Solutions Chemical Stewardship Be responsible in how you

Solutions

Chemical Stewardship • Be responsible in how you dispose of and use chemicals. • Chemical pollution can travel far – and harm organisms. Frog with three legs – it has mutated from chemical exposure.

Solutions • What the solute and the solvent are determines – whether a substance will dissolve. – how much will dissolve. • A substance dissolves faster if it is stirred or shaken. –The particles are made smaller. –The temperature is increased. Why?

Solution = Solute + Solvent • Solute - gets dissolved • Solvent - does the dissolving – Aqueous – Tincture – Amalgam – Organic • Polar • Non-polar (water) (alcohol) (mercury) Dental filling Nightmare on White Street Chem Matters, December 1996

Solution Definitions solution: a homogeneous mixture -- evenly mixed at the particle level -- e. g. , salt water alloy: a solid solution of metals -- e. g. , bronze = Cu + Sn; brass = Cu + Zn solvent: the substance that dissolves the solute water salt soluble: “will dissolve in” miscible: refers to two gases or two liquids that form a solution; more specific than “soluble” -- e. g. , food coloring and water

Types of Solutions Solute Solvent Solution gas air (nitrogen, oxygen, argon gases) humid air (water vapor in air) liquid carbonated drinks (CO 2 in water) vinegar (CH 3 COOH in water) salt water (Na. Cl in water) solid dental amalgam (Hg in Ag) sterling silver (Cu in Ag) Gaseous Solutions gas liquid Liquid Solutions gas liquid solid Solutions liquid solid Charles H. Corwin, Introductory Chemistry 2005, page 369

Factors Affecting the Rate of Dissolution 1. temperature 2. particle size 3. mixing 4. nature of solvent or solute As To , rate As size , rate More mixing, rate

Classes of Solutions aqueous solution: solvent = water = “the universal solvent” amalgam: solvent = Hg e. g. , dental amalgam tincture: solvent = alcohol e. g. , tincture of iodine (for cuts) organic solution: solvent contains carbon e. g. , gasoline, benzene, toluene, hexane
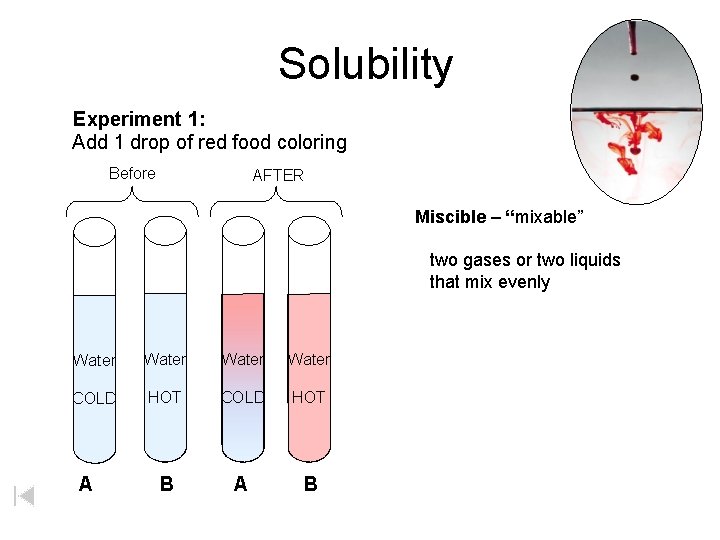
Solubility Experiment 1: Add 1 drop of red food coloring Before AFTER Miscible – “mixable” two gases or two liquids that mix evenly Water COLD HOT B A

Solubility Experiment 2: Add oil to water and shake AFTER Before Immiscible – “does not mix” two liquids or two gases that DO NOT MIX Oil Water T 0 sec T 30 sec
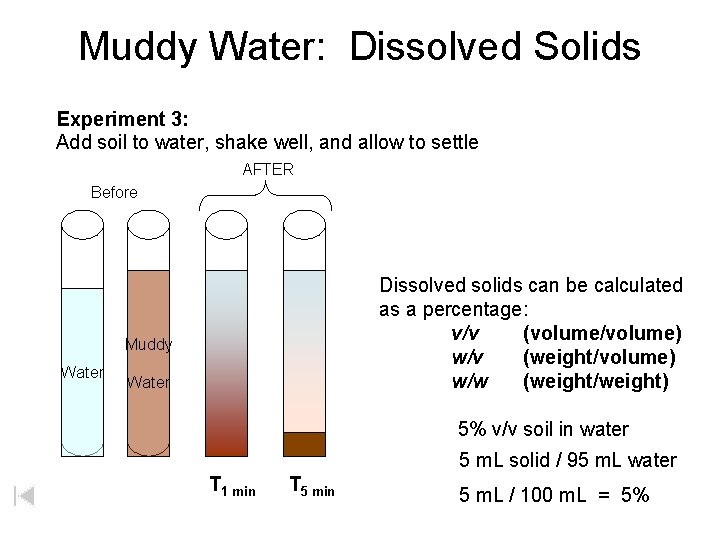
Muddy Water: Dissolved Solids Experiment 3: Add soil to water, shake well, and allow to settle AFTER Before Dissolved solids can be calculated as a percentage: v/v (volume/volume) w/v (weight/volume) w/w (weight/weight) Muddy Water 5% v/v soil in water 5 m. L solid / 95 m. L water T 1 min T 5 min 5 m. L / 100 m. L = 5%
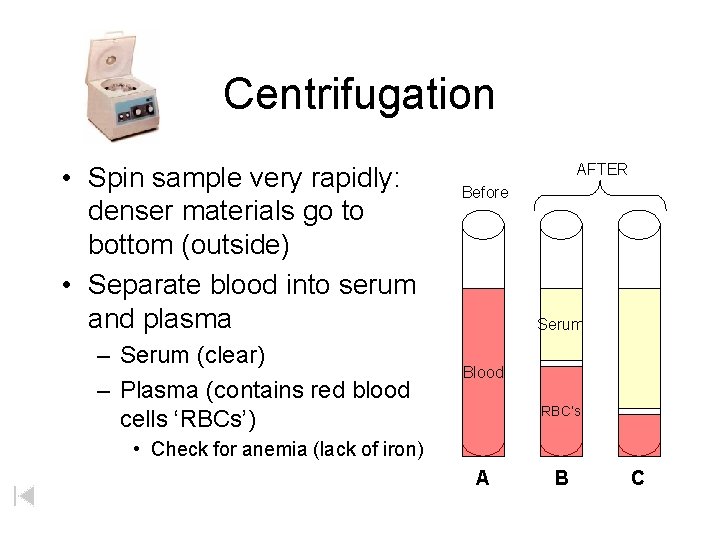
Centrifugation • Spin sample very rapidly: denser materials go to bottom (outside) • Separate blood into serum and plasma – Serum (clear) – Plasma (contains red blood cells ‘RBCs’) AFTER Before Serum Blood RBC’s • Check for anemia (lack of iron) A B C

Making solutions • In order to dissolve - the solvent molecules must come in contact with the solute. • Stirring moves fresh solvent next to the solute. • The solvent touches the surface of the solute. • Smaller pieces increase the amount of surface of the solute.
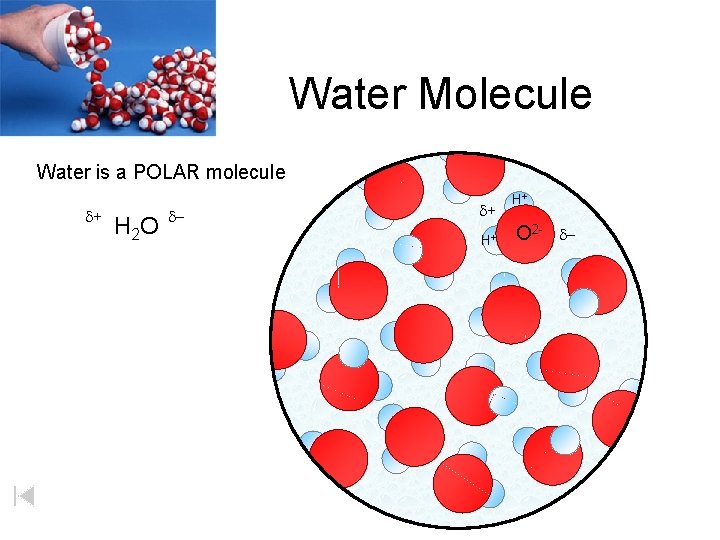
Water Molecule Water is a POLAR molecule d+ H 2 O d- d+ H+ H+ O 2 - d-

Water molecules “stick” together to create surface tension to support light weight objects. Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.

