Solutions Vocabulary suspension colloid soluble miscible insoluble Immiscible

Solutions Vocabulary: suspension colloid soluble miscible insoluble Immiscible Concentration Solute Solvent Concentrated Dilute Molarity Solvation Unsaturated solution Saturated solution supersaturated solution

Objectives: 1. Identify types of solutions 2. Describe concentration and explain why water is used as the solvent 3. Calculate Molarity and dilutions 4. Explain how to make a solution and dilution 5. Explain how solubility can be changed for different states of matter 6. Describe how solutes change freezing point and boiling point

Heterogeneous Mixtures • A heterogeneous mixture is a mixture that does not have a uniform composition and in which the individual substances remain distinct. • Suspensions are mixtures containing particles that settle out if left undisturbed.

Heterogeneous Mixtures • Colloids are heterogeneous mixtures of intermediate sized particles (between 1 nm and 1000 nm) and do not settle out. • The most abundant substance in a mixture is the dispersion medium.

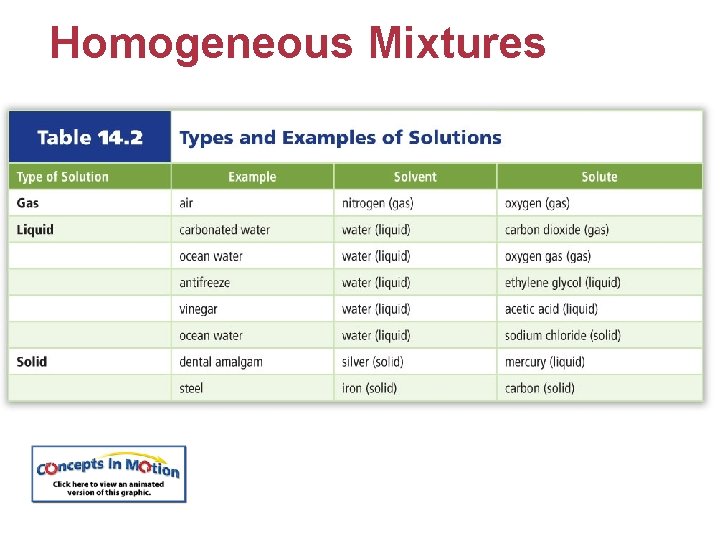
Homogeneous Mixtures • Solutions are homogeneous mixtures that contain two or more substances called the solute and solvent. − Solute: what is being dissolved − Solvent: what is doing the dissolving • Most solutions are liquids, but gaseous and solid solutions exist.
Homogeneous Mixtures

Homogeneous Mixtures • A substance that dissolves in a solvent is soluble. • Two liquids that are soluble in each other in any proportion are miscible. • A substance that does not dissolve in a solvent is insoluble. • Two liquids that can be mixed but separate shortly after are immiscible.

Section 14. 1 Assessment Miscible substances are: A. two liquids that are not soluble in each other B. solids that dissolve in liquids C. solids that do not dissolve in liquids D. two liquids that are soluble in each other A. B. C. D. A B C D

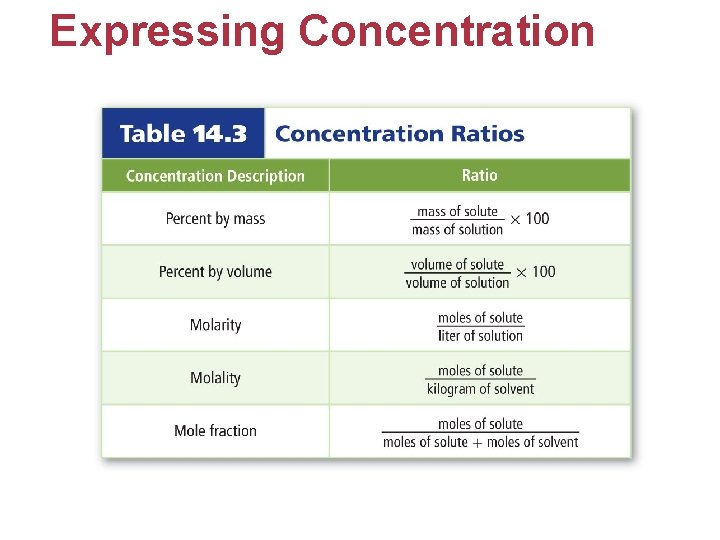
Expressing Concentration • The concentration of a solution is a measure of how much solute is dissolved in a specific amount of solvent or solution. • Concentration is described as concentrated or dilute. • Concentrated: contains large amounts of solute • Dilute: contains small amounts of solute
Expressing Concentration

