Some Basic Chemistry Single and Double Bonds C
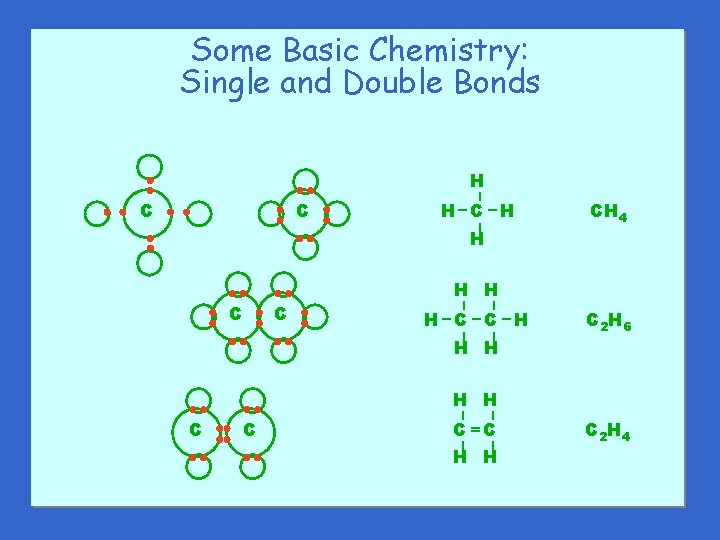
Some Basic Chemistry: Single and Double Bonds C H–C –H – C – H H – – – C H H – C CH 4 H–C – C –H H H C 2 H 6 – – C – H H C–C H H C 2 H 4
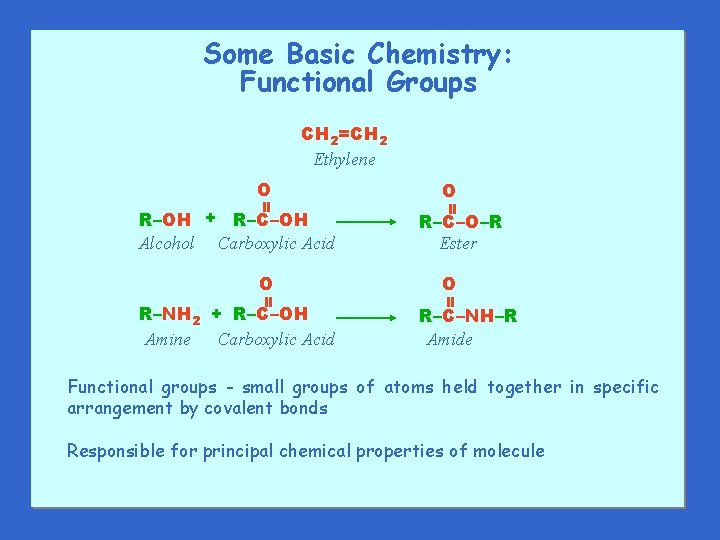
Some Basic Chemistry: Functional Groups CH 2=CH 2 Ethylene R–OH + R–C–OH Alcohol Carboxylic Acid R–NH 2 + R–C–OH Amine Carboxylic Acid R–C–O–R Ester O = = O R–C–NH–R Amide Functional groups - small groups of atoms held together in specific arrangement by covalent bonds Responsible for principal chemical properties of molecule
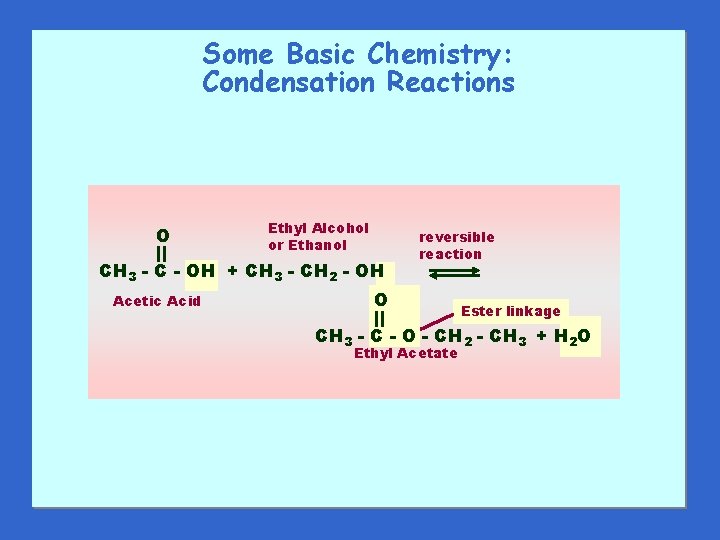
Some Basic Chemistry: Condensation Reactions O Ethyl Alcohol or Ethanol CH 3 - C - OH + CH 3 - CH 2 - OH Acetic Acid reversible reaction O Ester linkage CH 3 - C - O - CH 2 - CH 3 + H 2 O Ethyl Acetate