Water Molecule • What is a polar molecule? d. Hydrogen bond d+ • How does the polarity of water effect this molecule? H O H
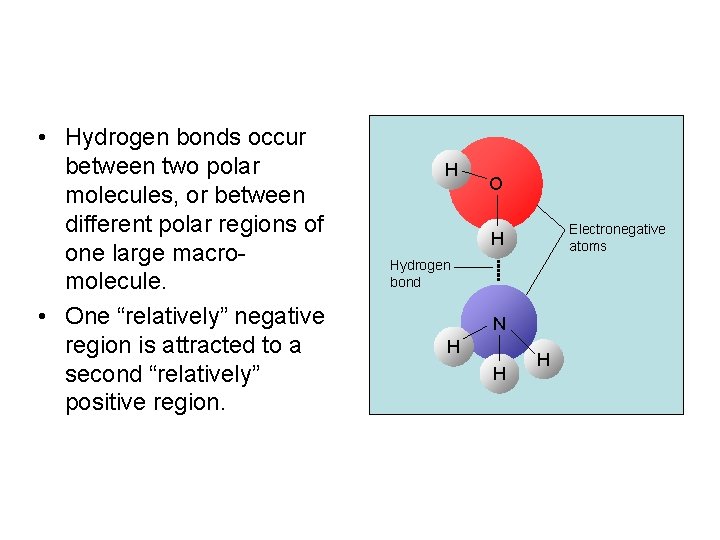
• Hydrogen bonds occur between two polar molecules, or between different polar regions of one large macromolecule. • One “relatively” negative region is attracted to a second “relatively” positive region. H O Electronegative atoms H Hydrogen bond N H H H
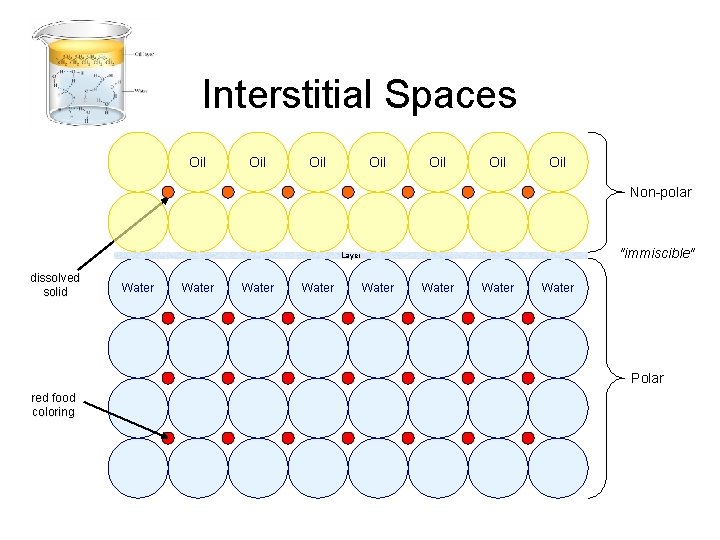
Interstitial Spaces Oil Oil Non-polar "immiscible" Layer dissolved solid Water Water Polar red food coloring
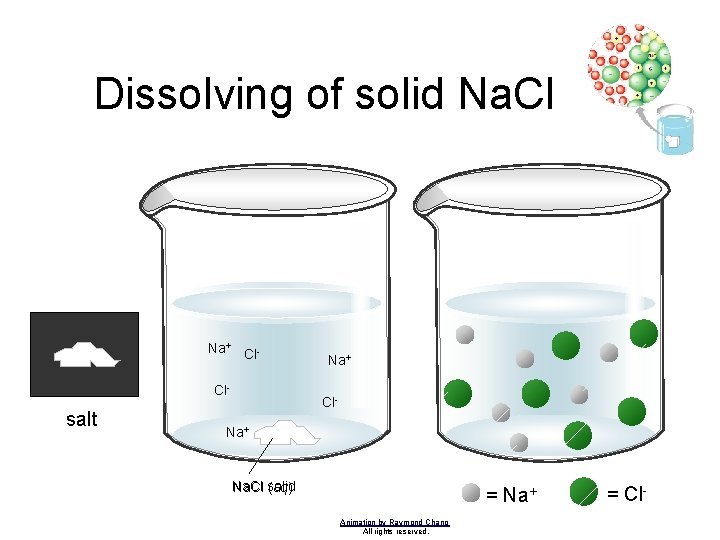
Dissolving of solid Na. Cl Na+ Cl. Cl- salt Na+ Cl- Na+ Na. Cl solid (aq) = Na+ Animation by Raymond Chang All rights reserved. = Cl-
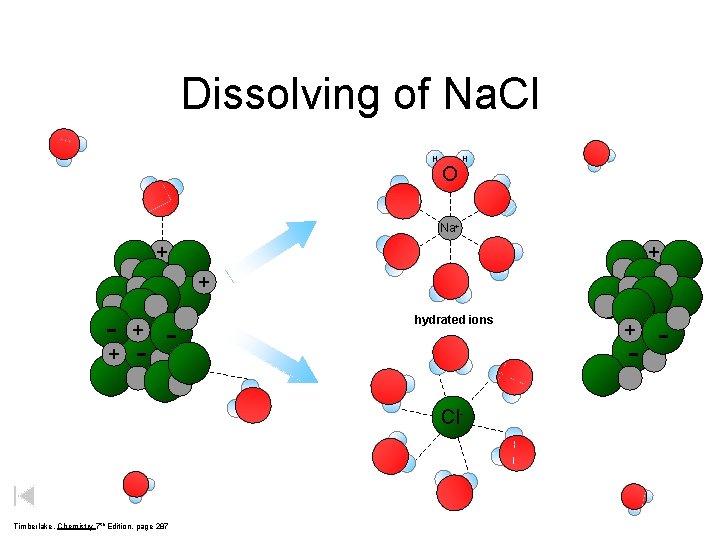
Dissolving of Na. Cl H O H Na+ + - - hydrated ions - Cl- Timberlake, Chemistry 7 th Edition, page 287 + -
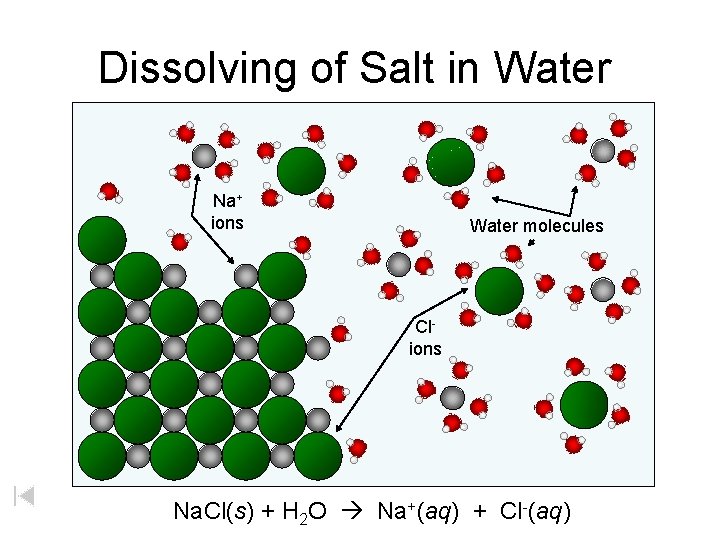
Dissolving of Salt in Water Na+ ions Water molecules Clions Na. Cl(s) + H 2 O Na+(aq) + Cl-(aq)
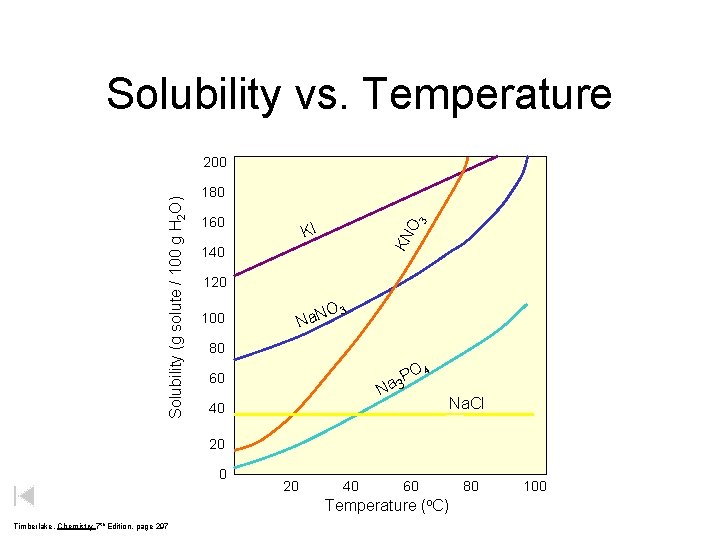
Solubility vs. Temperature 180 160 O 3 KI KN Solubility (g solute / 100 g H 2 O) 200 140 120 O Na. N 100 3 80 O 4 P a 3 60 N Na. Cl 40 20 40 60 Temperature (o. C) Timberlake, Chemistry 7 th Edition, page 297 80 100
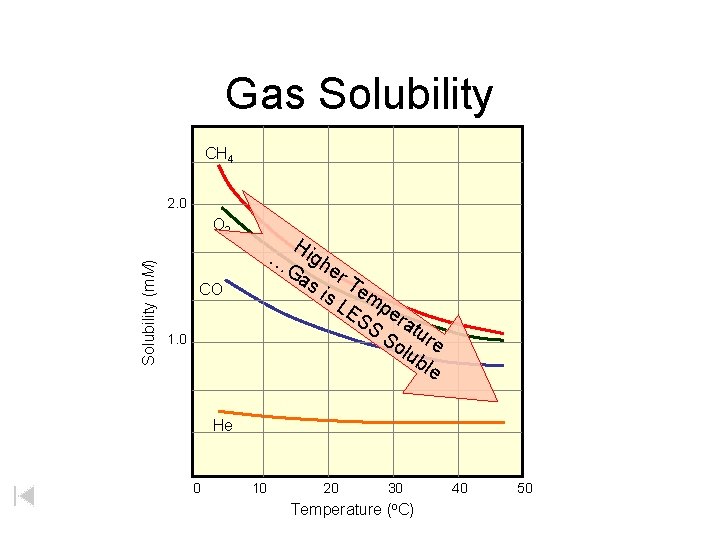
Gas Solubility CH 4 2. 0 Solubility (m. M) O 2 Hi g Ga her s i Te s. L m ES per S atu So re lub le … CO 1. 0 He 0 10 20 30 Temperature (o. C) 40 50

