Some Aspects of Calcium Looping Research at Imperial









- Slides: 23

Some Aspects of Calcium Looping Research at Imperial College, London J. Blamey, N. H. Florin, N. Paterson, D. R. Dugwell and P. S. Fennell* Department of Chemical Engineering, Imperial College, London *p. fennell@imperial. ac. uk 1 st I. E. A. High Temperature Solid Looping Cycles Network Meeting INCAR, Oviedo, Spain, 17 th September, 2009

Overview • Introduction to calcium looping / using Ca. O-based sorbents for CO 2 capture • Current projects relating to calcium looping in our group • Focus on a project related to hydration of spent sorbent • Focus on a project related to co-precipitation of synthetic sorbents

Introduction to Calcium Looping • Thank you to the previous speakers on the subject!

Current Projects • Optimisation of reactivation strategies for exhausted sorbents for CO 2, focusing on hydration [sponsored by EPSRC] • Design of synthetic sorbents by co-precipitation in a slurry bubble column [sponsored by the Grantham Institute for Climate Change, Imperial College] • Applications of the calcium looping cycle to cement manufacture [joint with industrial partner] • Morphology changes of limestone sorbent particles during carbonation/calcination looping cycles in a TGA (also useful for sorbent enhanced H 2 production) and reactivation with steam [joint project with CANMET, Canada, funded by KAUST] • H 2 production via sorbent-enhanced water-gas shift reactions [IC / KAUST] • UK /China H 2 production network (Imperial, Cambridge, Cranfield, Sheffield, Tsinghua, Taiyuan, NCEPU, TPRI, EPSRC funded) • Many other projects in the field of CO 2 capture, including amines, oxyfuel and chemical looping, as part of the Imperial College Centre for Carbon Capture and Storage (IC 4 S)

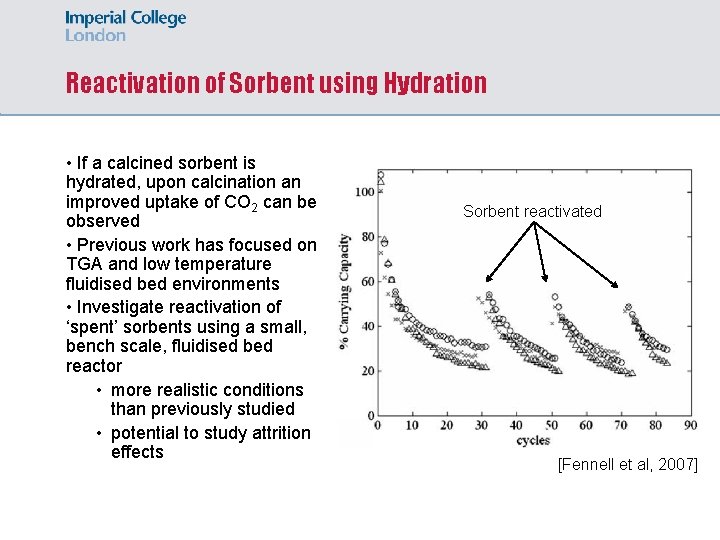
Reactivation of Sorbent using Hydration • If a calcined sorbent is hydrated, upon calcination an improved uptake of CO 2 can be observed • Previous work has focused on TGA and low temperature fluidised bed environments • Investigate reactivation of ‘spent’ sorbents using a small, bench scale, fluidised bed reactor • more realistic conditions than previously studied • potential to study attrition effects Sorbent reactivated [Fennell et al, 2007]

