Solutions COLLEGE CHEMISTRY Types of Solutions Remember a

Solutions COLLEGE CHEMISTRY

Types of Solutions �Remember, a solution is a homogenous mixture of two or more substances �Saturated solution – contains the maximum number of solute that will dissolve in a solvent at a specific temperature �Supersaturate solution – contains more solute than is present in a saturated solution Not stable Eventually the extra solute will come out through crystallization as crystals �Unsaturated solution – contains less solute that is present in a saturated solution
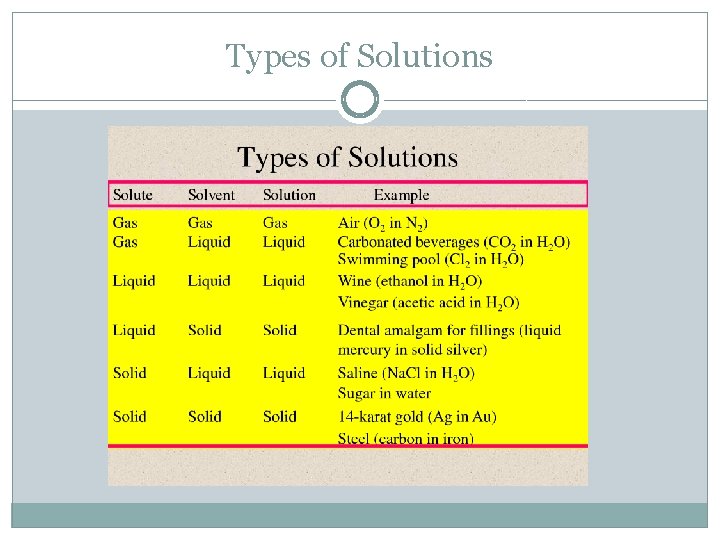
Types of Solutions

Molecular View of the Solution Process � the intermolecular attractions that hold molecules together in liquids in solids is also responsible for the formation of solutions �The ease of solution formation depends on these three types of interactions: 1. solvent-solvent interaction 2. solute-solute interaction 3. solvent-solute interaction

Molecular View of the Solution Process �For simplicity, we can think of the solution process as happening in three distinct steps � 1. separation of solvent molecules � 2. separation of solute molecules � 3. solvent and solute molecules mix �Process can be endothermic or exothermic DHsoln = DH 1 + DH 2 + DH 3
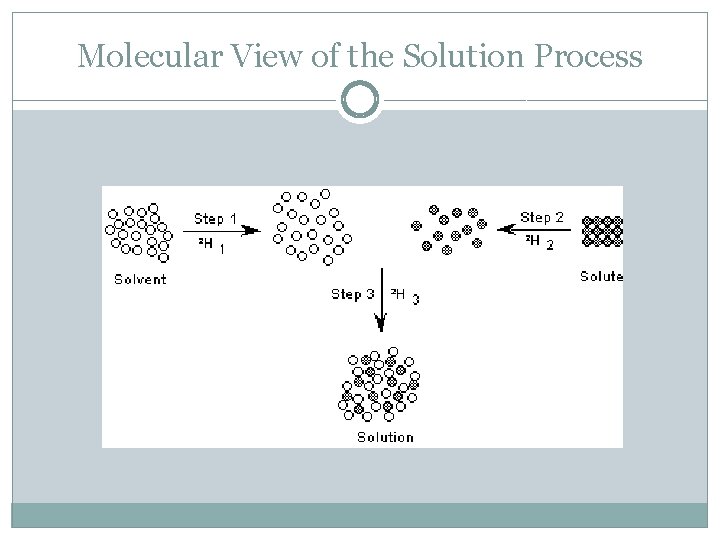
Molecular View of the Solution Process

Molecular View of the Solution Process �Solute-solvent interaction > solute-solute AND solvent-solvent Reaction is favorable and exothermic DHsoln < 0 �Solute-solvent interaction < solute-solute AND solvent-solvent Reaction isn’t favorable and is endothermic DHsoln > 0

