Solvent Effects on the Excited State Dynamics of






- Slides: 17

Solvent Effects on the Excited State Dynamics of 1 -cyclohexyluracil Patrick M. Hare Bern Kohler The Ohio State University Department of Chemistry 100 West 18 th Avenue Columbus, OH 43210 International Symposium on Molecular Spectroscopy 23 June 2005

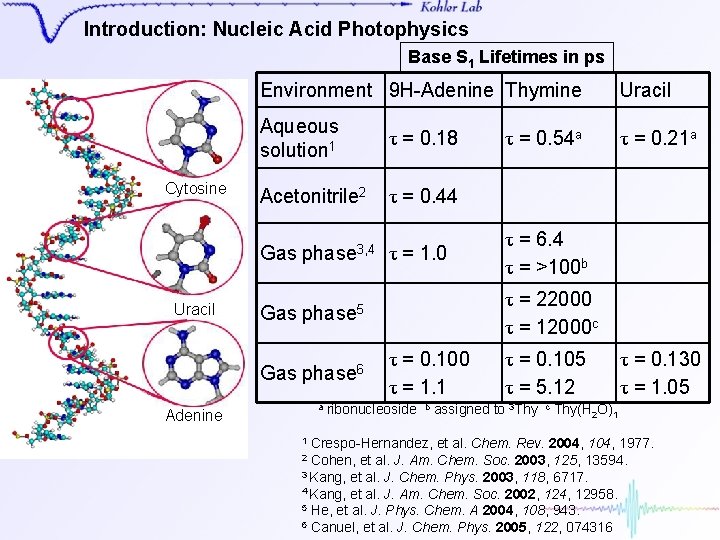
Introduction: Nucleic Acid Photophysics Base S 1 Lifetimes in ps Cytosine Thymine Uracil Environment 9 H-Adenine Thymine Uracil Aqueous solution 1 τ = 0. 18 τ = 0. 21 a Acetonitrile 2 τ = 0. 44 Gas phase 3, 4 Gas phase 5 Gas phase 6 a Adenine τ = 1. 0 τ = 0. 54 a τ = 6. 4 τ = >100 b τ = 22000 τ = 12000 c τ = 0. 100 τ = 1. 1 ribonucleoside b τ = 0. 105 τ = 5. 12 assigned to 3 Thy c τ = 0. 130 τ = 1. 05 Thy(H 2 O)1 Crespo-Hernandez, et al. Chem. Rev. 2004, 1977. Cohen, et al. J. Am. Chem. Soc. 2003, 125, 13594. 3 Kang, et al. J. Chem. Phys. 2003, 118, 6717. 4 Kang, et al. J. Am. Chem. Soc. 2002, 124, 12958. 5 He, et al. J. Phys. Chem. A 2004, 108, 943. 6 Canuel, et al. J. Chem. Phys. 2005, 122, 074316 1 2

Introduction: Uracil Photophysics The Triplet State in Uracil - Highest triplet yield of the bases - ΦISC reported in solvents with dielectric constants of 80 (H 2 O) – 25 (Et. OH) - 10 -fold increase in ΦISC in less polar solvents - μs lifetime quenched by O 2, selfquenching - Peaks at ~370 nm Our Tasks: Ø Investigate the excited state dynamics, especially in low polarity solvents and aprotic solvents Ø Determine the triplet yields ØInvestigate pump wavelength dependence The Singlet State in Uracil - ~200 fs lifetime in aqueous solution - ESA peaks at ~600 nm Aqueous Solutionsolution Gas phase / nonpolar np* pp** np T 0 S 0

