Solutions Vocabulary Solution homogeneous mixture Solid Brass steel

Solutions

Vocabulary Solution: homogeneous mixture Solid – Brass, steel Liquid – Salt dissolved in water Gas – Air Solute: substance that dissolves Solvent: substance in which the solute dissolves

Vocabulary Cont’d Insoluble: substance that cannot be dissolved Soluble: substance that can be dissolved Miscible: 2 liquids mix completely Immiscible: 2 liquids that do not mix completely

Soluble vs. Insoluble “Like dissolves Like” Rule: Only substances that are similar/alike will dissolve into one another Polar and Polar (dissolve) Nonpolar and Nonpolar (dissolve) Polar and Nonpolar (do NOT dissolve) Water is Polar! (IMPORTANT)
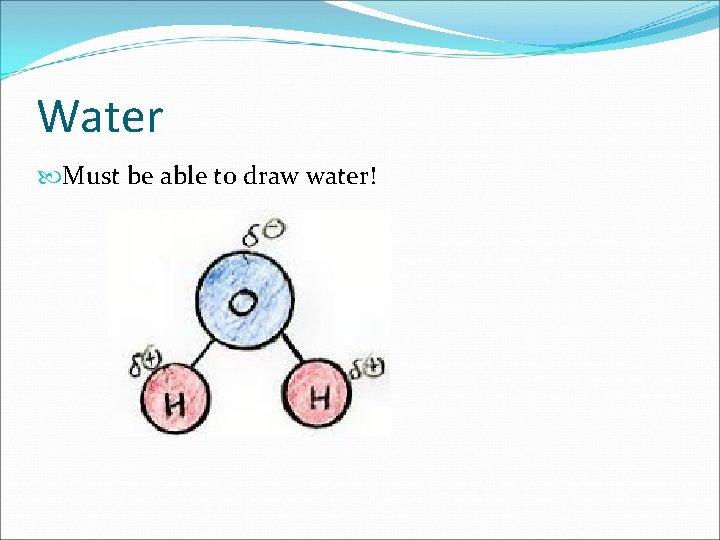
Water Must be able to draw water!

Dissolving & Solubility Factors that can effect the RATE (how fast/slow) dissolving: Temperature Stirring/Agitation Surface Area Remember Clear Spring High (CSH) stands for Heat, Stir, Crush

Heat of Solution Endothermic: energy is put into the compound (feels cold) Exothermic: energy is released from the compound (feels hot) The overall change is the heat of solution

Solubility The maximum amount of solute that will dissolve at a specified temperature If there are more grams of solute than can dissolve, some of the solute will fall out of solution

Saturation Unsaturated: when a solution can hold more solute and it does dissolve Saturated: when a solution cannot dissolve any more solute Supersaturated: contains more solute than a saturated solution at the same temperature extremely unstable and if a small amount of solute (seed crystal) is added the excess solute quickly falls out of solution (Example: hand warmers, rock candy, and cloud seeding)
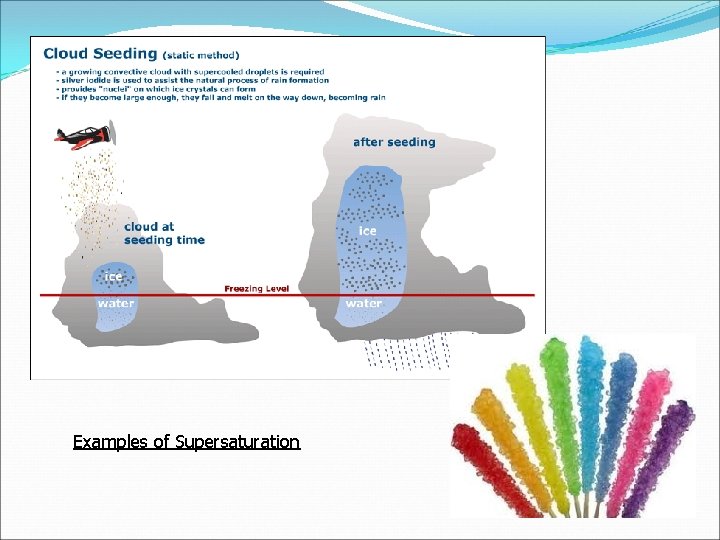
Examples of Supersaturation
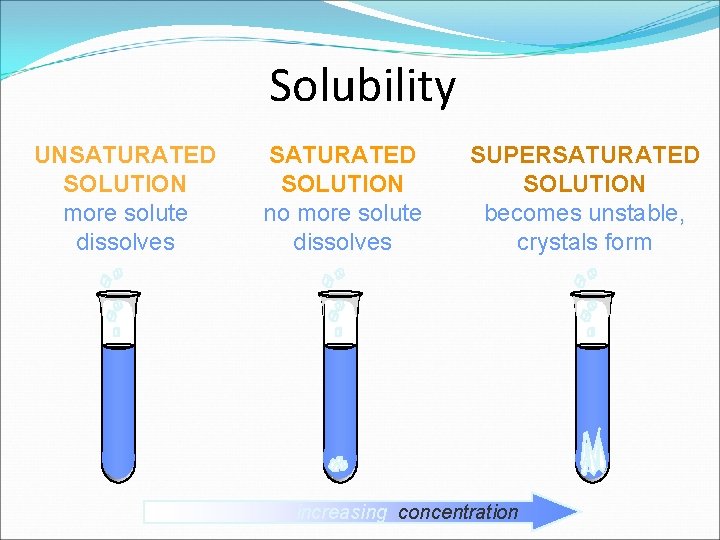
Solubility UNSATURATED SOLUTION more solute dissolves SATURATED SOLUTION no more solute dissolves SUPERSATURATED SOLUTION becomes unstable, crystals form increasing concentration

