Solutions Types of Mixtures Heterogeneous mixturenot uniform throughout







- Slides: 12

Solutions

Types of Mixtures • Heterogeneous mixture-not uniform throughout; particles are large enough to see under a microscope; layers form – Suspension-the particles do not remain mixed unless the mixture is constantly stirred or agitated – Example: liquid medications

Types of Mixtures • Colloid-particles that are bigger than the particles in a solution but smaller than the particles in a suspension; can be noticed under a microscope. – Example: Milk

Types of Mixtures • Homogeneous mixture-uniform throughout; can’t distinguish between the types of particles; no layers form.

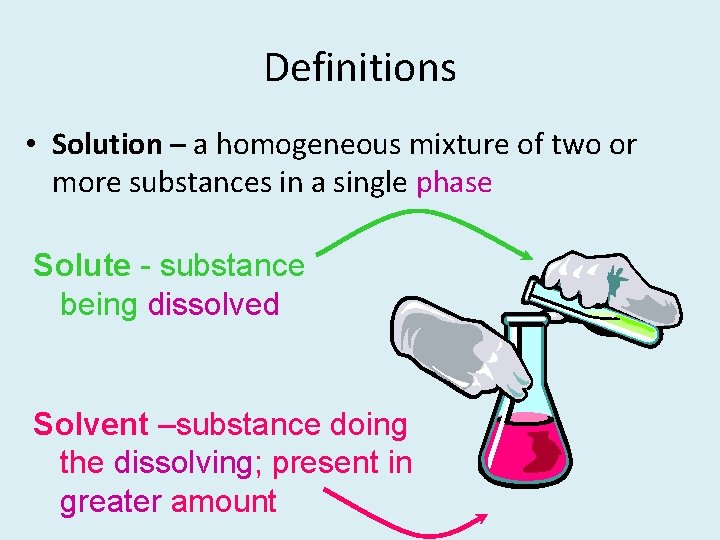
Definitions • Solution – a homogeneous mixture of two or more substances in a single phase Solute - substance being dissolved Solvent –substance doing the dissolving; present in greater amount

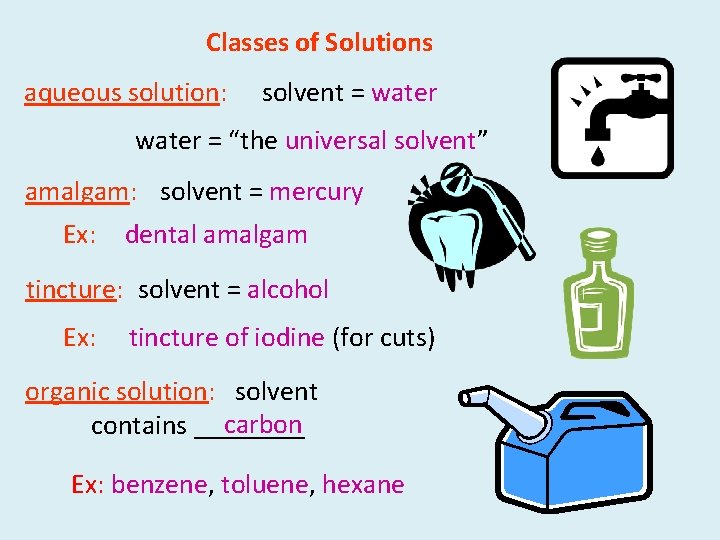
Classes of Solutions aqueous solution: solvent = water = “the universal solvent” amalgam: solvent = mercury Ex: dental amalgam tincture: solvent = alcohol Ex: tincture of iodine (for cuts) organic solution: solvent carbon contains ____ Ex: benzene, toluene, hexane

Types of Solutions • Gaseous solutions – Ex: air • Solid solutions-one solid is mixed with another solid and is called an alloy – Ex: brass-a mixture of copper and zinc