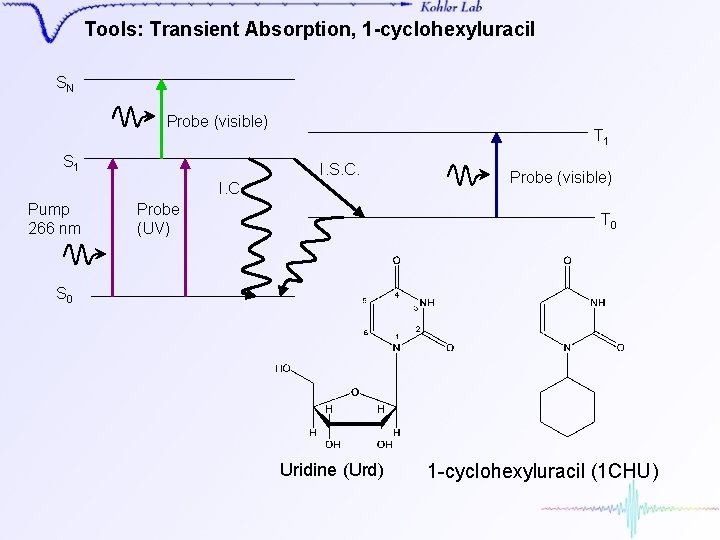
Tools: Transient Absorption, 1 -cyclohexyluracil SN Probe (visible) S 1 T 1 I. S. C. I. C. Pump 266 nm Probe (UV) Probe (visible) T 0 S 0 Uridine (Urd) 1 -cyclohexyluracil (1 CHU)

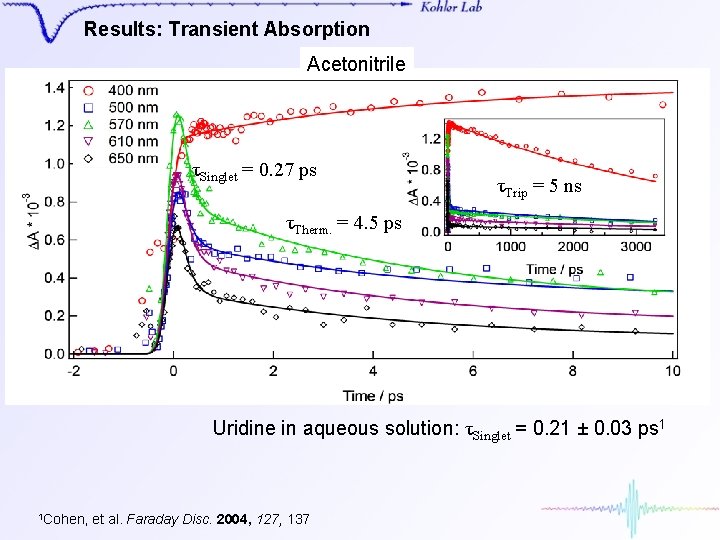
Results: Transient Absorption Acetonitrile τSinglet = 0. 27 ps τTrip = 5 ns τTherm. = 4. 5 ps Uridine in aqueous solution: τSinglet = 0. 21 ± 0. 03 ps 1 1 Cohen, et al. Faraday Disc. 2004, 127, 137

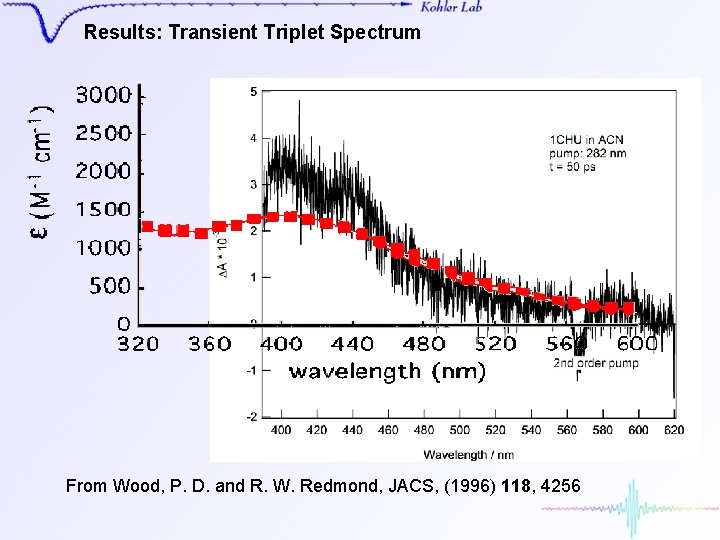
Results: Transient Triplet Spectrum From Wood, P. D. and R. W. Redmond, JACS, (1996) 118, 4256