Electrolytes - solutions that carry an electric current strong electrolyte Na. Cl(aq) Na+ + Cl- Timberlake, Chemistry 7 th Edition, page 290 weak electrolyte HF(aq) H + + F- nonelectrolyte
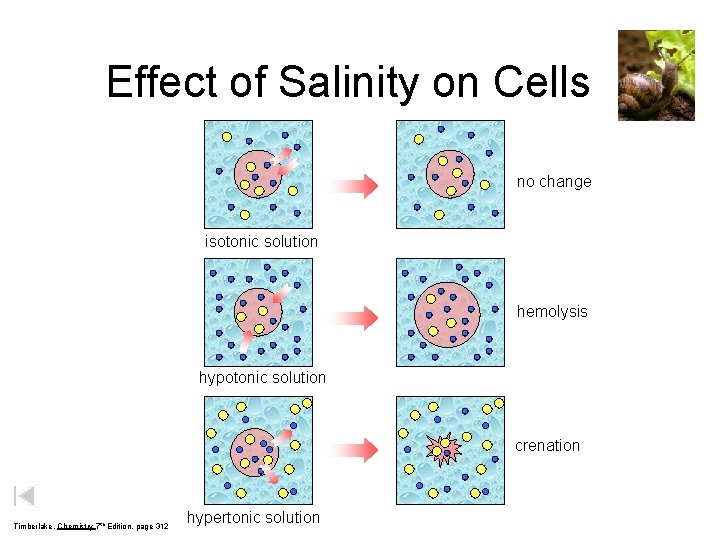
Effect of Salinity on Cells no change isotonic solution hemolysis hypotonic solution crenation Timberlake, Chemistry 7 th Edition, page 312 hypertonic solution
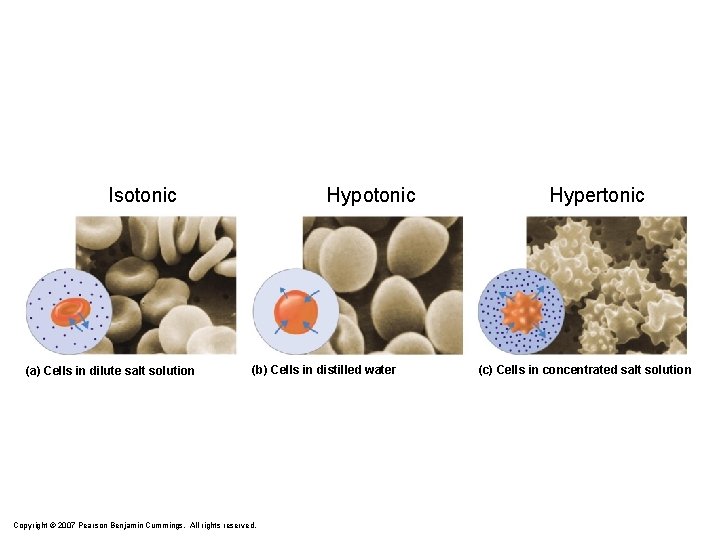
Isotonic (a) Cells in dilute salt solution Hypotonic (b) Cells in distilled water Copyright © 2007 Pearson Benjamin Cummings. All rights reserved. Hypertonic (c) Cells in concentrated salt solution
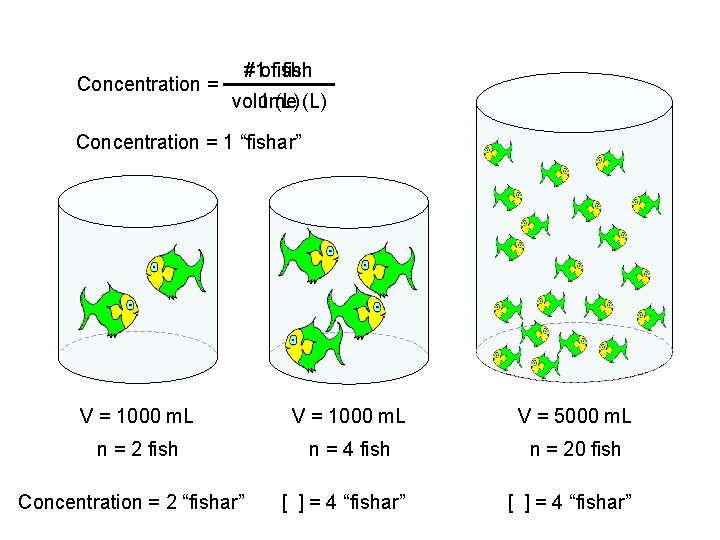
Concentration = #1 offish volume 1 (L) Concentration = 1 “fishar” V = 1000 m. L V = 5000 m. L n = 2 fish n = 4 fish n = 20 fish Concentration = 2 “fishar” [ ] = 4 “fishar”
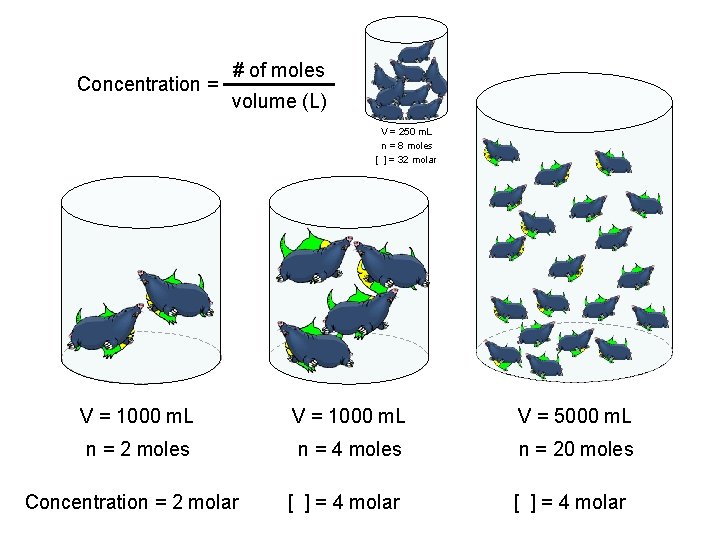
Concentration = # of moles volume (L) V = 250 m. L n = 8 moles [ ] = 32 molar V = 1000 m. L V = 5000 m. L n = 2 moles n = 4 moles n = 20 moles Concentration = 2 molar [ ] = 4 molar