Solubility Curves Know how to read this curve and answer questions Examples + If 70 grams of Na. NO 3 are dissolved in water at 10°C, is the solution saturated, unsaturated, or supersaturated? + If 20 grams of Na. Cl are dissolved in water at 80° C, then how many more grams can be added to make a saturated solution?
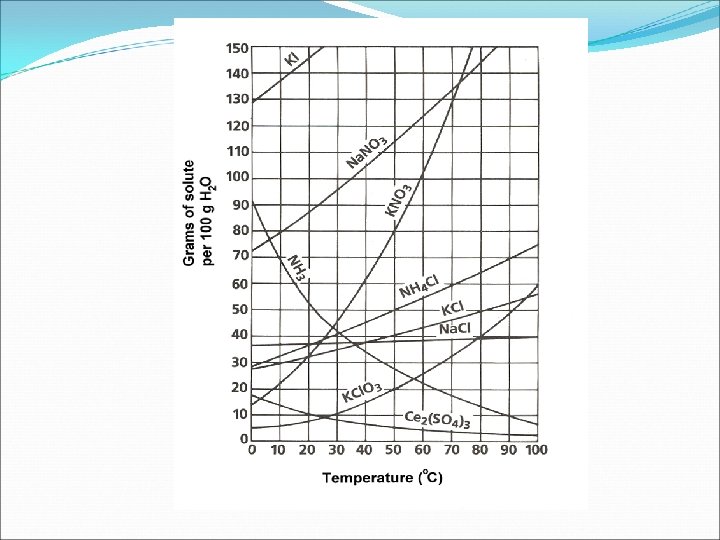

Temperature and Solubility Solids Increase Temp. will Increase Dissolving (Solubility) Decrease Temp. will Decrease Dissolving (Solubility) Gases (Different from Solids) Increase Temp. will Decrease Dissolving (Solubility) Decrease Temp. will Increase Dissolving (Solubility)
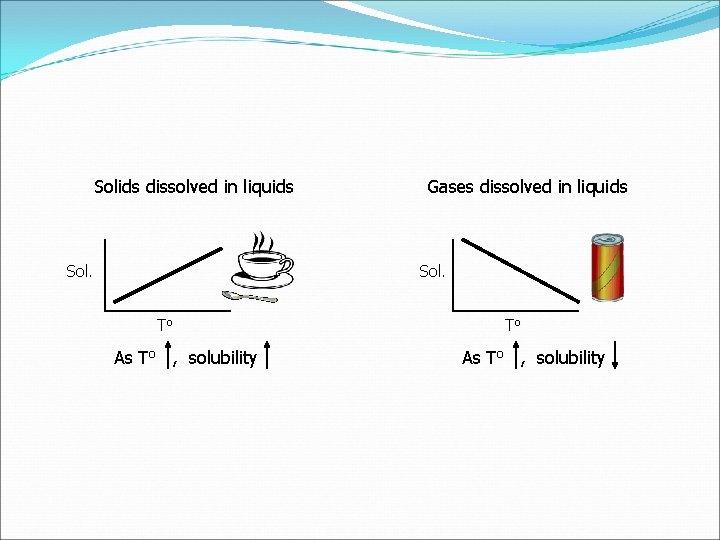
Solids dissolved in liquids Sol. Gases dissolved in liquids Sol. To As To , solubility