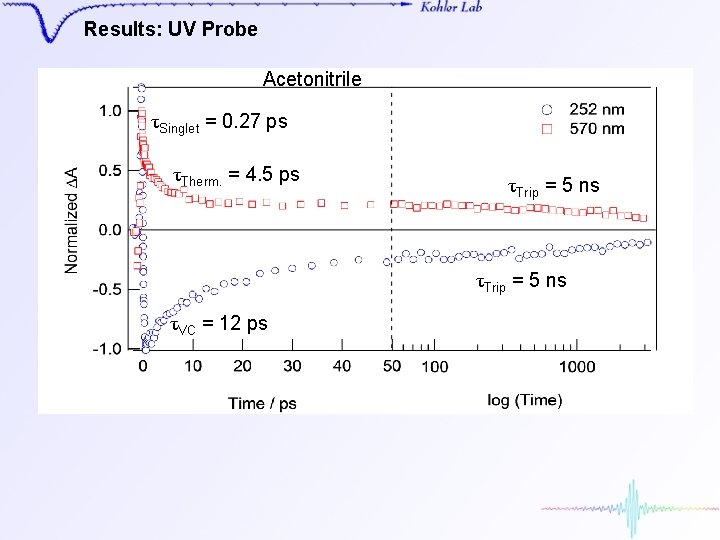
Results: UV Probe Acetonitrile τSinglet = 0. 27 ps τTherm. = 4. 5 ps τTrip = 5 ns τVC = 12 ps

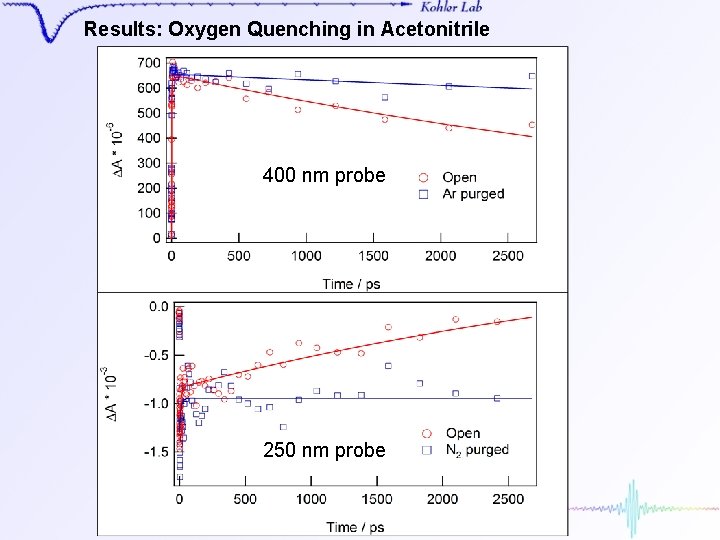
Results: Oxygen Quenching in Acetonitrile 400 nm probe 250 nm probe

