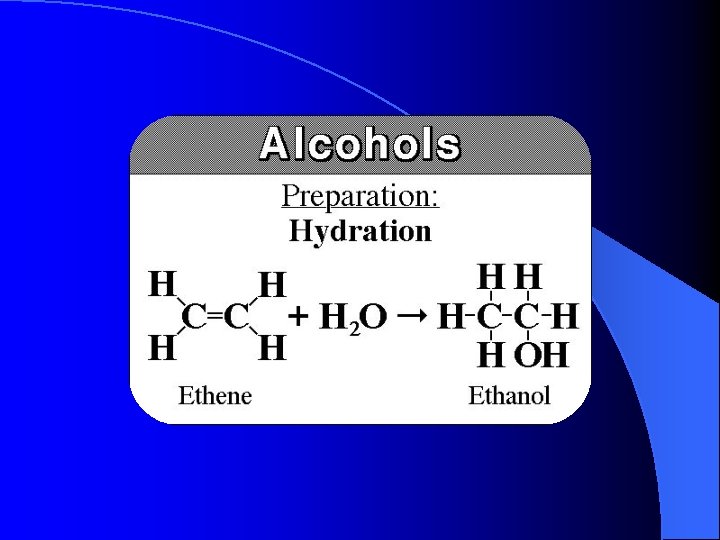
Preparation and properties of Ethene gas Aluminium oxide(CATALYST) removes a water molecule from ethanol and produces ethene gas
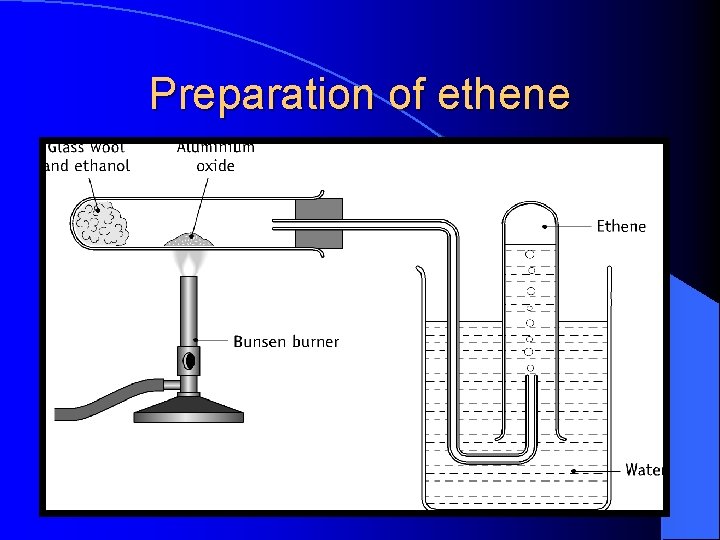
Preparation of ethene

Practical precautions of ethene • Avoid suck back by removing heat before dismantling the equipment • Discard first gas jar as it will have a mix of air and ethene

Precautions • • • Why is it desirable to push the glass wool into the tube after the ethanol has been added? To ensure that all of the ethanol is soaked up. Why should the ethanol not be heated strongly? Strong heating of the ethanol will cause it to evaporate too quickly and escape from the tube before it can be dehydrated.

PHYSICAL PROPERTIES: It is a colourless gas with a sweetish smell. It is insoluble in water so you can collect it over water. COMBUSTION C₂H₄ + 3 O₂ → 2 CO₂ + 2 H₂O LIME WATER TEST