Water and Oxygen Dissolved Oxygen – The amount of oxygen that is dissolved in water If dissolved oxygen is too low, animal and aquatic life is unable to survive and the water becomes toxic Happens commonly in lakes and ponds during the summer months

Pressure and Solubility (Gases) A change in pressure on solution with a dissolved solid or liquid has very little effect on solubility. A change in pressure on a solution with a dissolved gas has a large effect on solubility. Solubility of a gas increases as the external pressure increases When pressure on the outside decreases some of the gas escapes.

Henry’s Law – At a given temperature the solubility (S) of a gas in a liquid is directly proportional to the pressure (P) above liquid S 1 P 2 = S 2 P 1 Show work, answer, units Units must match Label properly 1=initial condition and 2=final condition Only applies at constant temperature

Practice If 0. 24 g of a gas dissolves in 1. 0 L of water at 1. 5 atm of pressure, how much of the gas will dissolve if the pressure is raised to 6. 0 atm? Assume the temperature is held constant. Answer: 0. 96 g/L

Practice The solubility of a gas changes from 0. 95 g/L to 0. 72 g/L. If the initial pressure was 2. 8 atm, what is the final pressure? Answer: 2. 12 atm

Concentration How much solute is dissolved in a specific amount of solvent or solution. Can be expressed as Percent by mass - Tuesday Percent by volume - Tuesday Molarity – Wednesday Molality and Normality – We will not learn these, but they are mentioned in the book

Percent by Mass (Mass of solute/Mass of solution) x 100 Used when a solid is dissolved in a liquid Mass of solution is equal to the mass of the solute + the mass of the solvent

Percent by mass problems In order to maintain a sodium chloride concentration similar to ocean water, an aquarium must contain 30 g of Na. Cl per 500 grams of water. What is the percent mass of the solution? Answer: 5. 66%

Percent by Mass Problems You have 150 grams of Cu. Cl 2 solution. The percent by mass of the solute is 8. 0%. How many grams of Cu. Cl 2 are in solution? How many grams of solvent are in the solution? Answer: 12 g of Cu. Cl 2 138 g of water

Percent by Volume Usually describes solutions where both solute and solvent are liquids Volume of solute/Volume of solution x 100

Percent by Volume Problems What is the percent by volume of ethanol in a solution that contains 50 ml of ethanol distilled in 430 ml of water? Answer: 10. 42%

Percent by Volume Problems If you have 30 ml of a 5% aqueous solution of H 2 O 2, what volumes of H 2 O 2 and water are in the solution? Answer: 1. 5 m. L of H 2 O 2 28. 5 m. L of H 2 O

Molarity Its unit is a M = mol/L Moles of solute/Liters of solution

A 100. 5 ml solution contains 5. 10 grams of Glucose (C 6 H 12 O 6). What is the molarity of this solution? The molar mass of glucose is 180. 18 g/mol Answer: 0. 28 M

Calculate the molarity of 1. 60 liters of a solution containing 1. 55 grams of dissolved KBr. Answer: 0. 0081 M

Making a molar solution Using the molarity of the solution figure out how many moles of the solute will be needed. Using the moles of solute, convert it to grams of solute

How many grams of Ca. Cl 2 would be dissolved in 1. 0 liters of a. 10 M solution? Answer: 110. 98 g Ca. Cl 2

How many grams of Ca. Cl 2 should be dissolved to make 500 ml of a. 20 M solution? Answer: 11. 10 g Ca. Cl 2

Diluting a stock solution Stock solution is a concentrated solution M 1 V 1 = M 2 V 2 Lab: 1. 0 M H 2 SO 4 is needed 130 students Each set of lab partners needs 30 m. L Glassware 0. 5 L, 1 L, or 2 L How much should I make?