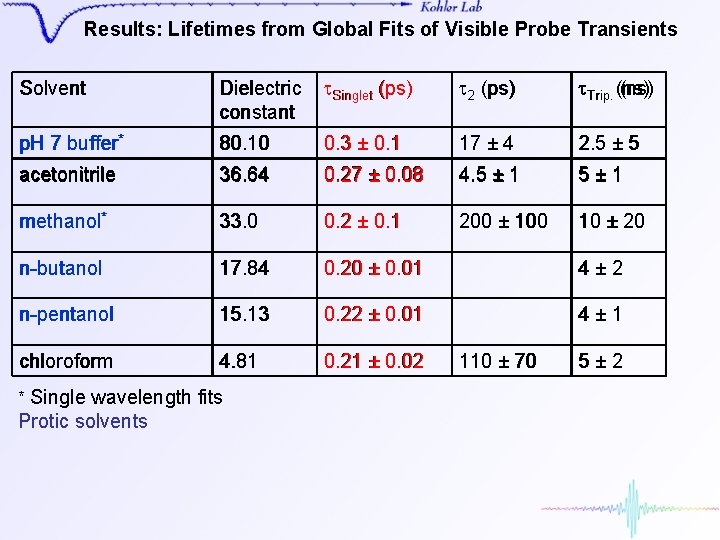
Results: Lifetimes from Global Fits of Visible Probe Transients Solvent Dielectric constant t. Singlet (ps) t 2 (ps) t. Trip (ns) Trip. (ns) p. H 7 buffer* 80. 10 0. 3 ± 0. 1 17 ± 4 2. 5 ± 5 acetonitrile 36. 64 0. 27 ± 0. 08 4. 5 ± 1 5± 1 methanol* 33. 0 0. 2 ± 0. 1 200 ± 100 10 ± 20 n-butanol 17. 84 0. 20 ± 0. 01 4± 2 n-pentanol 15. 13 0. 22 ± 0. 01 4± 1 chloroform 4. 81 0. 21 ± 0. 02 * Single wavelength fits Protic solvents 110 ± 70 5± 2

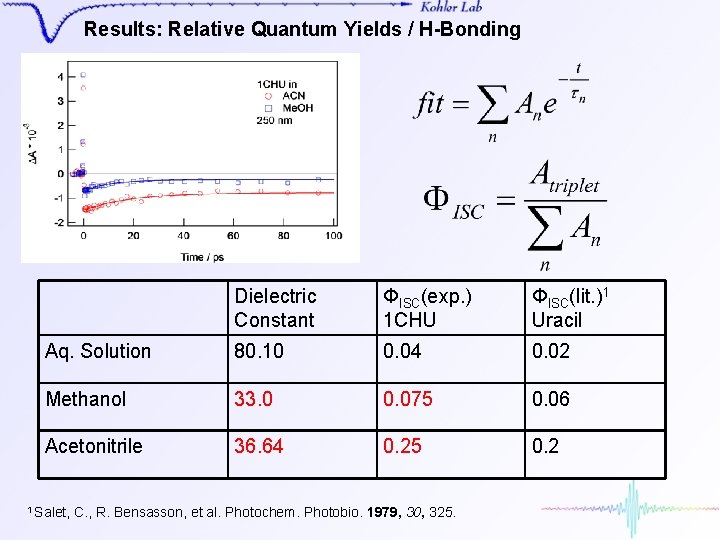
Results: Relative Quantum Yields / H-Bonding Dielectric Constant ΦISC(exp. ) 1 CHU ΦISC(lit. )1 Uracil Aq. Solution 80. 10 0. 04 0. 02 Methanol 33. 0 0. 075 0. 06 Acetonitrile 36. 64 0. 25 0. 2 1 Salet, C. , R. Bensasson, et al. Photochem. Photobio. 1979, 30, 325.

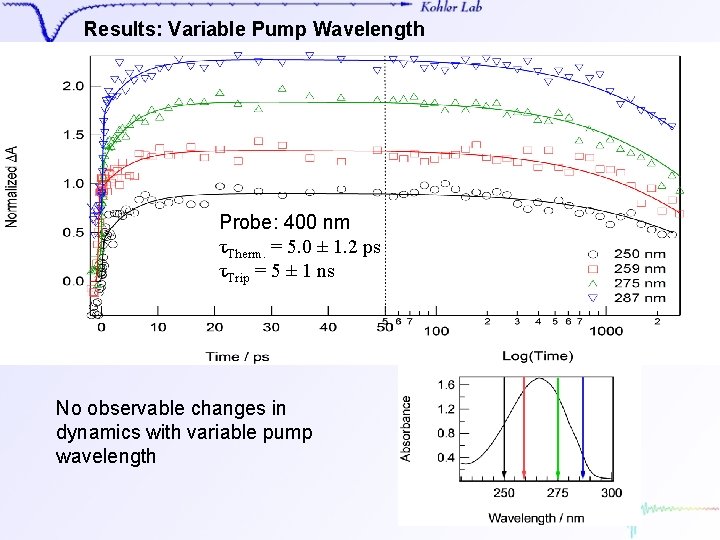
Results: Variable Pump Wavelength Probe: 400 nm τTherm. = 5. 0 ± 1. 2 ps τTrip = 5 ± 1 ns No observable changes in dynamics with variable pump wavelength