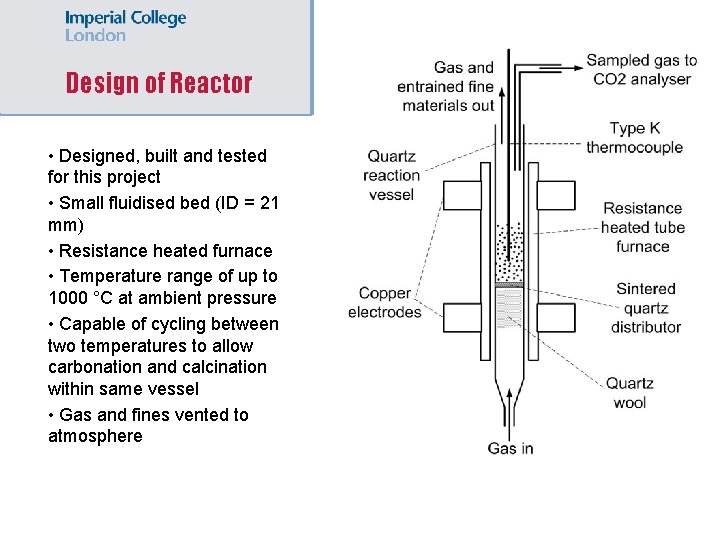
Design of Reactor • Designed, built and tested for this project • Small fluidised bed (ID = 21 mm) • Resistance heated furnace • Temperature range of up to 1000 °C at ambient pressure • Capable of cycling between two temperatures to allow carbonation and calcination within same vessel • Gas and fines vented to atmosphere

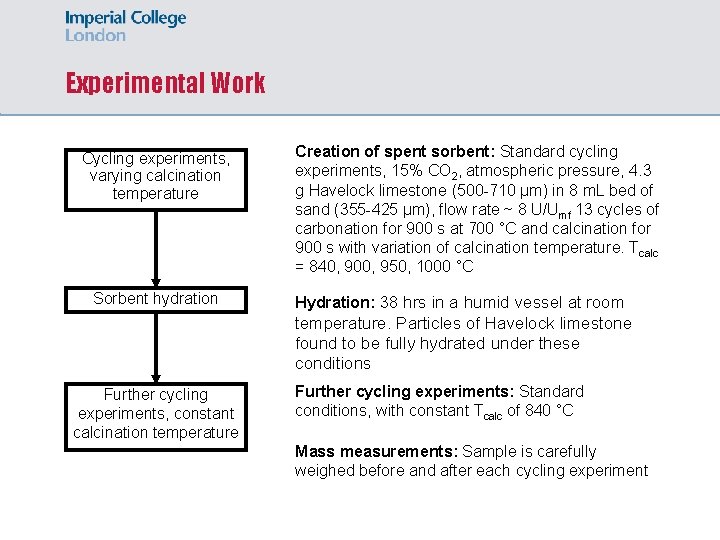
Experimental Work Cycling experiments, varying calcination temperature Sorbent hydration Creation of spent sorbent: Standard cycling experiments, 15% CO 2, atmospheric pressure, 4. 3 g Havelock limestone (500 -710 µm) in 8 m. L bed of sand (355 -425 µm), flow rate ~ 8 U/Umf 13 cycles of carbonation for 900 s at 700 °C and calcination for 900 s with variation of calcination temperature. Tcalc = 840, 900, 950, 1000 °C Hydration: 38 hrs in a humid vessel at room temperature. Particles of Havelock limestone found to be fully hydrated under these conditions Further cycling experiments, constant calcination temperature Further cycling experiments: Standard conditions, with constant Tcalc of 840 °C Mass measurements: Sample is carefully weighed before and after each cycling experiment