Making Molar Solutions …from liquids (More accurately, from stock solutions)

Concentration…a measure of solute-to-solvent ratio concentrated “lots of solute” vs. dilute “not much solute” “watery” Add water to dilute a solution; boil water off to concentrate it.
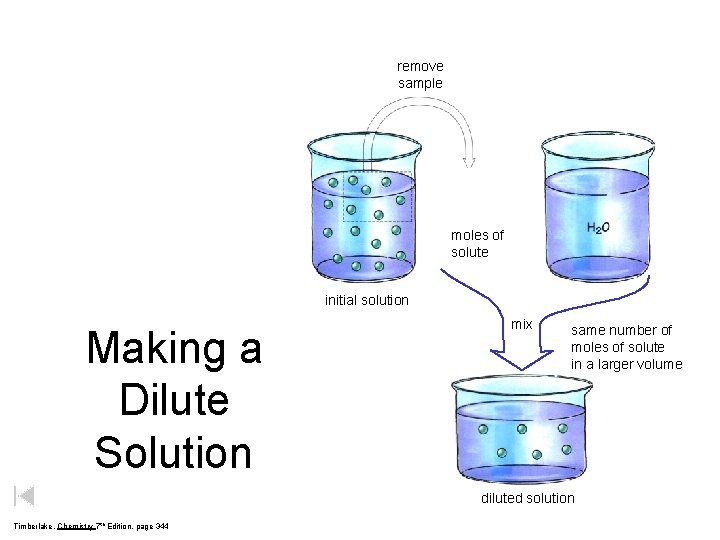
remove sample moles of solute initial solution Making a Dilute Solution mix same number of moles of solute in a larger volume diluted solution Timberlake, Chemistry 7 th Edition, page 344

Concentration “The amount of solute in a solution” A. mass % = mass of solute mass of sol’n % by mass – medicated creams % by volume – rubbing alcohol B. parts per million (ppm) also, ppb and ppt – commonly used for minerals or contaminants in water supplies C. molarity (M) = moles of solute L of sol’n mol – used most often in this class M = D. mol L molality (m) = moles of solute kg of solvent M L
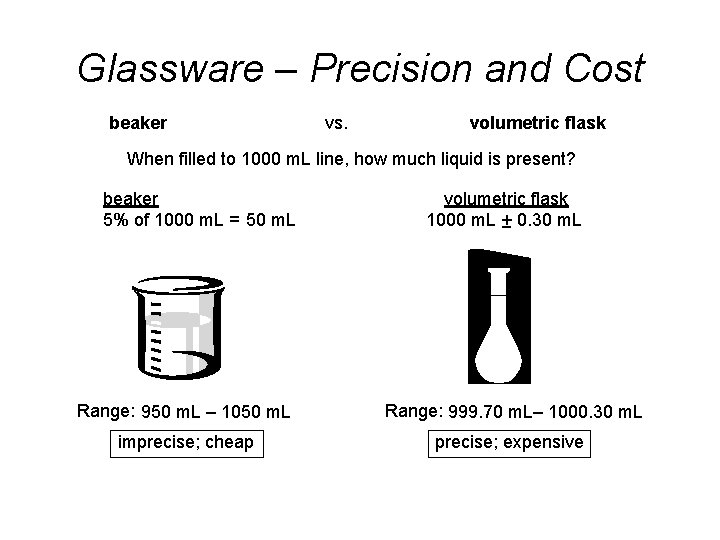
Glassware – Precision and Cost beaker vs. volumetric flask When filled to 1000 m. L line, how much liquid is present? beaker 5% of 1000 m. L = 50 m. L volumetric flask 1000 m. L + 0. 30 m. L Range: 950 m. L – 1050 m. L Range: 999. 70 m. L– 1000. 30 m. L imprecise; cheap precise; expensive

Markings on Glassware Beaker 500 m. L + 5% Range = 500 m. L + 25 m. L 475 – 525 m. L Graduated Cylinder 500 m. L + 5 m. L Range = 500 m. L + 5 m. L 495 – 505 m. L Volumetric Flask 500 m. L + 0. 2 m. L Range = 499. 8 – 500. 2 m. L TC 20 o. C “to contain at a temperature of 20 o. C” TD “to deliver” 22 s T “time in seconds”

How to mix solid chemicals Lets mix chemicals for the upcoming soap lab. We will need 1000 m. L of 3 M Na. OH per class. How much sodium hydroxide will I need, for five classes, for this lab? mol M = L ? mol 3 M = 1 L How much will this weigh? ? = 3 mol Na. OH/class x 5 classes 15 mol Na. OH 1 Na @ 23 g/mol + 1 O @ 16 g/mol + 1 H @ 1 g/mol MMNa. OH = 40 g/mol X g Na. OH = 15. 0 mol Na. OH 40. 0 g Na. OH = 600 g Na. OH 1 mol Na. OH FOR EACH CLASS: To mix this, add 120 g Na. OH into 1 L volumetric flask with ~750 m. L cold H 2 O. Mix, allow to return to room temperature – bring volume to 1 L.

How to mix a Standard Solution Wash bottle Volume marker (calibration mark) Weighed amount of solute Zumdahl, De. Coste, World of Chemistry 2002, page 480