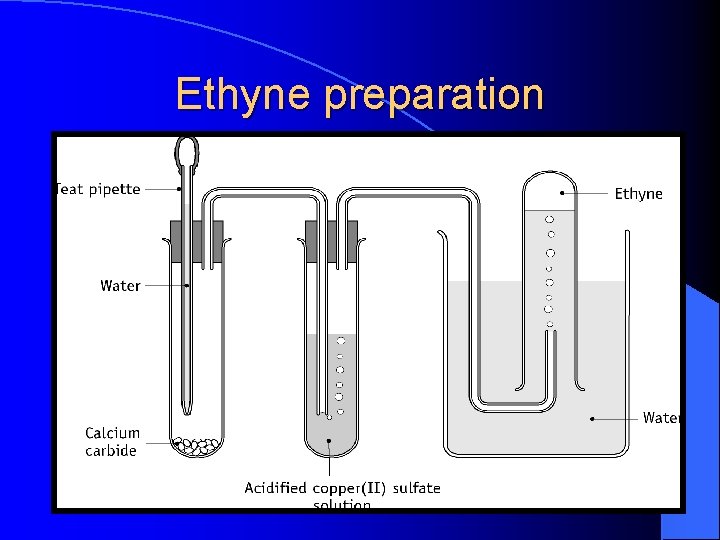
Ethyne preparation
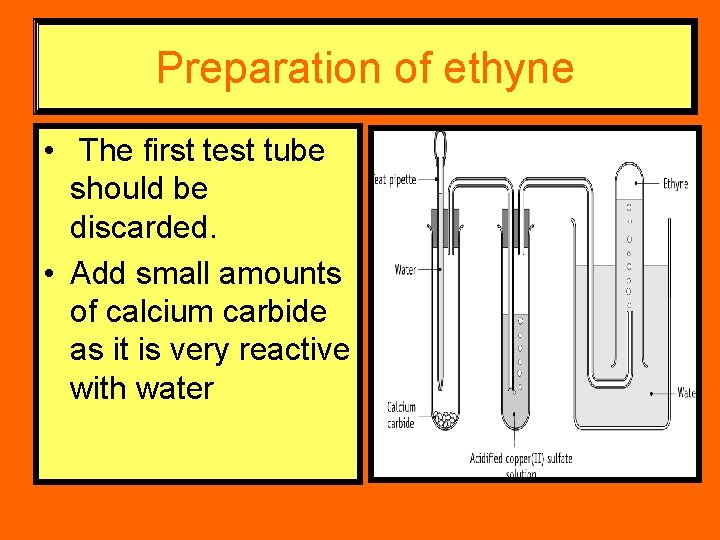
Preparation of ethyne • The first test tube should be discarded. • Add small amounts of calcium carbide as it is very reactive with water

























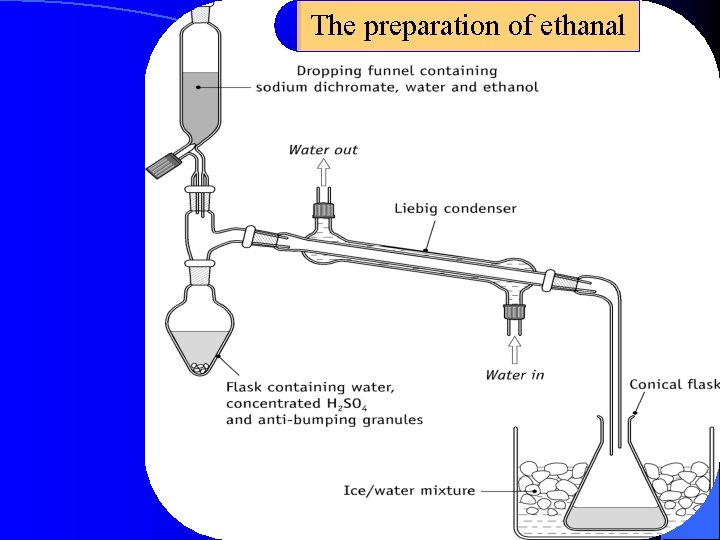
The preparation of ethanal
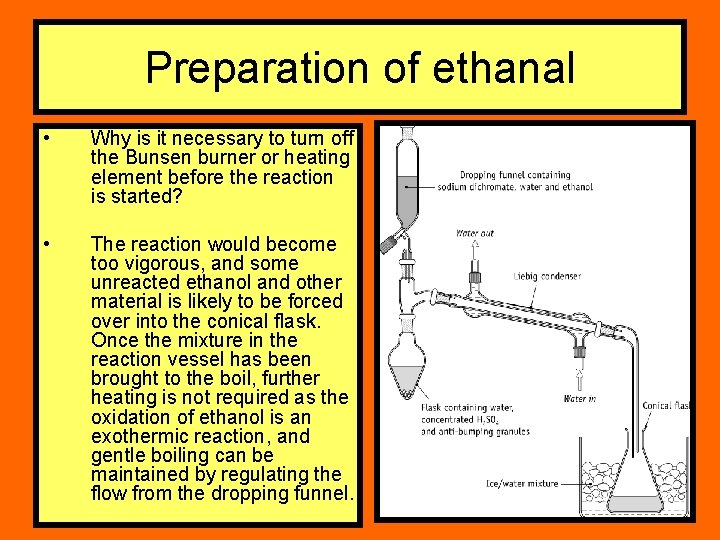
Preparation of ethanal • Why is it necessary to turn off the Bunsen burner or heating element before the reaction is started? • The reaction would become too vigorous, and some unreacted ethanol and other material is likely to be forced over into the conical flask. Once the mixture in the reaction vessel has been brought to the boil, further heating is not required as the oxidation of ethanol is an exothermic reaction, and gentle boiling can be maintained by regulating the flow from the dropping funnel.