Expressing Concentration • Molarity is the number of moles of solute dissolved per liter of solution. • Dilution equation: M 1 V 1 = M 2 V 2

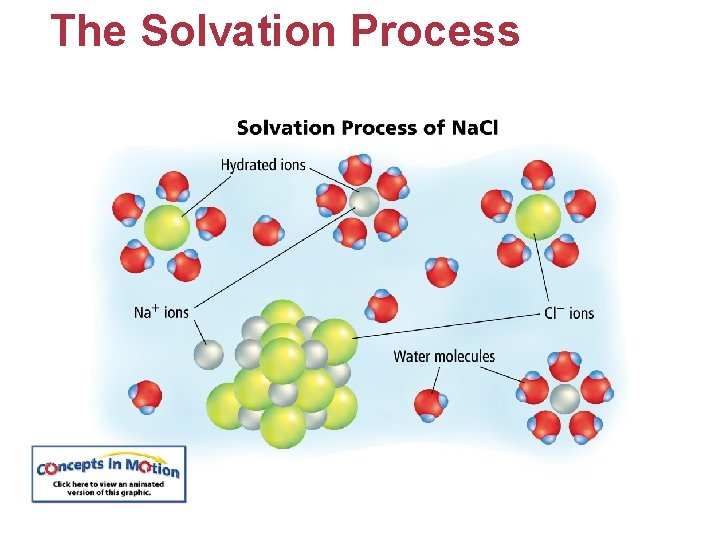
The Solvation Process • Solvation is the process of surrounding solute particles with solvent particles to form a solution. • Solvation in water is called hydration. • The attraction between dipoles of a water molecule and the ions of a crystal are greater than the attraction among ions of a crystal.
The Solvation Process

1. 2. 3. 4. 5. 6. Why is Water the “Universal Solvent” Water is BENT and POLAR and therefore forms HYDROGEN BONDS! High Surface tension: acts like a skin on the surface of the liquid. Low vapor pressure and High boiling point: this means the amount of energy necessary to boil is very large! High Specific Heat Crystallizes into hexagonal patterns Dissolves other polar substances and ionic salts
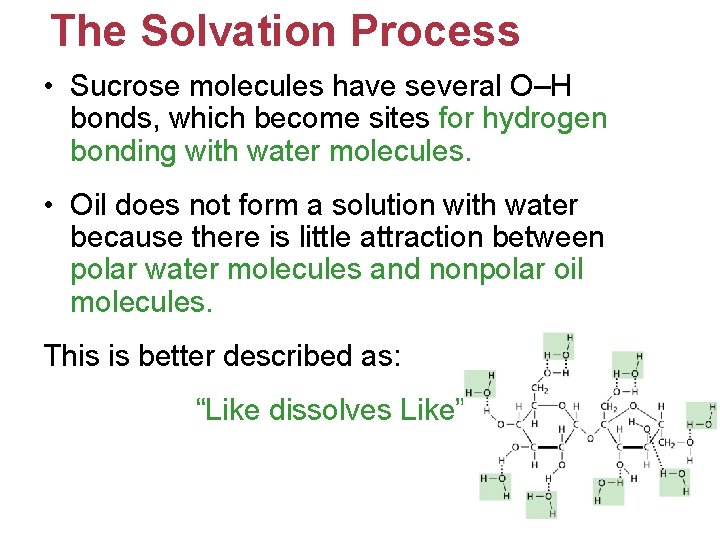
The Solvation Process • Sucrose molecules have several O–H bonds, which become sites for hydrogen bonding with water molecules. • Oil does not form a solution with water because there is little attraction between polar water molecules and nonpolar oil molecules. This is better described as: “Like dissolves Like”