Process of Making a Standard Solution from Liquids Zumdahl, De. Coste, World of Chemistry 2002, page 483
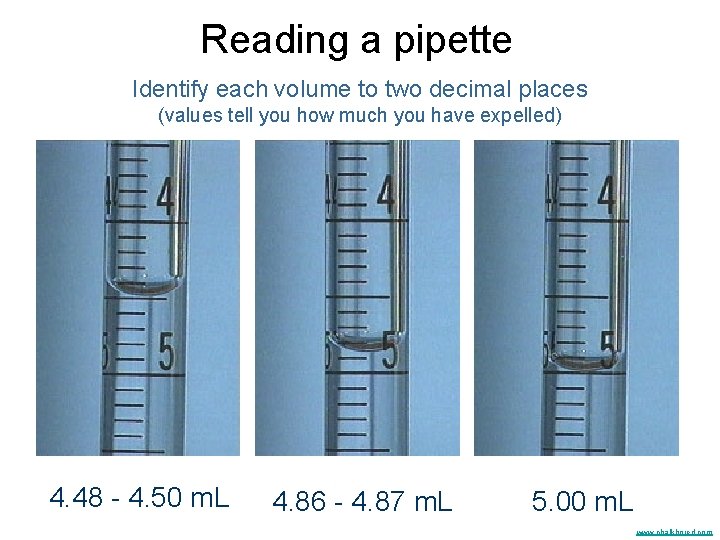
Reading a pipette Identify each volume to two decimal places (values tell you how much you have expelled) 4. 48 - 4. 50 m. L 4. 86 - 4. 87 m. L 5. 00 m. L www. chalkbored. com
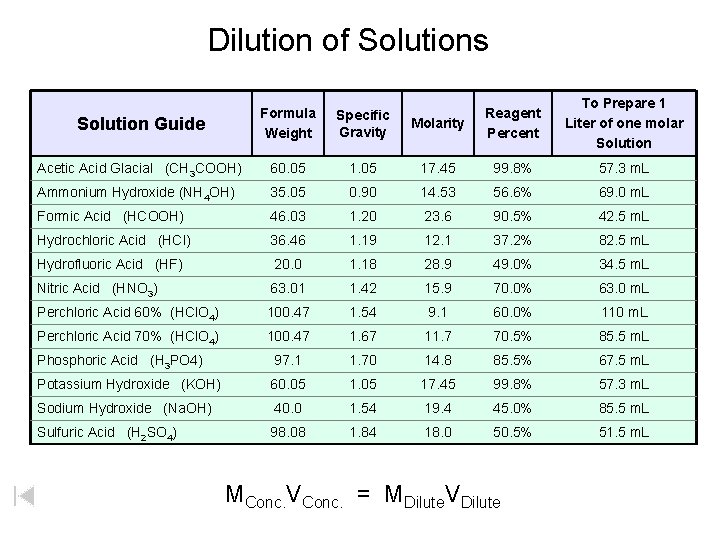
Dilution of Solutions Molarity Reagent Percent To Prepare 1 Liter of one molar Solution 1. 05 17. 45 99. 8% 57. 3 m. L 35. 05 0. 90 14. 53 56. 6% 69. 0 m. L Formic Acid (HCOOH) 46. 03 1. 20 23. 6 90. 5% 42. 5 m. L Hydrochloric Acid (HCl) 36. 46 1. 19 12. 1 37. 2% 82. 5 m. L Hydrofluoric Acid (HF) 20. 0 1. 18 28. 9 49. 0% 34. 5 m. L Nitric Acid (HNO 3) 63. 01 1. 42 15. 9 70. 0% 63. 0 m. L Perchloric Acid 60% (HCl. O 4) 100. 47 1. 54 9. 1 60. 0% 110 m. L Perchloric Acid 70% (HCl. O 4) 100. 47 1. 67 11. 7 70. 5% 85. 5 m. L Phosphoric Acid (H 3 PO 4) 97. 1 1. 70 14. 8 85. 5% 67. 5 m. L Potassium Hydroxide (KOH) 60. 05 17. 45 99. 8% 57. 3 m. L Sodium Hydroxide (Na. OH) 40. 0 1. 54 19. 4 45. 0% 85. 5 m. L Sulfuric Acid (H 2 SO 4) 98. 08 1. 84 18. 0 50. 5% 51. 5 m. L Solution Guide Formula Weight Specific Gravity Acetic Acid Glacial (CH 3 COOH) 60. 05 Ammonium Hydroxide (NH 4 OH) MConc. VConc. = MDilute. VDilute

Dilutions of Solutions Acids (and sometimes bases) are purchased in concentrated form (“concentrate”) and are easily diluted to any desired concentration. **Safety Tip: When diluting, add acid or base to water. ** Dilution Equation: C = concentrate D = dilute Concentrated H 3 PO 4 is 14. 8 M. What volume of concentrate is required to make 25. 00 L of 0. 500 M H 3 PO 4? VC = 0. 845 L = 845 m. L

How would you mix the above solution? 1. Measure out 0. 845 L of concentrated H 3 PO 4. 2. In separate container, obtain ~20 L of cold H 2 O. 3. In fume hood, slowly pour [H 3 PO 4] into cold H 2 O. 4. Add enough H 2 O until 25. 00 L of solution is obtained. Be sure to wear your safety glasses! glasses

You have 75 m. L of conc. HF (28. 9 M); you need 15. 0 L of 0. 100 M HF. Do you have enough to do the experiment? MCVC = MDVD 28. 9 M (0. 075 L) = 0. 100 M (15. 0 L) Yes; we’re OK. 2. 1675 mol HAVE > 1. 50 mol NEED

Dilution • Preparation of a desired solution by adding water to a concentrate. • Moles of solute remain the same.

Dilution • What volume of 15. 8 M HNO 3 is required to make 250 m. L of a 6. 0 M solution? GIVEN: M 1 = 15. 8 M V 1 = ? M 2 = 6. 0 M V 2 = 250 m. L WORK: M 1 V 1 = M 2 V 2 (15. 8 M) V 1 = (6. 0 M)(250 m. L) V 1 = 95 m. L of 15. 8 M HNO 3

Preparing Solutions How to prepare 500 m. L of 1. 54 M Na. Cl solution – mass 45. 0 g of Na. Cl – add water until total volume is 500 m. L – Shake to dissolve salt 500 m. L mark 45. 0 g Na. Cl solute 500 m. L volumetric flask