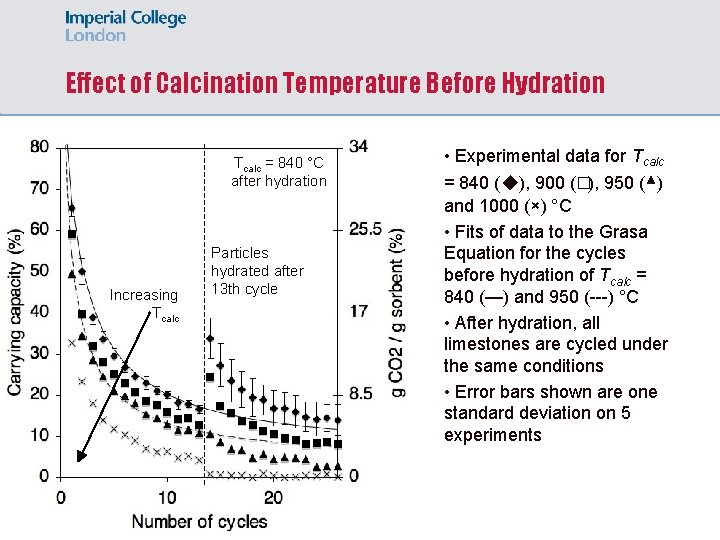
Effect of Calcination Temperature Before Hydration Tcalc = 840 °C after hydration Increasing Tcalc Particles hydrated after 13 th cycle • Experimental data for Tcalc = 840 (◆), 900 (�), 950 (▴) and 1000 (×) °C • Fits of data to the Grasa Equation for the cycles before hydration of Tcalc = 840 (—) and 950 (---) °C • After hydration, all limestones are cycled under the same conditions • Error bars shown are one standard deviation on 5 experiments

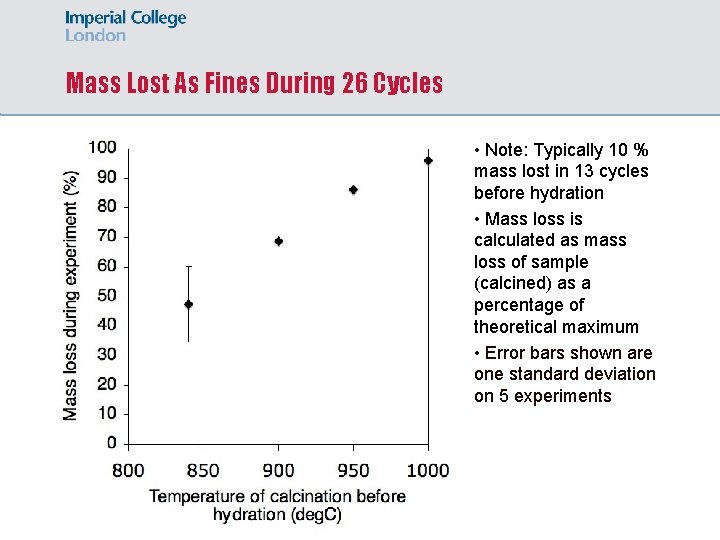
Mass Lost As Fines During 26 Cycles • Note: Typically 10 % mass lost in 13 cycles before hydration • Mass loss is calculated as mass loss of sample (calcined) as a percentage of theoretical maximum • Error bars shown are one standard deviation on 5 experiments

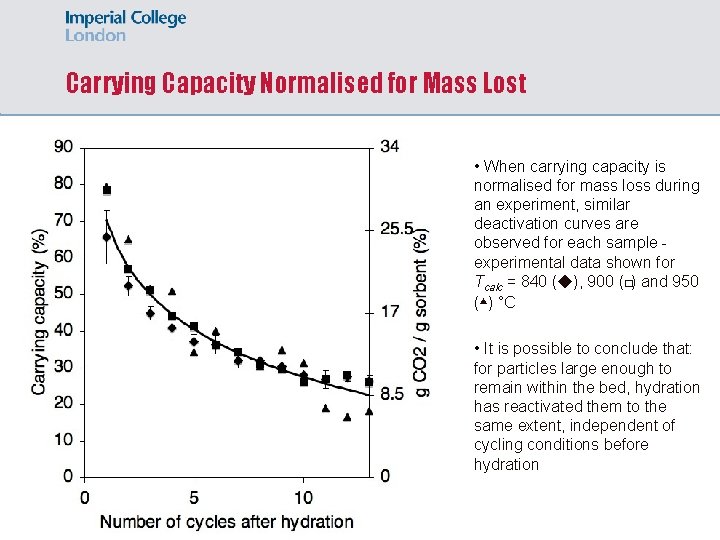
Carrying Capacity Normalised for Mass Lost • When carrying capacity is normalised for mass loss during an experiment, similar deactivation curves are observed for each sample experimental data shown for Tcalc = 840 (◆), 900 (�) and 950 (▴) °C • It is possible to conclude that: for particles large enough to remain within the bed, hydration has reactivated them to the same extent, independent of cycling conditions before hydration

