Solutions Solubility Percent Concentration Colloids and Suspensions Solubility

Solutions Solubility Percent Concentration Colloids and Suspensions

Solubility The maximum amount of solute that can dissolve in a specific amount of solvent usually 100 g. g of solute 100 g water

Saturated and Unsaturated l A saturated solution contains the maximum amount of solute that can dissolve. Undissolved solute remains. l An unsaturated solution does not contain all the solute that could dissolve

Self test At 40 C, the solubility of KBr is 80 g/100 g H 2 O. Indicate if the following solutions are (1) saturated or (2) unsaturated A. ___60 g KBr in 100 g of water at 40 C B. ___200 g KBr in 200 g of water at 40 C C. ___25 KBr in 50 g of water at 40 C

Answer At 40 C, the solubility of KBr is 80 g/100 g H 2 O. Indicate if the following solutions are (1) saturated or (2) unsaturated A. 2 Less than 80 g/100 g H 2 O B. 1 Same as 100 g KBr in 100 g of water at 40 C, which is greater than its solubility C. 2 Same as 60 g KBr in 100 g of water, which is less than its solubility
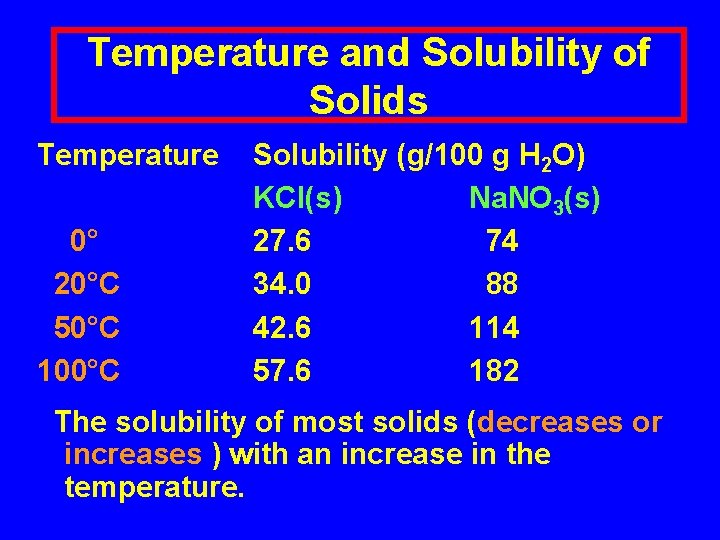
Temperature and Solubility of Solids Temperature 0° 20°C 50°C 100°C Solubility (g/100 g H 2 O) KCl(s) Na. NO 3(s) 27. 6 74 34. 0 88 42. 6 114 57. 6 182 The solubility of most solids (decreases or increases ) with an increase in the temperature.