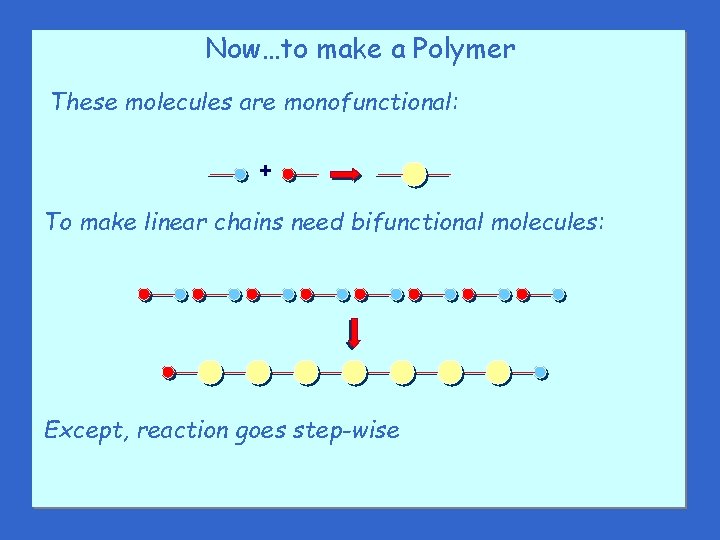
Now…to make a Polymer These molecules are monofunctional: + To make linear chains need bifunctional molecules: Except, reaction goes step-wise
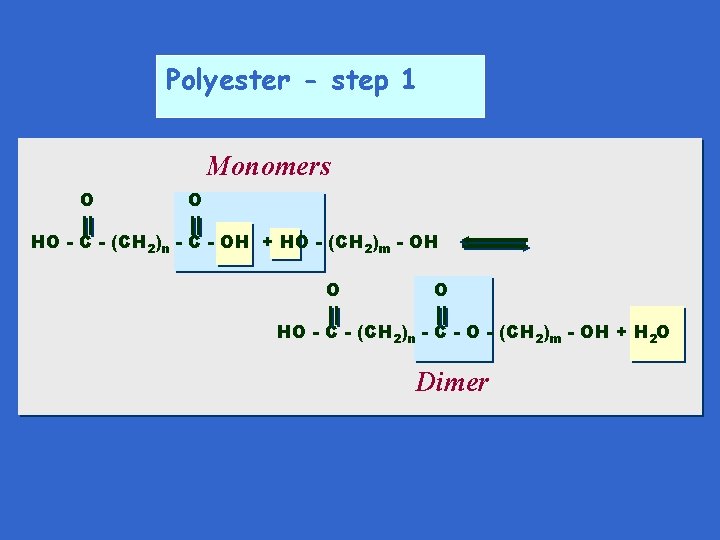
Polyester - step 1 Monomers O O HO - C - (CH 2)n - C - OH + HO - (CH 2)m - OH O O HO - C - (CH 2)n - C - O - (CH 2)m - OH + H 2 O Dimer
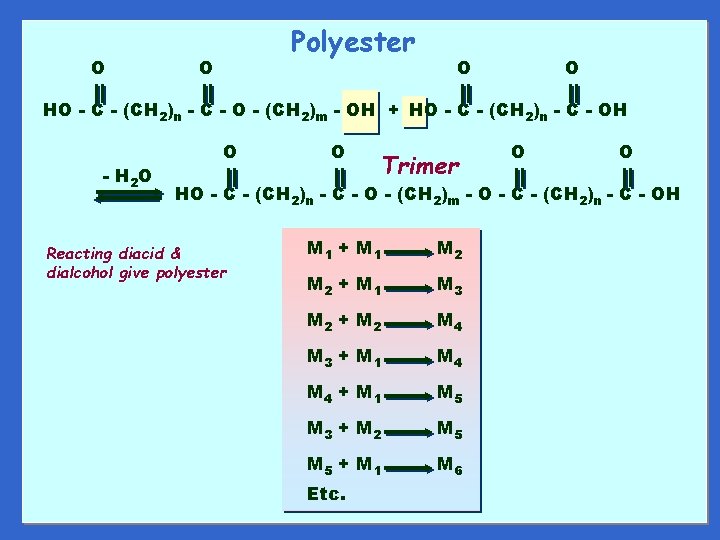
O Polyester O O O HO - C - (CH 2)n - C - O - (CH 2)m - OH + HO - C - (CH 2)n - C - OH - H 2 O O O Trimer O O HO - C - (CH 2)n - C - O - (CH 2)m - O - C - (CH 2)n - C - OH Reacting diacid & dialcohol give polyester M 1 + M 1 M 2 + M 1 M 3 M 2 + M 2 M 4 M 3 + M 1 M 4 + M 1 M 5 M 3 + M 2 M 5 + M 1 M 6 Etc.
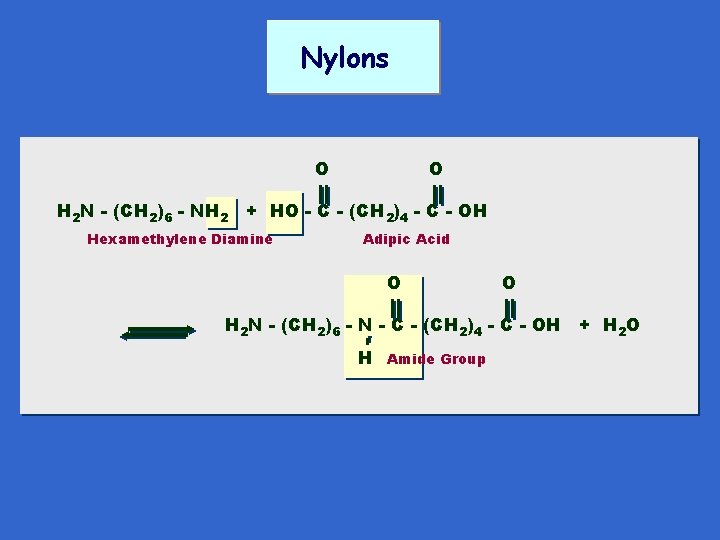
Nylons O O H 2 N - (CH 2)6 - NH 2 + HO - C - (CH 2)4 - C - OH Hexamethylene Diamine Adipic Acid O O H 2 N - (CH 2)6 - N - C - (CH 2)4 - C - OH + H 2 O Amide Group H

O - N - (CH 2)6 - N - C - (CH 2)4 - C H “I am making the announcement of a brand new chemical textile fiber ---derivable from coal, air and water -- and characterized by extreme toughness and strength --” Charles Stine V. P. for research, Du Pont, 1938 6 H Nylon 6, 6 O 6 n

Nylon “I am making the announcement of a brand new chemical textile fiber ---derivable from coal, air and water -- and characterized by extreme toughness and strength --” Charles Stine V. P. for research, Du Pont, 1938

Nylon May 15 1940 - “Nylon Day” Four million pairs go on sale throughout US Supply exhausted in 4 days.

Nylon Parachute WWII

Post WWII stocking sale, San Francisco.
