Some important terms Elements Chemical symbol Mixtures Heterogenous


Some important terms: � Elements �Chemical symbol � Mixtures �Heterogenous �Homogenous � Kinetic �Atoms � Compounds � Chemical theory � Diffusion � States of matter/symbols Properties/Changes �Melting/freezing �Chemical �Evaporation/condensation �Reactants �Products equation �Sublimation/deposition

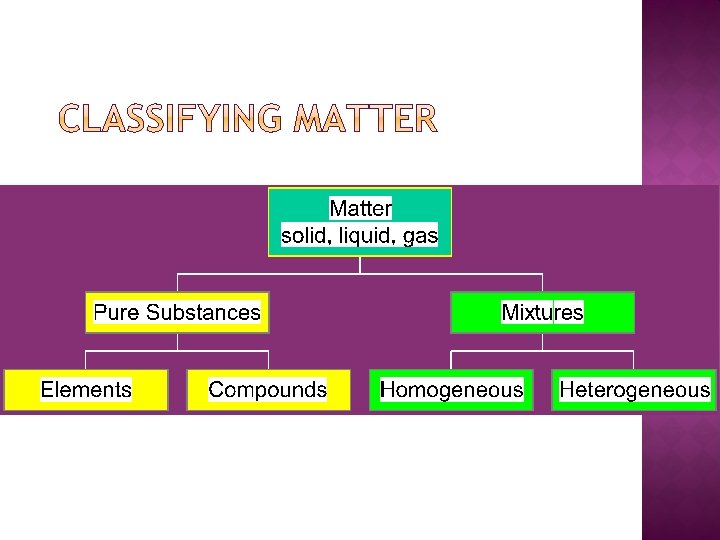
� Elements: a single atom or diatomic molecule that cannot be broken down by chemical means � Exs (chemical symbols): Ar Ga H 2 Cl 2 Li � Compounds: 2 or more elements chemically combined � Exs: H 2 O Ca. Cl 2 BF 3 CH 3 OH NH 3 � Mixtures: 2 or more compounds physically combined �Heterogenous: non-uniform composition throughout �Exs: salsa salad milk wood oil/water �Homogenous uniform composition throughout �Exs: kool-aid salt water air tea water/alcohol

� Pure substances: each sample of that substance has the same properties or characteristics as every other sample � Mixtures: made up of several substances, each which retains their individual properties

� Elements: a single atom or diatomic molecule that cannot be broken down by chemical means � Exs: Ar Ga H 2 Cl 2 Li V � Compounds: 2 or more elements chemically combined � Exs: H 2 O Ca. Cl 2 BF 3 CH 3 OH NH 3 � Mixtures: 2 or more compounds physically combined �Heterogenous: non-uniform composition throughout �Exs: salsa salad milk wood oil/water �Homogenous uniform composition throughout �Exs: kool-aid salt water air tea water/alcohol

� Elements: a single atom or diatomic molecule that cannot be broken down by chemical means � Exs: Ar Ga H 2 Cl 2 Li V � Compounds: 2 or more elements chemically combined � Exs: H 2 O Ca. Cl 2 BF 3 CH 3 OH NH 3 � Mixtures: 2 or more compounds physically combined �Heterogenous: non-uniform composition throughout �Exs: salsa salad milk wood oil/water �Homogenous uniform composition throughout �Exs: kool-aid salt water air tea water/alcohol

Compounds • Elements lose their chemical characteristics Mixtures • Parts retain their properties • can be separated by physical means chemical means • composition may vary • same composition by mass for a compound
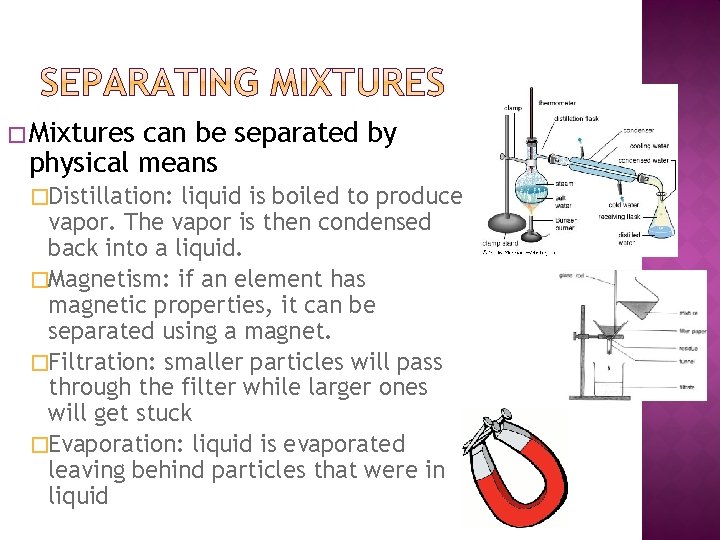
� Mixtures can be separated by physical means �Distillation: liquid is boiled to produce vapor. The vapor is then condensed back into a liquid. �Magnetism: if an element has magnetic properties, it can be separated using a magnet. �Filtration: smaller particles will pass through the filter while larger ones will get stuck �Evaporation: liquid is evaporated leaving behind particles that were in liquid

� Water? � Answer: compound � Air? � Answer: mixture � Salt? � Answer: compound � Carbon dioxide? � Answer: compound

� Air � Answer: Homogeneous mixture � Granite � Answer: Heterogeneous mixture � Sand water � Answer: Heterogeneous mixture

� Chemical equation: � Ca + O 2 Ca. O � Balanced chemical equation: � 2 Ca + O 2 2 Ca. O � Chemical equation with state symbols: � 2 Ca(s) + O 2(g) 2 Ca. O(s) �State symbols: �Gas = (g) �Liquid = (l) �Solid = (s) �Aqueous (in solution with water) = (aq)

� States of matter are determined by the amount of kinetic energy the particles have �Temperature �Pressure � Therefore, KT states: the average kinetic energy of the particles is directly related to the temp of the system (and thus affects P as well) � Inter-particle forces = Strength of forces between particles � KE=1/2 mv 2 (the larger the mass, the slower it flows)

- Slides: 14