Preparing Solutions molality molarity 1. 54 m Na. Cl in 0. 500 kg of water 500 m. L of 1. 54 M Na. Cl – mass 45. 0 g of Na. Cl – add 0. 500 kg of water – mass 45. 0 g of Na. Cl – add water until total volume is 500 m. L water 45. 0 g Na. Cl 500 m. L mark 500 m. L volumetric flask
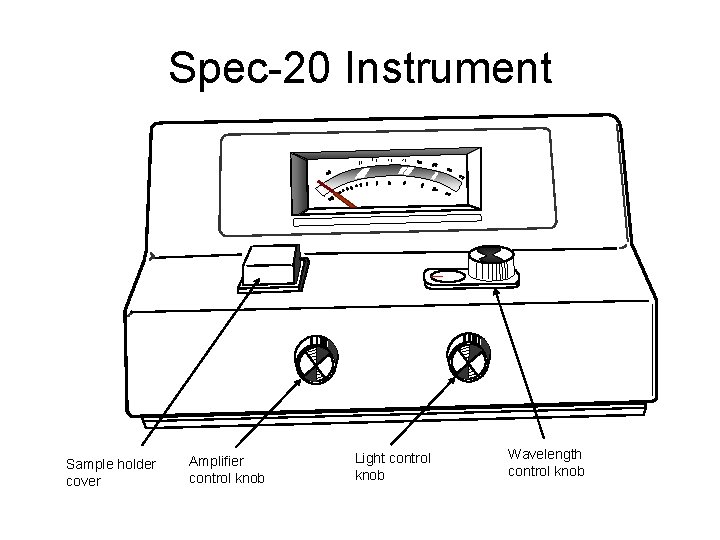
Spec-20 Instrument Sample holder cover Amplifier control knob Light control knob Wavelength control knob
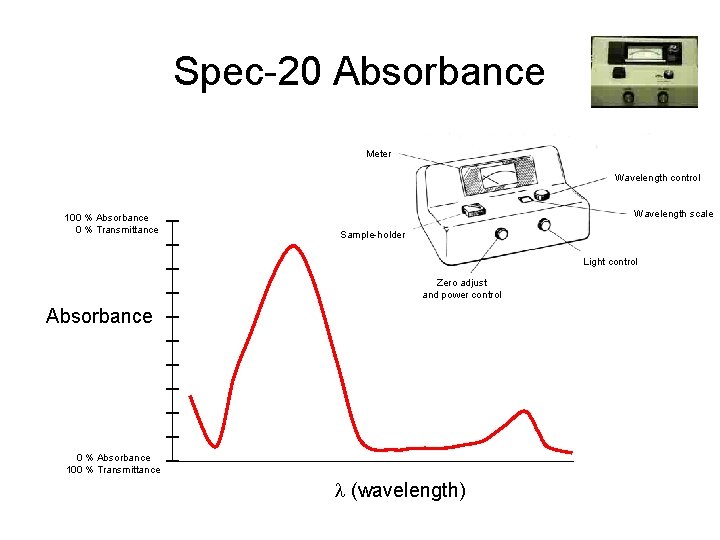
Spec-20 Absorbance Insert Photograph Spec-20 Meter Wavelength control 100 % Absorbance 0 % Transmittance Wavelength scale Sample-holder Light control Zero adjust and power control Absorbance 0 % Absorbance 100 % Transmittance l (wavelength)
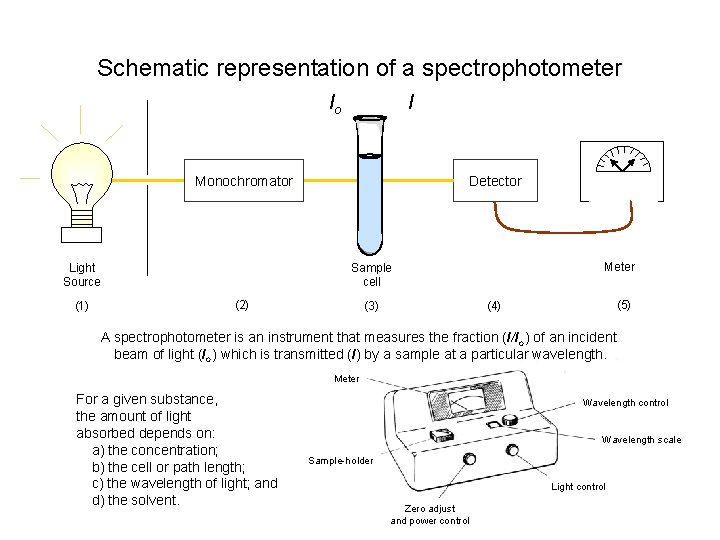
Schematic representation of a spectrophotometer I Io Monochromator Light Source (1) Detector Meter Sample cell (2) (5) (4) (3) A spectrophotometer is an instrument that measures the fraction (I/Io) of an incident beam of light (Io) which is transmitted (I) by a sample at a particular wavelength. Meter For a given substance, the amount of light absorbed depends on: a) the concentration; b) the cell or path length; c) the wavelength of light; and d) the solvent. Wavelength control Wavelength scale Sample-holder Light control Zero adjust and power control
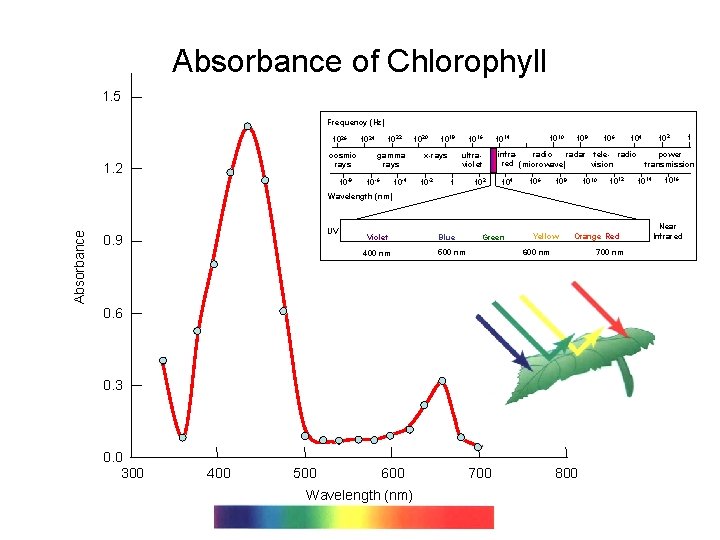
Absorbance of Chlorophyll 1. 5 Frequency (Hz) 1026 cosmic rays 1. 2 10 -8 1022 1024 gamma rays 10 -4 10 -6 1020 1018 ultraviolet x-rays 10 -2 1016 1 102 1010 1014 106 108 infraradio radar tele- radio red (microwave) vision 104 106 108 1010 1012 104 power transmission 1014 1016 Absorbance Wavelength (nm) UV 0. 9 Violet 400 nm Blue Green Yellow Orange Red 700 nm 600 nm 500 nm 0. 6 0. 3 0. 0 300 400 500 600 Wavelength (nm) 700 800 1 Near Infrared
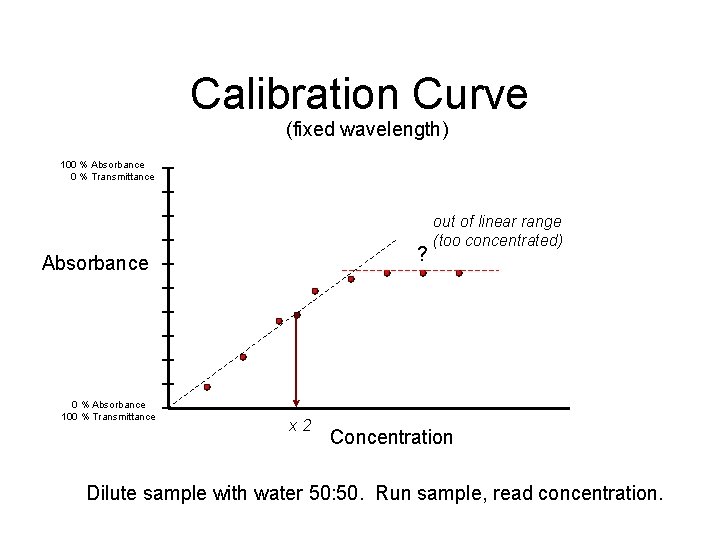
Calibration Curve (fixed wavelength) 100 % Absorbance 0 % Transmittance ? Absorbance 0 % Absorbance 100 % Transmittance x 2 out of linear range (too concentrated) Concentration Dilute sample with water 50: 50. Run sample, read concentration.

What mass of Ca. F 2 must be added to 1, 000 L of water so that fluoride atoms are present at a conc. of 1. 5 ppm? 23 m’cule 1000 m. L 1 g 1 mol 6. 02 x 10 X m’cule H 2 O = 1000 L = 3. 34 x 1028 m’cules H 2 O 1 L 1 m. L 18 g 1 mol 1. 5 atom F = 1, 000 m’cule H 2 O X = 5. 01 x 1022 atoms F times X atoms F 3. 34 x 1028 m’cule H 2 O 1 molecule Ca. F 2 = 2. 505 x 1022 molecules Ca. F 2 2 atoms F 1 mol Ca. F 2 78. 1 g Ca. F 2 X g Ca. F 2 = 2. 505 x 1022 molecules = 3. 25 g Ca. F 2 6. 02 x 1023 molecules 1 mol Ca. F 2
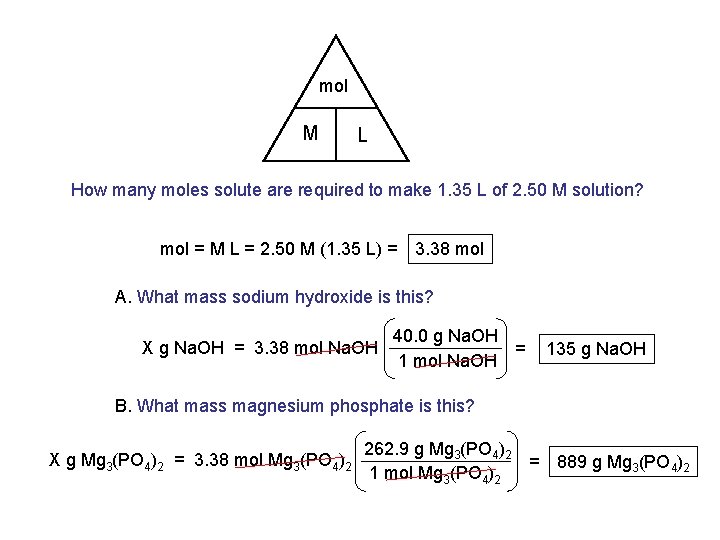
mol M L How many moles solute are required to make 1. 35 L of 2. 50 M solution? mol = M L = 2. 50 M (1. 35 L) = 3. 38 mol A. What mass sodium hydroxide is this? X g Na. OH = 3. 38 mol Na. OH 40. 0 g Na. OH = 1 mol Na. OH 135 g Na. OH B. What mass magnesium phosphate is this? X g Mg 3(PO 4)2 = 3. 38 mol Mg 3(PO 4)2 262. 9 g Mg 3(PO 4)2 = 889 g Mg 3(PO 4)2 1 mol Mg 3(PO 4)2
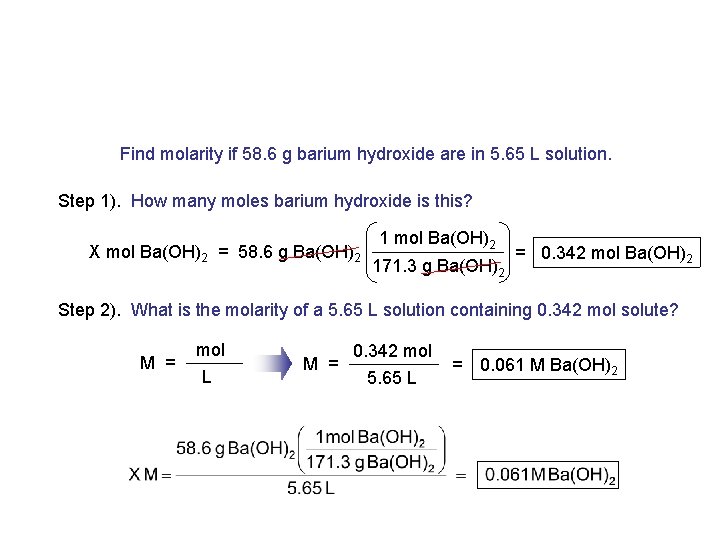
Find molarity if 58. 6 g barium hydroxide are in 5. 65 L solution. Step 1). How many moles barium hydroxide is this? X mol Ba(OH)2 = 58. 6 g Ba(OH)2 1 mol Ba(OH)2 171. 3 g Ba(OH)2 = 0. 342 mol Ba(OH)2 Step 2). What is the molarity of a 5. 65 L solution containing 0. 342 mol solute? M = mol L M = 0. 342 mol 5. 65 L = 0. 061 M Ba(OH)2