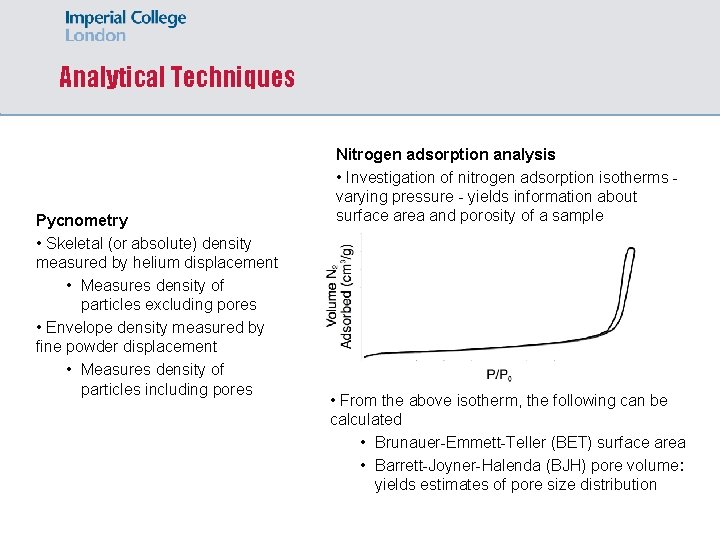
Analytical Techniques Pycnometry • Skeletal (or absolute) density measured by helium displacement • Measures density of particles excluding pores • Envelope density measured by fine powder displacement • Measures density of particles including pores Nitrogen adsorption analysis • Investigation of nitrogen adsorption isotherms varying pressure - yields information about surface area and porosity of a sample • From the above isotherm, the following can be calculated • Brunauer-Emmett-Teller (BET) surface area • Barrett-Joyner-Halenda (BJH) pore volume: yields estimates of pore size distribution

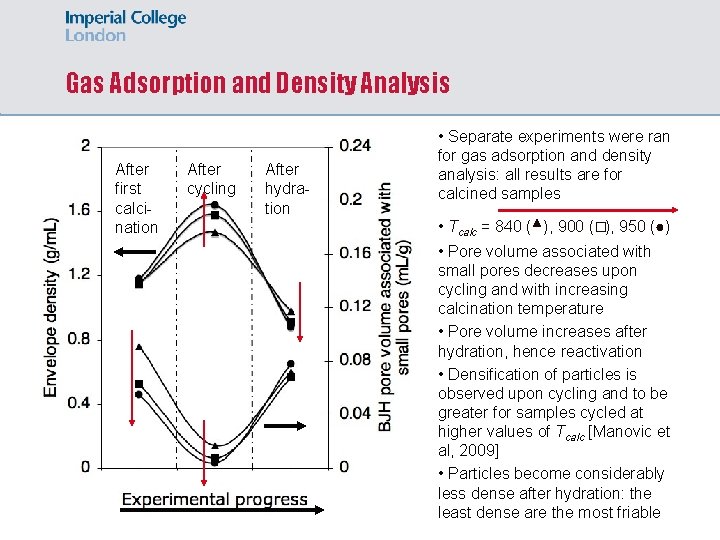
Gas Adsorption and Density Analysis After first calcination After cycling After hydration • Separate experiments were ran for gas adsorption and density analysis: all results are for calcined samples • Tcalc = 840 (▴), 900 (�), 950 (●) • Pore volume associated with small pores decreases upon cycling and with increasing calcination temperature • Pore volume increases after hydration, hence reactivation • Densification of particles is observed upon cycling and to be greater for samples cycled at higher values of Tcalc [Manovic et al, 2009] • Particles become considerably less dense after hydration: the least dense are the most friable

Optical Micrographs Before and After Hydration • Examples of particles before (left column) and after (right column) hydration. Two particles cycled at 1113 K show small increase in size upon hydration. One particle cycled at 1273 K shows larger increase in size upon hydration and mild fragmentation, whereas the other shatters. Quartz cylinders (diameter 0. 48 mm) used as reference distance.

