Solutions Mixing Mixing Mixture Physical combination Substances may

Solutions Mixing, Mixing!

Mixture Physical combination Substances may be physically separated Methods of separation: Boiling/evaporation Fractional crystalization Magnetism Electricity

Solution Homogeneous Single phase Soluble: capable of being dissolved Solvent: substance doing the dissolving Solute: substance(s) being dissolved
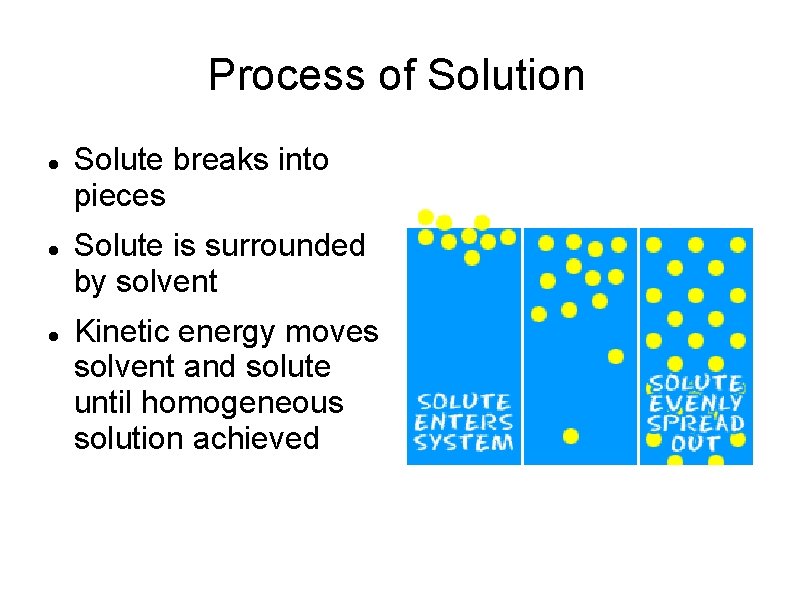
Process of Solution Solute breaks into pieces Solute is surrounded by solvent Kinetic energy moves solvent and solute until homogeneous solution achieved

Types of Solutions Solid, liquid, gas Table 13 -1 shows some combinations Common in chemistry labs: solid in liquid Generally: Like dissolves like! Particle size in solution is very small

Solute Particles Electrolyte vs nonelectrolyte Electrolyte: Conducts electricity in solution Ionic substance Dissociates when dissolved to form more than one solute particle Nonelectrolyte: Does not conduct Generally covalent substances

Suspension Heterogeneous mixture Larger particle size (over 1000 nm) Particles will settle out with time Particles can be separated from mixture by filtration

Colloid Mixture with homogeneous characteristics, but suspended particles Particle size between that of suspension and mixture (1 -1000 nm) Made of dispersing medium and dispersed phase Tyndall effect: scattering of light by dispersed phase

Tyndall Effect
- Slides: 9