TOLLENS REAGENT TEST FOR ALDEHYDES

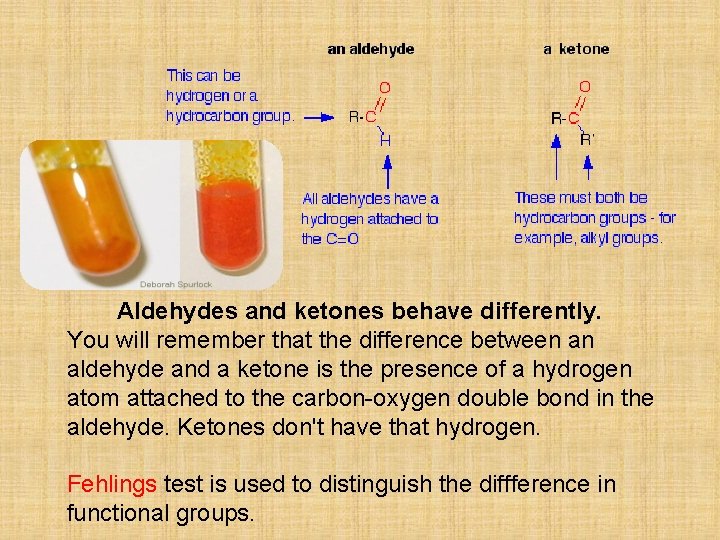
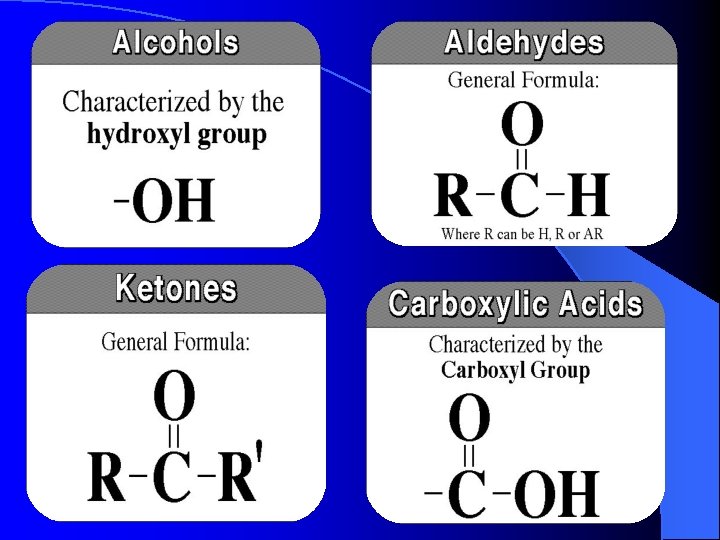
Aldehydes and ketones behave differently. You will remember that the difference between an aldehyde and a ketone is the presence of a hydrogen atom attached to the carbon-oxygen double bond in the aldehyde. Ketones don't have that hydrogen. Fehlings test is used to distinguish the diffference in functional groups.