Stress Distribution in the Growing Hydroxide Layer Balance on moles of Ca r 2 Ca(OH)2 r 0 Initially Ca. O, subsequently Ca(OH)2 Oxide grows from the inner boundary Geometry r 1

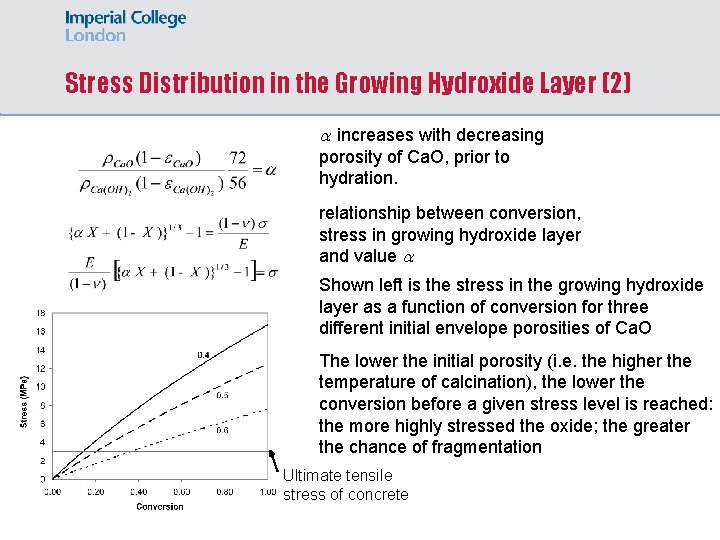
Stress Distribution in the Growing Hydroxide Layer (2) increases with decreasing porosity of Ca. O, prior to hydration. relationship between conversion, stress in growing hydroxide layer and value Shown left is the stress in the growing hydroxide layer as a function of conversion for three different initial envelope porosities of Ca. O The lower the initial porosity (i. e. the higher the temperature of calcination), the lower the conversion before a given stress level is reached: the more highly stressed the oxide; the greater the chance of fragmentation Ultimate tensile stress of concrete

Conclusions of Hydration Work • Upon hydration, more highly sintered sorbents are reactivated to a lesser extent • This is because of their increased friability upon hydration • A model has been developed that establishes a link between the porosity of a particle (which decreases upon sintering) and the tensile strength • The lower the initial porosity, the lower the conversion before a given stress level is reached

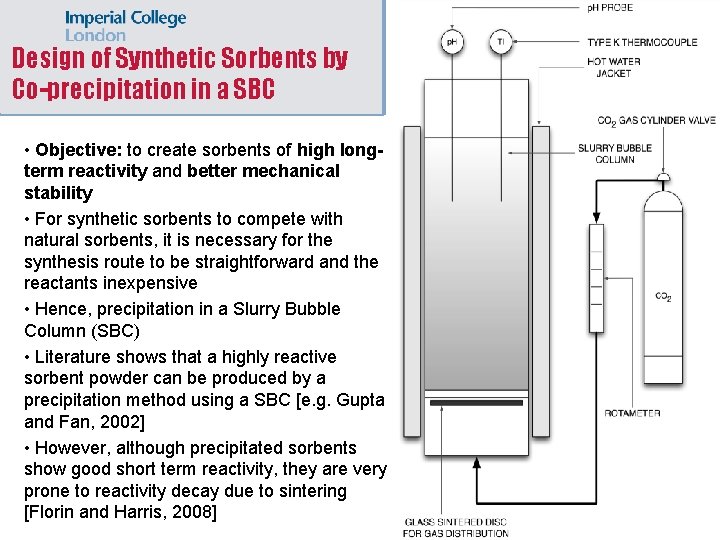
Design of Synthetic Sorbents by Co-precipitation in a SBC • Objective: to create sorbents of high longterm reactivity and better mechanical stability • For synthetic sorbents to compete with natural sorbents, it is necessary for the synthesis route to be straightforward and the reactants inexpensive • Hence, precipitation in a Slurry Bubble Column (SBC) • Literature shows that a highly reactive sorbent powder can be produced by a precipitation method using a SBC [e. g. Gupta and Fan, 2002] • However, although precipitated sorbents show good short term reactivity, they are very prone to reactivity decay due to sintering [Florin and Harris, 2008]