Molecular View of the Solution Process �So if particles are more attractive to each other than the solute-solvent, why does dissociation take place? Two things govern dissociation � 1. energy (exothermic is more favorable) � 2. tendency towards disorder (entropy) � Molecules want to be in a state of disorder (solution), do not enjoy being in order (just solute or just solvent)

BP Oil Spill Predicting Solubility �“like dissolves like” help us determine what is soluble (or dissolves) in another molecule �non-polar solute will dissolve in non-polar solvents and polar solutes will dissolve in polar solvents This is why oil (non-polar) and water (polar) will not mix �Also, compounds with similar intermolecular forces will be solubility in each other Ex: CCl 4 (carbon tetrachloride) and C 6 H 6 (benzene) are both non-polar and have dispersion forces only fairly soluble Ex: alcohols (with –OH) dissolve in water

Predicting Solubility �Miscibility – ability of two liquids to be completely soluble in each other in all proportions Immiscible – not soluble �Solubility rules allows us to determine what ionic compounds will dissolve in water Generally, ionic compounds will dissolve in polar solvents and covalent compounds will dissolve only in nonpolar solvents �Covalent compounds lack a dipole moment and cannot effectively solvate an ionic compound (such as Na. Cl) Solvation – process in which an ion or molecule is surrounded by solvent molecules in a specific manner
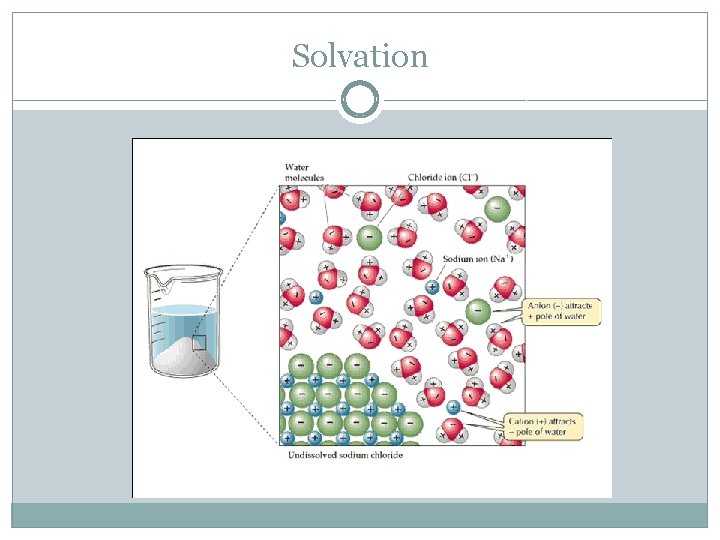
Solvation

Example 12. 1 �Predict the relative solubilities of the following cases: �(a) Bromine (Br 2) in benzene (C 6 H 6, m = 0 D) and in water (H 2 O, m = 1. 87 D) �(b) KCl in carbon tetrachloride (CCl 4, m = 0 D) and in liquid ammonia (NH 3, m = 1. 46 D) �(c) formaldehyde (CH 2 O) in carbon disulfide (CS 2, m = 0 D) and in water

Example 12. 1 �Predict the relative solubilities of the following cases: �(a) Bromine (Br 2) in benzene (C 6 H 6, m = 0 D) and in water (H 2 O, m = 1. 87 D) Br 2 is nonpolar as in benzene (dispersion forces), water is polar Br 2 should only dissolve in benzene �(b) KCl in carbon tetrachloride (CCl 4, m = 0 D) and in liquid ammonia (NH 3, m = 1. 46 D) KCl is ionic, more soluble in ammonia since it has a larger dipole �(c) formaldehyde (CH 2 O) in carbon disulfide (CS 2, m = 0 D) and in water Formaldehyde is polar, carbon disulfide is nonpolar, and water is polar Formaldehyde can also hydrogen bond with water and it is more soluble in water
- Slides: 13