Preparation of Ethanoic acid Reflux (20 mins) Mix 2 cm 3 of ethanol and 10 cm 3 of deionised water in the dropping funnel. 5 g of sodium dichromate 5 cm 3 of sulphuric acid Cool flask with tap water

Preparation of ethanoic acid 1. Mix 2 cm 3 of ethanol and 10 cm 3 of deionised water in the dropping funnel. 2. Add the solution from the dropping funnel dropwise down the condenser, while swirling the contents of the flask and cooling it if necessary to prevent too vigorous a reaction.
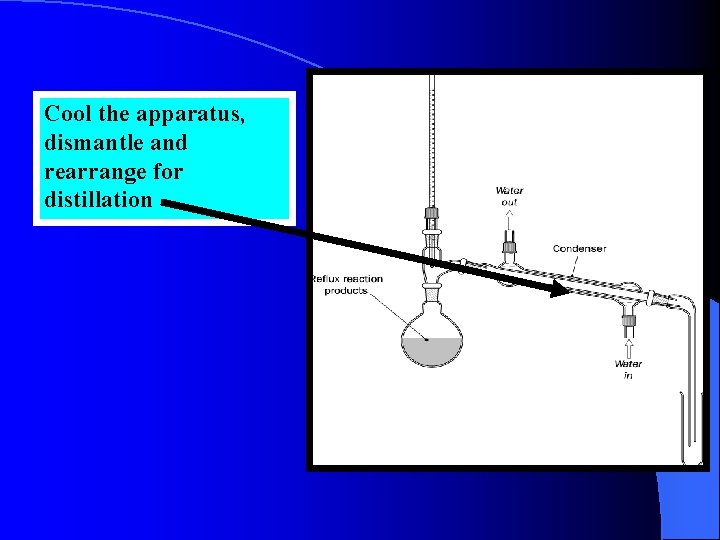
Cool the apparatus, dismantle and rearrange for distillation
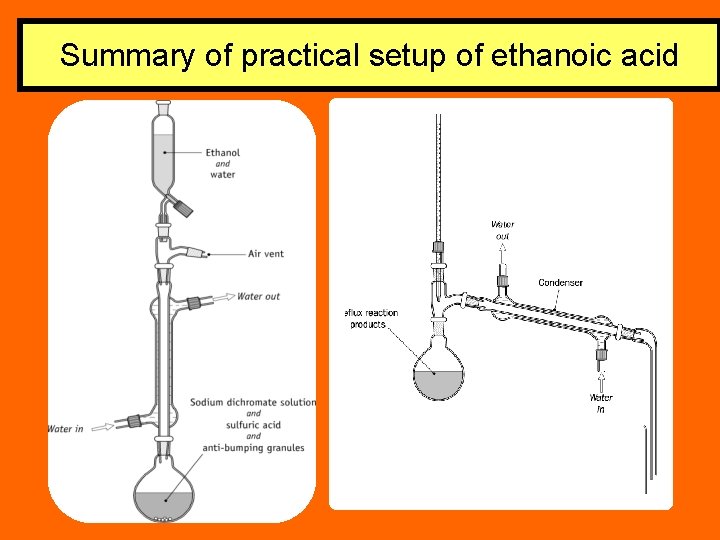
Summary of practical setup of ethanoic acid

Precautions • Reflux distillation involves condensing the vapour from a boiling liquid in such a way as to return the condensed material to the reaction vessel. • In this way a reaction may be carried out at quite a high temperature while preventing the loss of any of the reactants or products.



- Slides: 94