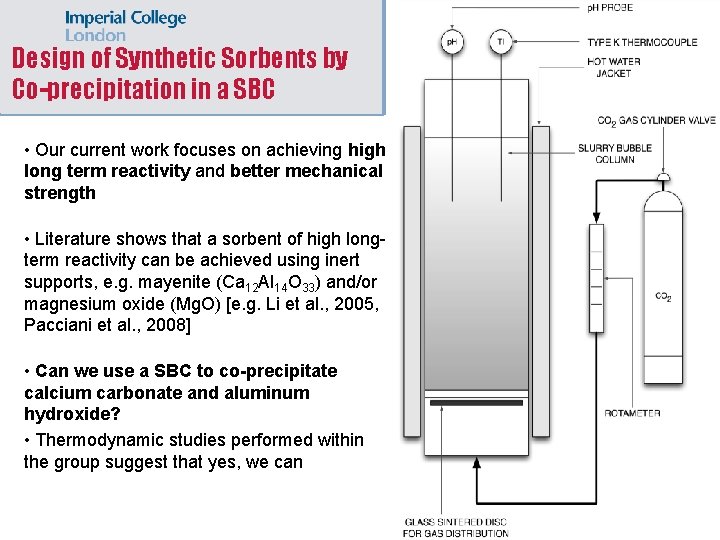
Design of Synthetic Sorbents by Co-precipitation in a SBC • Our current work focuses on achieving high long term reactivity and better mechanical strength • Literature shows that a sorbent of high longterm reactivity can be achieved using inert supports, e. g. mayenite (Ca 12 Al 14 O 33) and/or magnesium oxide (Mg. O) [e. g. Li et al. , 2005, Pacciani et al. , 2008] • Can we use a SBC to co-precipitate calcium carbonate and aluminum hydroxide? • Thermodynamic studies performed within the group suggest that yes, we can

Design of Synthetic Sorbents by Co-precipitation in a SBC: Exptl. • Main hypothesis: Better distribution of Ca. O achievable via co-precipitation vs. wet mixing or hydrolysis methods • Experiments performed in a SBC, varying: Ca(OH)2 loading and derivation amount of Al(NO)3. 9 H 2 O solution presence of dispersant (propan-2 -ol) presence of inorganic additives • carbonation initiated by bubbling through CO 2 – precipitating Ca. CO 3 seeded on finely dispersed Al(OH)3 • Preliminary results indicate high capture capacity on a g-CO 2/g- sorbent basis compared to Havelock; and method suited to using Ca 2+ dissolved from natural limestone

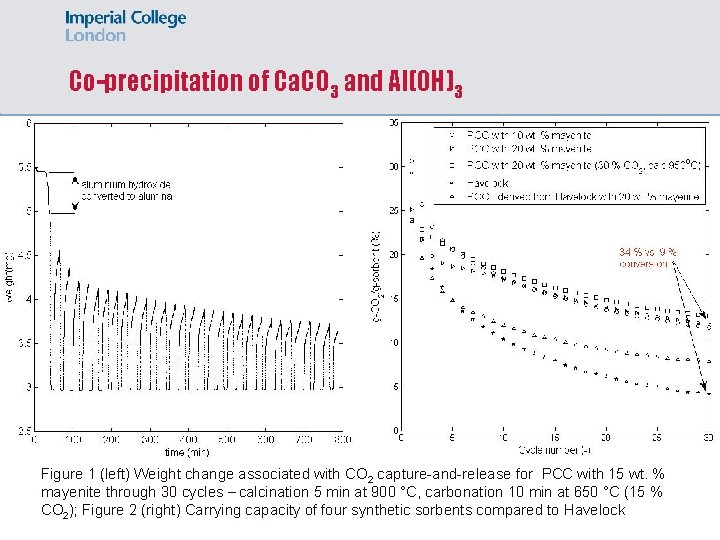
Co-precipitation of Ca. CO 3 and Al(OH)3 Figure 1 (left) Weight change associated with CO 2 capture-and-release for PCC with 15 wt. % mayenite through 30 cycles – calcination 5 min at 900 °C, carbonation 10 min at 650 °C (15 % CO 2); Figure 2 (right) Carrying capacity of four synthetic sorbents compared to Havelock