Factors That Affect Solvation • Stirring or shaking moves dissolved particles away from the contact surfaces more quickly and allows new collisions to occur. • Breaking the solute into small pieces increases surface area and allows more collisions to occur. • As temperature increases, rate of solvation increases.

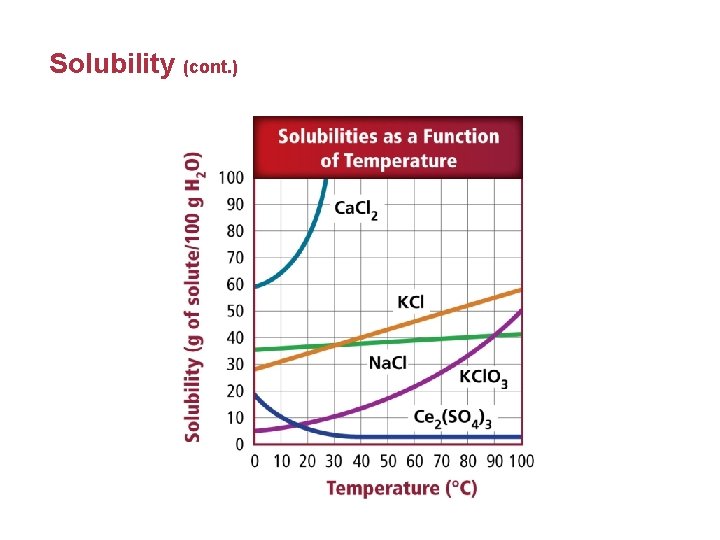
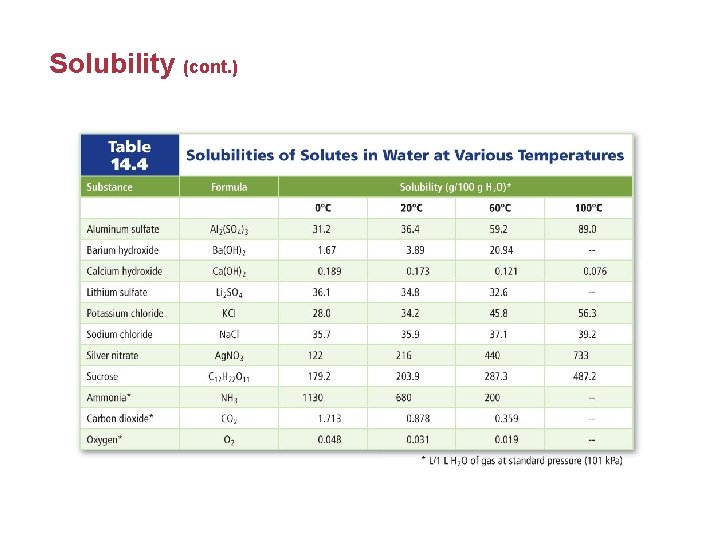
Solubility • Solubility depends on the nature of the solute and solvent. It is the solutes ability to dissolve in the solvent. • Unsaturated solutions are solutions that contain less dissolved solute for a given temperature and pressure than a saturated solution.

Solubility • Saturated solutions contain the maximum amount of dissolved solute for a given amount of solvent at a specific temperature and pressure. • Solubility is affected by increasing the temperature of the solvent because the kinetic energy of the particles increases.
Solubility (cont. )

Solubility • A supersaturated solution contains more dissolved solute than a saturated solution at the same temperature. • To form a supersaturated solution, a saturated solution is formed at high temperature and then slowly cooled. • Supersaturated solutions are unstable.
Solubility (cont. )
- Slides: 21