Temperature and Solubility of Solids Temperature 0° 20°C 50°C 100°C Solubility (g/100 g H 2 O) KCl(s) Na. NO 3(s) 27. 6 74 34. 0 88 42. 6 114 57. 6 182 The solubility of most solids increases with an increase in the temperature.
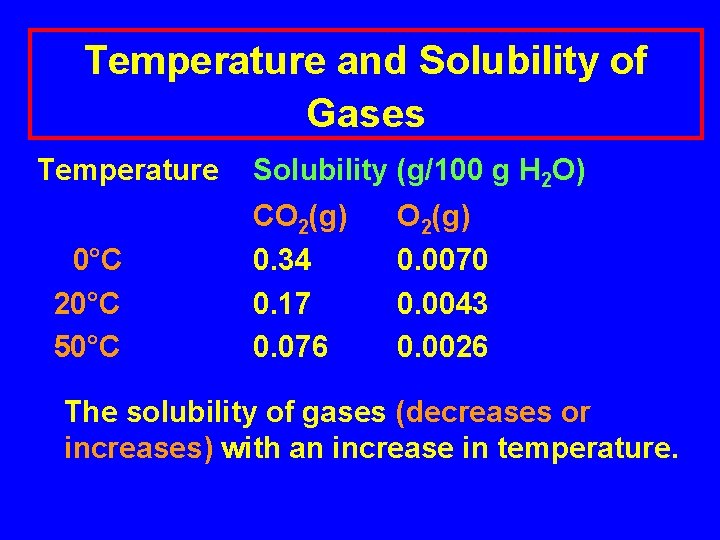
Temperature and Solubility of Gases Temperature 0°C 20°C 50°C Solubility (g/100 g H 2 O) CO 2(g) 0. 34 0. 17 0. 076 O 2(g) 0. 0070 0. 0043 0. 0026 The solubility of gases (decreases or increases) with an increase in temperature.
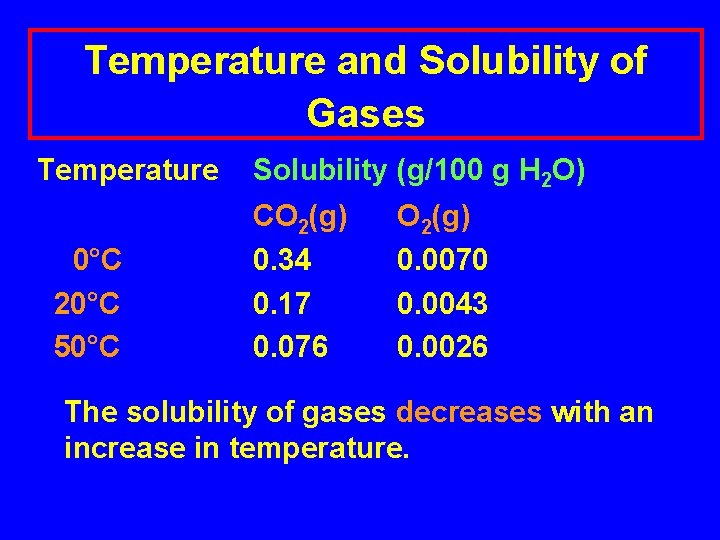
Temperature and Solubility of Gases Temperature 0°C 20°C 50°C Solubility (g/100 g H 2 O) CO 2(g) 0. 34 0. 17 0. 076 O 2(g) 0. 0070 0. 0043 0. 0026 The solubility of gases decreases with an increase in temperature.

Self test A. Why would a bottle of carbonated drink possibly burst (explode) when it is left out in the hot sun ? B. Why would fish die in water that gets too warm?

Answer A. Gas in the bottle builds up as the gas becomes less soluble in water at high temperatures, which may cause the bottle to explode. B. Because O 2 gas is less soluble in warm water, the fish may not obtain the needed amount of O 2 for their survival.

Soluble and Insoluble Salts A soluble salt is an ionic compound that dissolves in water. An insoluble salt is an ionic compound that does not dissolve in water

Rules of Solubility 1. A salt is soluble in water if it contains any one of the following positive ions: NH 4+ Li+ Na+ K+ Or this negative ion: NO 3 Examples: soluble salts Li. Cl Na 2 SO 4 KBr Ca(NO 3)2

Cl- Salts 2. Salts with Cl- are soluble, but not if the positive ion is Ag+, Pb 2+, or Hg 22+. Examples: soluble insoluble (will not dissolve) Mg. Cl 2 Ag. Cl Pb. Cl 2

SO 42 - Salts 3. Salts with SO 42 - are soluble, but not if the positive ion is Ba 2+, Pb 2+ or Ca 2+. Examples: soluble insoluble Mg. SO 4 Ba. SO 4 Pb. SO 4

Other Salts 4. Most salts containing CO 32 -, PO 43 -, S 2 and OH- are not soluble. Examples: soluble not soluble Na 2 CO 3 Ca. CO 3 K 2 S Cu. S

Self test Indicate if each salt is (1) soluble or (2) insoluble: A. ______ Na 2 SO 4 B. ______ Mg. CO 3 C. ______ Pb. Cl 2 D. ______ Mg. Cl 2

Answer Indicate if each salt is (1) soluble or (2) not soluble: A. _1_ Na 2 SO 4 B. _2_ Mg. CO 3 C. _2_ Pb. Cl 2 D. _1_ Mg. Cl 2
- Slides: 18