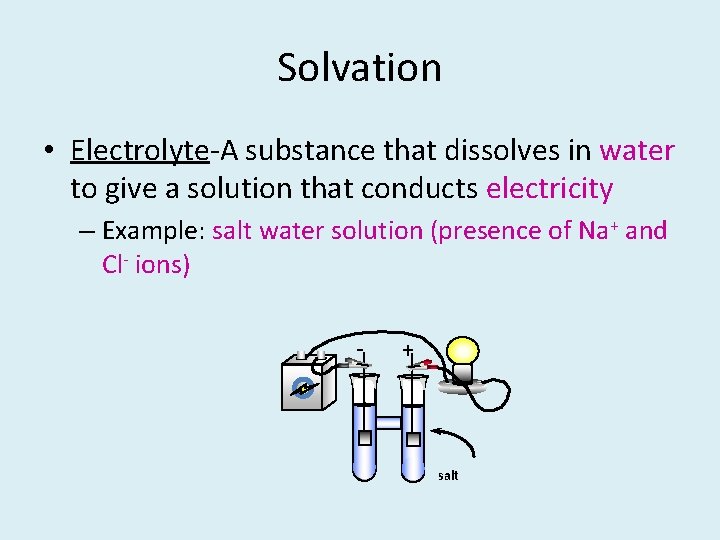
Solvation • Electrolyte-A substance that dissolves in water to give a solution that conducts electricity – Example: salt water solution (presence of Na+ and Cl- ions) - + salt

Solvation • Nonelectrolyte-A substance that dissolves in water to give a solution that does not conduct electricity. – Example: sugar solution (no ions in solution) - + sugar

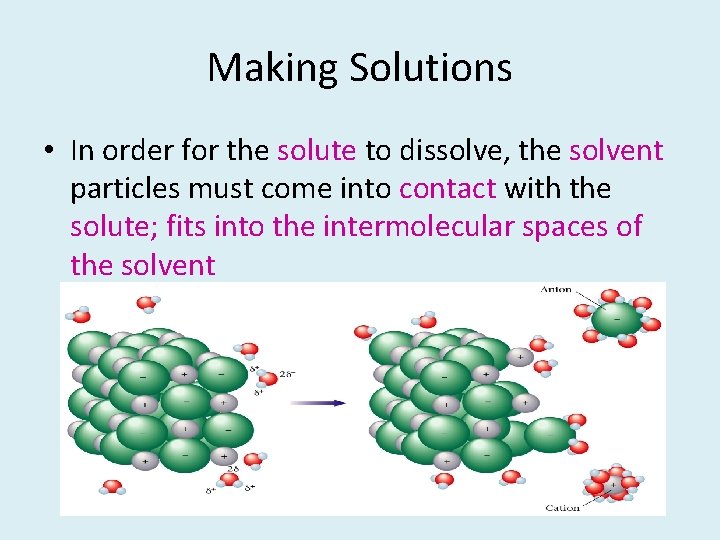
Making Solutions • In order for the solute to dissolve, the solvent particles must come into contact with the solute; fits into the intermolecular spaces of the solvent

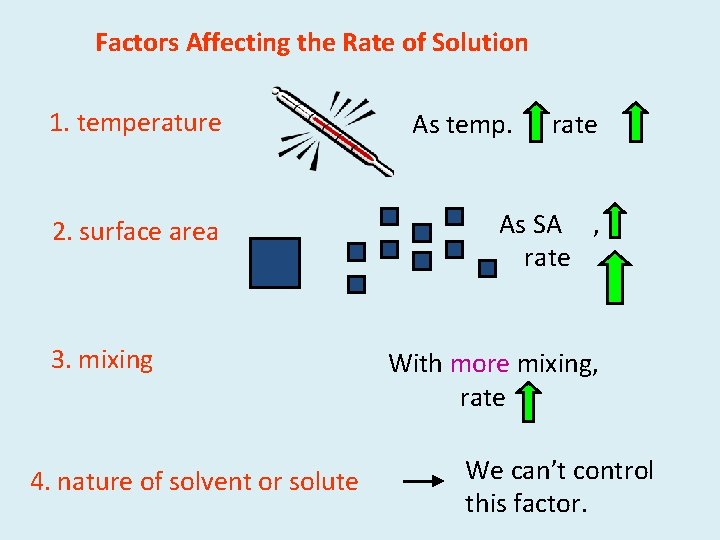
Factors Affecting the Rate of Solution 1. temperature As temp. , rate 2. surface area As SA , rate 3. mixing 4. nature of solvent or solute With more mixing, rate We can’t control this factor.

Factors Affecting Rate of Solubility • Temperature-the particles move around quicker and can interact with each other more frequently • Surface area-as more particles are exposed on the surface, they can interact with each other more • Mixing-stirring helps to disperse the particles with each other