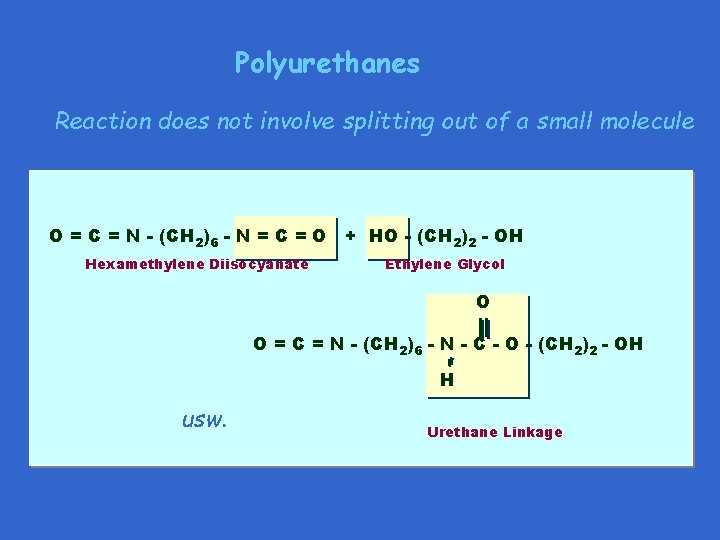
Polyurethanes Reaction does not involve splitting out of a small molecule O = C = N - (CH 2)6 - N = C = O + HO - (CH 2)2 - OH Hexamethylene Diisocyanate Ethylene Glycol O O = C = N - (CH 2)6 - N - C - O - (CH 2)2 - OH H usw. Urethane Linkage

Linear & branched polymers ex: polyethylene Linear Branched - short branches
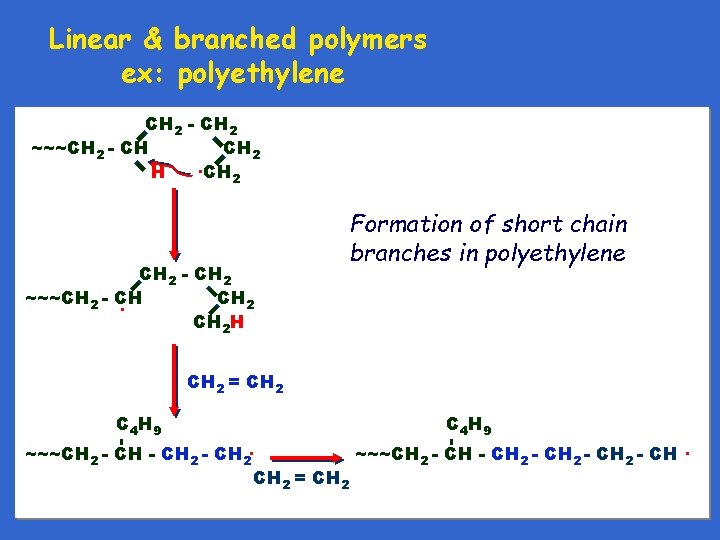
Linear & branched polymers ex: polyethylene CH 2 - CH 2 ~~~CH 2 - CH CH 2. CH H 2 CH 2 - CH 2 ~~~CH 2 - CH CH 2 H Formation of short chain branches in polyethylene CH 2 = CH 2 C 4 H 9 - - C 4 H 9 ~~~CH 2 - CH 2 - CH 2 = CH 2