How many ml of a 18. 0 M H 2 SO 4 stock solution would you need to prepare 2 L of 1. 0 M H 2 SO 4? Answer: 111 m. L

How Do You Perform This Dilution? 1. Fill a 2 liter volumetric flask half-full of distilled water. 2. Measure 111 m. L of 18. 0 M H 2 SO 4 in graduated cylinder. 3. Slowly pour the acid into the water. 4. After each addition, swirl the flask to enhance mixing and to allow the exothermic heat of solution to dissipate. 5. Fill the volumetric to the “line” with distilled water. 6. Your flask now contains 2. 0 L of a 1 M solution of H 2 SO 4 that can be used for the lab.

Viscosity Physical Property Fluids resistance to flow Fluid pours fast means LOW viscosity (example: water, coffee, tea) Fluid pours slowly means HIGH viscosity (example: syrup, ketchup)

Dissolving Ionization: The breaking up of a compound into separate ions Electrolyte: An ionic/soluble compound whose aqueous solutions conducts electricity Nonelectrolyte: A molecular (covalent) substance whose aqueous solutions do not conduct electricity Precipitate: A product of a reaction that is insoluble and falls out of solution as a solid.
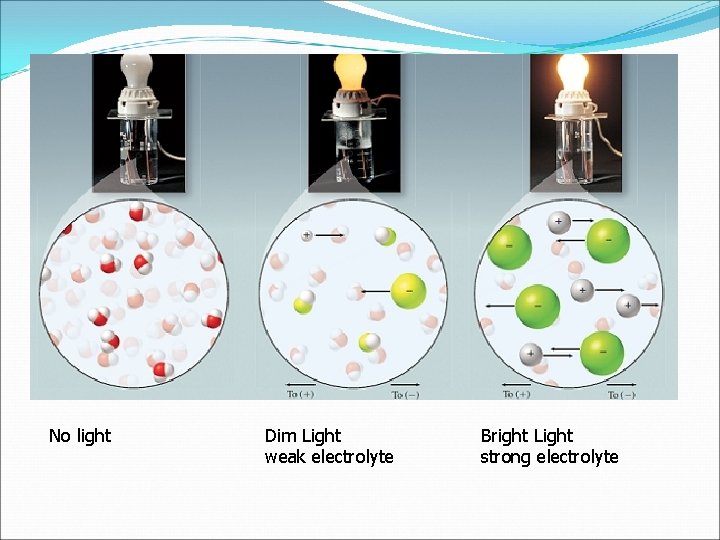
No light Dim Light weak electrolyte Bright Light strong electrolyte

How to Determine if a Substance is an Electrolyte or Precipitate? 1. Is the substance ionic or covalent? (Where are they on periodic table? ) 1. Is the substance soluble or insoluble? (Use the solubility rules)

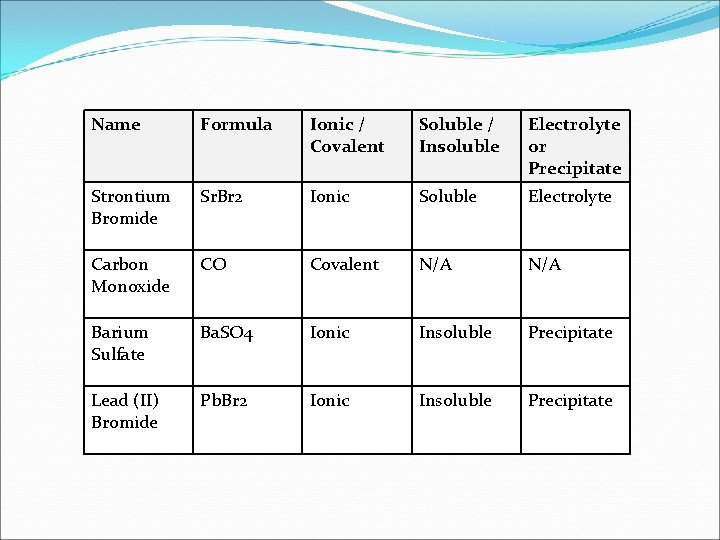
Name Formula Ionic / Covalent Soluble / Insoluble Electrolyte or Precipitate Strontium Bromide Sr. Br 2 Ionic Soluble Electrolyte Carbon Monoxide CO Covalent N/A Barium Sulfate Ba. SO 4 Ionic Insoluble Precipitate Lead (II) Bromide Pb. Br 2 Ionic Insoluble Precipitate
- Slides: 43