You have 10. 8 g potassium nitrate. How many m. L of solution will make this a 0. 14 M solution? convert to m. L

Dissociation occurs when neutral combinations of particles separate into ions while in aqueous solution. sodium chloride Na. Cl Na 1+ + Cl 1– sodium hydroxide Na. OH Na 1+ + OH 1– hydrochloric acid HCl H 1+ + Cl 1– sulfuric acid H 2 SO 4 2 H 1+ + SO 42– acetic acid CH 3 COOH CH 3 COO 1– + H 1+ In general, acids yield hydrogen (H? 1+) ions ? 1–) ions. in aqueous solution; bases yield hydroxide (OH
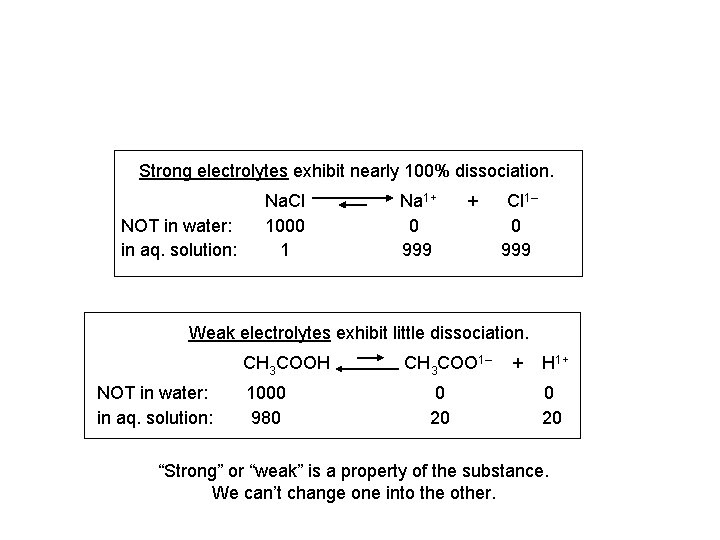
Strong electrolytes exhibit nearly 100% dissociation. NOT in water: in aq. solution: Na. Cl 1000 1 Na 1+ 0 999 + Cl 1– 0 999 Weak electrolytes exhibit little dissociation. CH 3 COOH NOT in water: in aq. solution: 1000 980 CH 3 COO 1– 0 20 + H 1+ 0 20 “Strong” or “weak” is a property of the substance. We can’t change one into the other.

electrolytes: solutes that dissociate in solution -- conduct electric current because of free-moving ions e. g. , acids, bases, most ionic compounds -- are crucial for many cellular processes -- obtained in a healthy diet -- For sustained exercise or a bout of the flu, sports drinks ensure adequate electrolytes. nonelectrolytes: solutes that DO NOT dissociate -- DO NOT conduct electric current (not enough ions) e. g. , any type of sugar

Colligative Properties depend on concentration of a solution Compared to solvent’s… a solution w/that solvent has a… …normal freezing point (NFP) …lower FP …normal boiling point (NBP) …higher BP FREEZING PT. DEPRESSION BOILING PT. ELEVATION
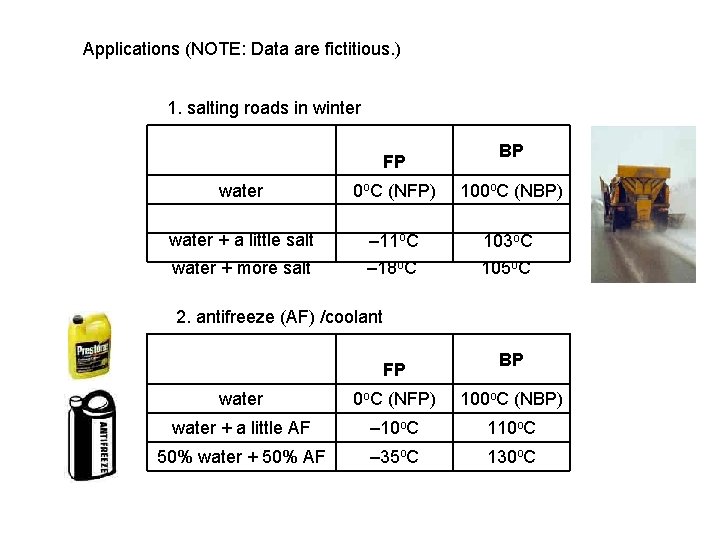
Applications (NOTE: Data are fictitious. ) 1. salting roads in winter FP BP water 0 o. C (NFP) 100 o. C (NBP) water + a little salt – 11 o. C water + more salt – 18 o. C 103 o. C 105 o. C 2. antifreeze (AF) /coolant FP BP water 0 o. C (NFP) 100 o. C (NBP) water + a little AF – 10 o. C 110 o. C 50% water + 50% AF – 35 o. C 130 o. C
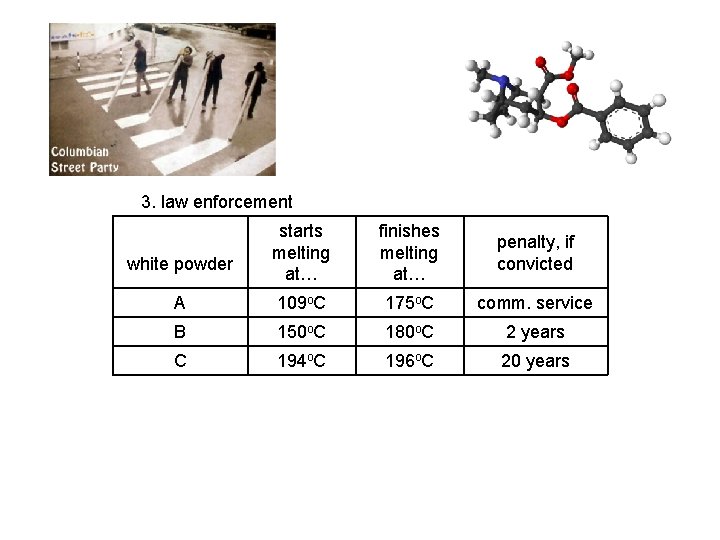
3. law enforcement starts melting at… finishes melting at… penalty, if convicted A 109 o. C 175 o. C comm. service B 150 o. C 180 o. C 2 years C 194 o. C 196 o. C 20 years white powder
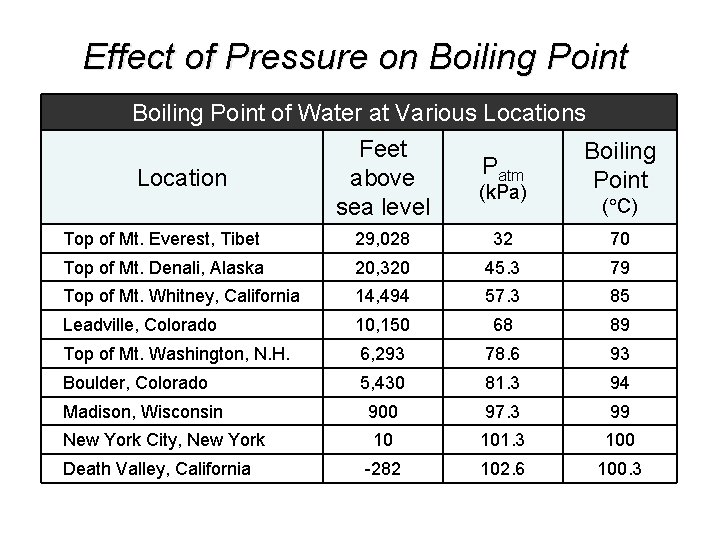
Effect of Pressure on Boiling Point of Water at Various Locations Feet Boiling Patm Location above Point (k. Pa) ( C) sea level Top of Mt. Everest, Tibet 29, 028 32 70 Top of Mt. Denali, Alaska 20, 320 45. 3 79 Top of Mt. Whitney, California 14, 494 57. 3 85 Leadville, Colorado 10, 150 68 89 Top of Mt. Washington, N. H. 6, 293 78. 6 93 Boulder, Colorado 5, 430 81. 3 94 Madison, Wisconsin 900 97. 3 99 New York City, New York 10 101. 3 100 -282 102. 6 100. 3 Death Valley, California