Low density polyethylene (LDPE) (short branches)

Other types of branching short long star Branching suppresses or prevents chain movement & "crystallization" in polymers
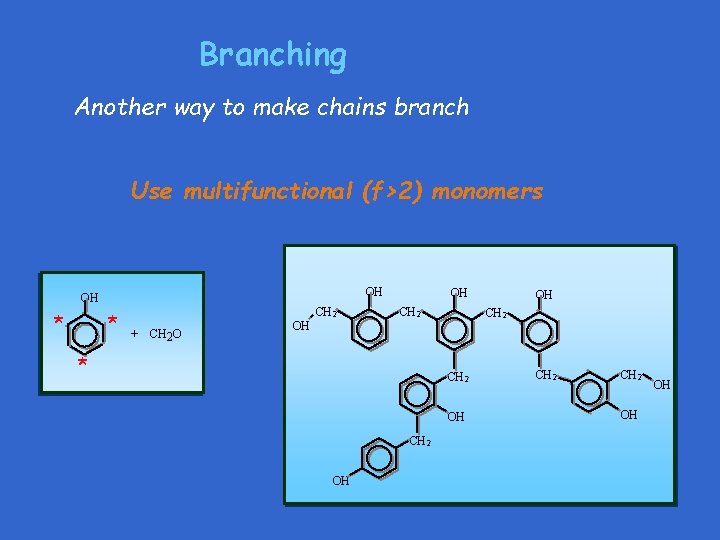
Branching Another way to make chains branch Use multifunctional (f>2) monomers OH OH * * CH 2 + CH 2 O OH CH 2 OH * CH 2 OH OH
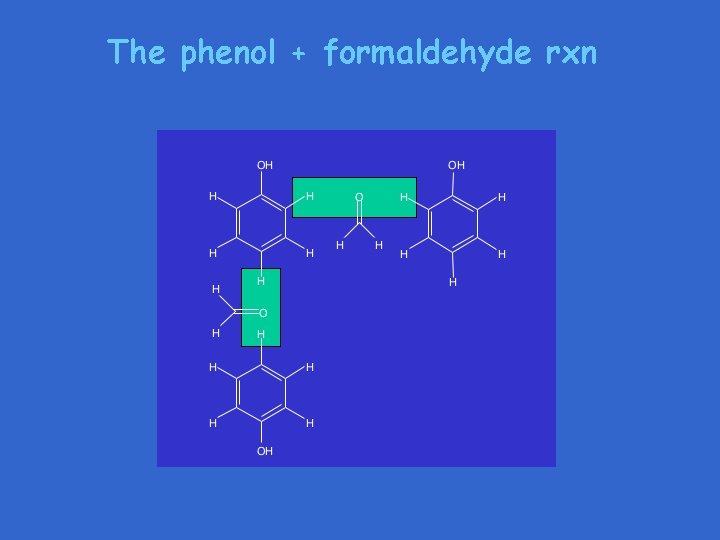
The phenol + formaldehyde rxn
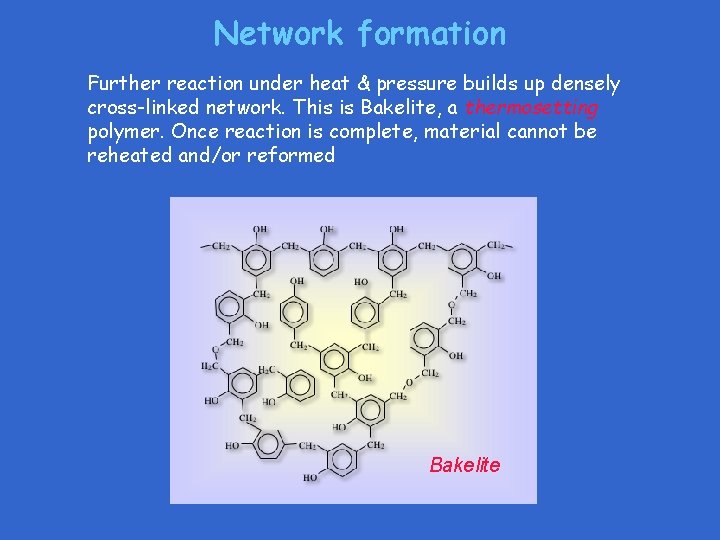
Network formation Further reaction under heat & pressure builds up densely cross-linked network. This is Bakelite, a thermosetting polymer. Once reaction is complete, material cannot be reheated and/or reformed Bakelite