Conclusions 1 CHU is soluble in solvents with ε >5 ns lifetime observed in 1 CHU is the triplet state Triplet is formed very rapidly Excited state lifetime shows no appreciable solvent effects Hydrogen bonding is important for decreasing ΦISC No change in lifetimes with varying pump wavelength Future work Determine ΦISC in more solvents Further investigation of the variation of ΦISC with pump wavelength

Acknowledgements Dr. Bern Kohler Group Funding National Institutes of Health Dr. Terry Gustafson

Obligatory Laser System Slide 1

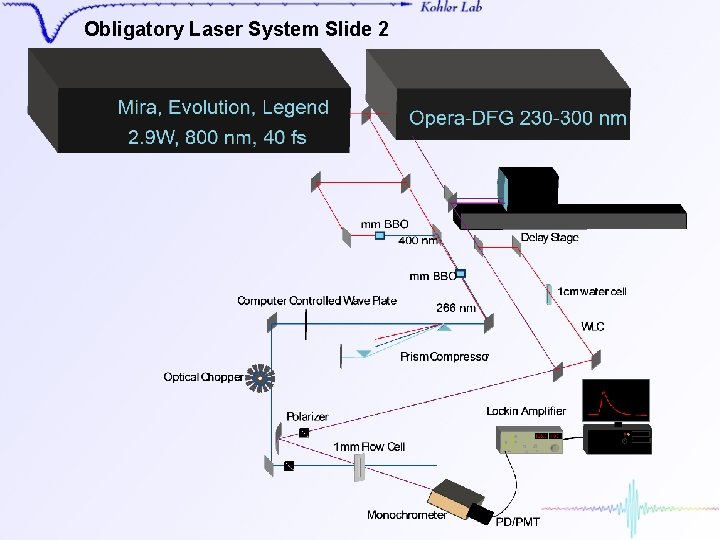
Obligatory Laser System Slide 2

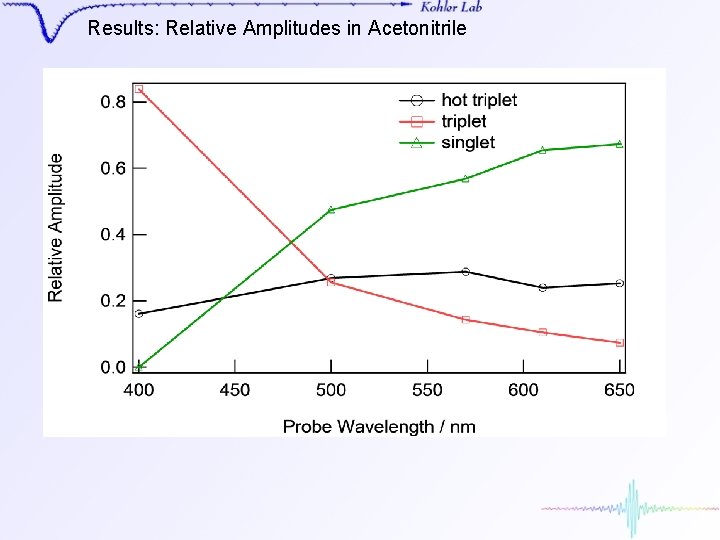
Results: Relative Amplitudes in Acetonitrile

Results: Summary of Dynamics in Acetonitrile SN ~600 nm T 1 S 1 τIC = 0. 2 ps τISC ~ 0. 2 ps ~370 nm τTherm ~ 5 ps 266 nm T 0 τVC = 12 ps S 0 τISC ~ 5000 ps