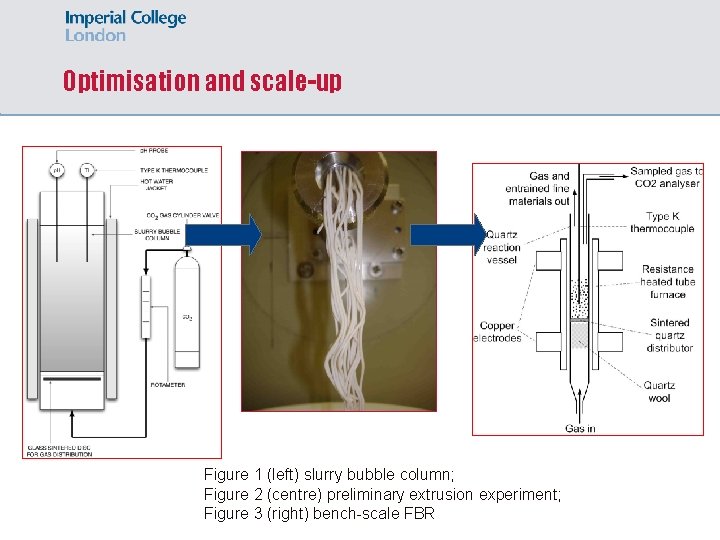
Optimisation and scale-up Figure 1 (left) slurry bubble column; Figure 2 (centre) preliminary extrusion experiment; Figure 3 (right) bench-scale FBR

Acknowledgements Engineering and Physical Sciences Research Council (EPSRC), UK King Abdullah University of Science and Technology (KAUST), Saudi Arabia National Resources, Canada The Grantham Institute for Climate Change, Imperial College Prof. Ben Anthony Prof. Rafael Kandiyoti Dr. Yinghai Wu Charles Dean Kelvin Okpoko

References Fennell, P. S. , Davidson, J. F. , Dennis, J. S. , and Hayhurst, A. N. , Regeneration of sintered limestone sorbents for the sequestration of CO 2 from combustion and other systems. Journal of the Energy Institute, 2007. 80(2): p. 116 -119. Florin, N. H. and Harris, A. T. , Screening Ca. O-based sorbents for CO 2 in biomass gasifiers. Energy and Fuels, 2008. 22(4): p. 2734 -2742. Gupta, H. and Fan, L. S. , Carbonation-calcination cycle using high reactivity calcium oxide for carbon dioxide separation from flue gas. Industrial & engineering chemistry research, 2002. 41(16): p. 4035 -4042. Li, Z. -S. , Cai, N. -S. , Huang, Y. -Y. , and Han, H. -J. , Synthesis, experimental studies, and analysis of a new calcium-based carbon dioxide absorbent. Energy & Fuels, 2005. 19(4): p. 1447 -1452. Manovic, V. , Charland, J. -P. , Blamey, J. , Fennell, P. S. , Lu, D. Y. , and Anthony, E. J. , Influence of calcination conditions on carrying capacity of Ca. O-based sorbent in CO 2 looping cycles. Fuel, 2009. 88(10): p. 1893 -1900. Pacciani, R. , Muller, C. R. , Davidson, J. F. , Dennis, J. S. , and Hayhurst, A. N. , Synthetic Cabased solid sorbents suitable for capturing CO 2 in a fluidized bed. The Canadian journal of chemical engineering, 2008. 86(3): p. 356 -366.