Bakelite - Material of a Thousand Uses Clear Bakelite items Bakelite camera Phenolic resin/celluloid clock Bakelite telephone Bakelite radio Bakelite microphone
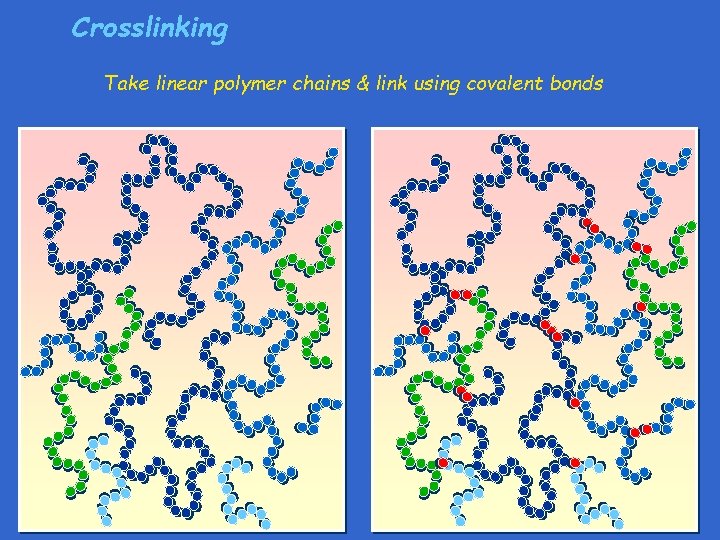
Crosslinking Take linear polymer chains & link using covalent bonds
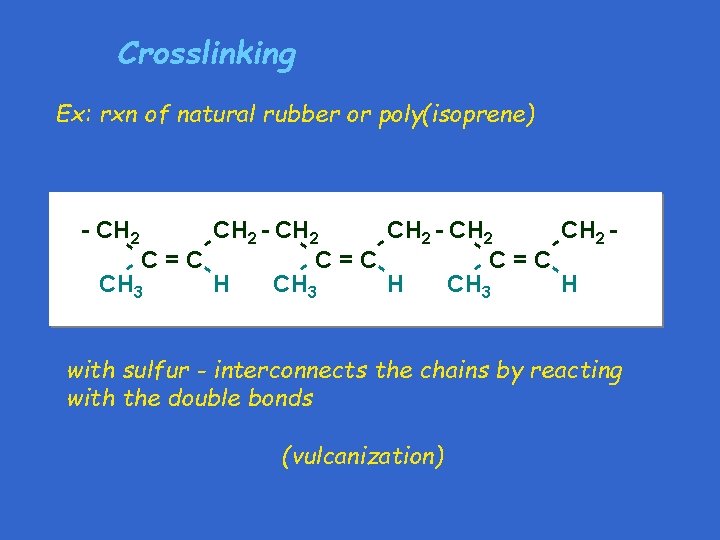
Crosslinking Ex: rxn of natural rubber or poly(isoprene) CH - CH 2 CH - 2 - 2 -C=C CH 3 H - - - CH 2 - - with sulfur - interconnects the chains by reacting with the double bonds (vulcanization)
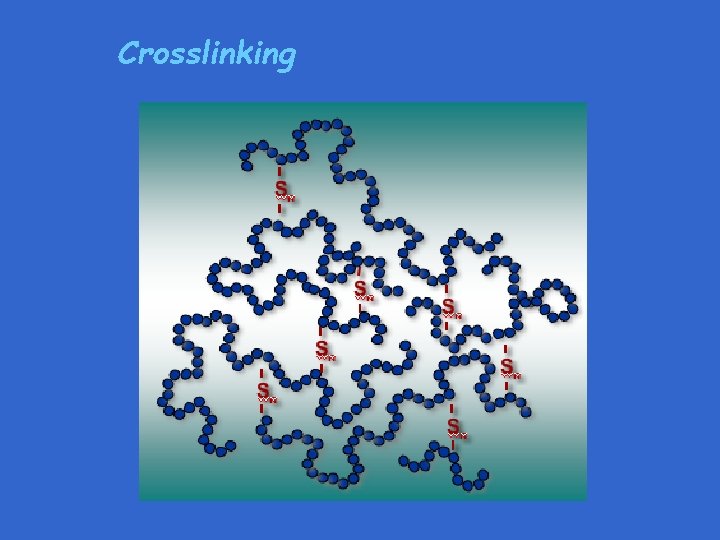
Crosslinking
- Slides: 24