Calculations for Colligative Properties The change in FP or BP is found using… DTx = Kx m i DTx = change in To (below NFP or above NBP) Kx = constant depending on… (A) solvent (B) freezing or boiling m = molality of solute = mol solute / kg solvent i = integer that accounts for any solute dissociation any sugar (all nonelectrolytes)……………. . . i = 1 table salt, Na. Cl Na 1+ + Cl 1–………………i = 2 barium bromide, Ba. Br 2 Ba 2+ + 2 Br 1–……i = 3

Freezing Point Depression DTf = Kf m i Boiling Point Elevation DTb = Kb m i Then use these in conjunction with the NFP and NBP to find the FP and BP of the mixture. (Kf = cryoscopic constant, which is 1. 86 K kg/mol for the freezing point of water) (Kb = ebullioscopic constant, which is 0. 51 K kg/mol for the boiling point of water)

168 g glucose (C 6 H 12 O 6) are mixed w/2. 50 kg H 2 O. Find BP and FP of mixture. For H 2 O, Kb = 0. 512, Kf = – 1. 86. i=1 (NONELECTROLYTE) DTb = Kb m i = 0. 512 (0. 373) (1) = 0. 19 o. C BP = (100 + 0. 19)o. C = 100. 19 o. C DTf = Kf m i = – 1. 86 (0. 373) (1) = – 0. 69 o. C FP = (0 + – 0. 69)o. C = – 0. 69 o. C

168 g cesium bromide are mixed w/2. 50 kg H 2 O. Find BP and FP of mixture. For H 2 O, Kb = 0. 512, Kf = – 1. 86. Cs 1+ Br 1– i=2 Cs. Br Cs 1+ + Br 1– DTb = Kb m i = 0. 512 (0. 316) (2) = 0. 32 o. C BP = (100 + 0. 32)o. C = 100. 32 o. C DTf = Kf m i = – 1. 86 (0. 316) (2) = – 1. 18 o. C FP = (0 + – 1. 18)o. C = – 1. 18 o. C
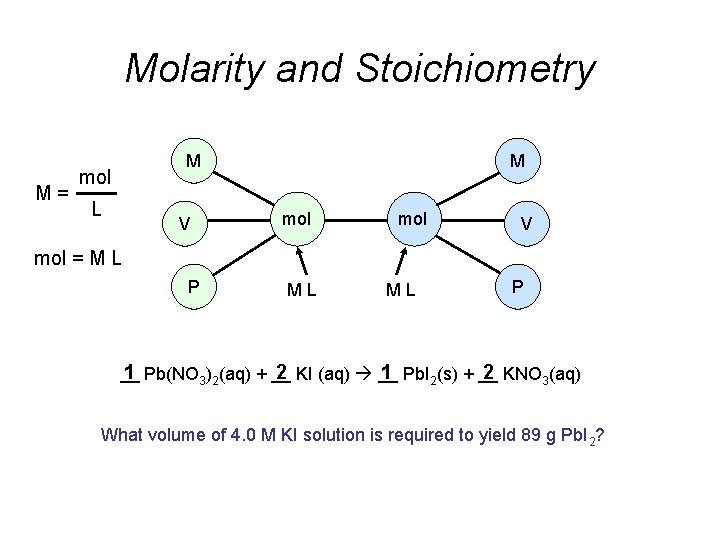
Molarity and Stoichiometry M= M mol L V M mol V mol = M L P ML ML P __ 1 Pb(NO 3)2(aq) + __ 2 KI (aq) __ 1 Pb. I 2(s) + __ 2 KNO 3(aq) What volume of 4. 0 M KI solution is required to yield 89 g Pb. I 2?

Stoichiometry for Reactions in Solution Step 1) Identify the species present in the combined solution, and determine what reaction occurs. Step 2) Write the balanced net ionic equation for the reaction. Step 3) Calculate the moles of reactants. Step 4) Determine which reactant is limiting. Step 5) Calculate the moles of product or products, as required. Step 6) Convert to grams or other units, as required. Stoichiometry steps for reactions in solution

1 Pb(NO 3)2(aq) + 2 KI (aq) 1 Pb. I 2(s) + 2 KNO 3(aq) ? L 4. 0 M 89 g What volume of 4. 0 M KI solution is required to yield 89 g Pb. I 2? Strategy: (1) Find mol KI needed to yield 89 g Pb. I 2. (2) Based on (1), find volume of 4. 0 M KI solution. X mol KI = 89 g Pb. I 2 M= mol L L= 1 mol Pb. I 2 2 mol KI 461 g Pb. I 2 1 mol Pb. I 2 = 0. 39 mol KI mol 0. 39 mol KI = 0. 098 L of 4. 0 M KI = M 4. 0 M KI

How many m. L of a 0. 500 M Cu. SO 4 solution will react w/excess Al to produce 11. 0 g Cu? Al 3+ SO 42– __Cu. SO 4(aq) + __Al (s) __Cu(s) + __Al 2(SO 4)3(aq) 3 Cu. SO 4(aq) + 2 Al (s) 3 Cu(s) + 1 Al 2(SO 4)3(aq) x mol 11 g X mol Cu. SO 4 = 11 g Cu mol M = L 1 mol Cu 63. 5 g Cu mol L = M 3 mol Cu. SO 4 = 0. 173 mol Cu. SO 4 3 mol Cu 0. 173 mol Cu. SO 4 0. 500 M Cu. SO 4 0. 346 L 1000 m. L = 346 m. L 1 L = 0. 346 L

Stoichiometry Problems • How many grams of Cu are required to react with 1. 5 L of 0. 10 M Ag. NO 3? Cu + 2 Ag. NO 3 2 Ag + ? g 1. 5 L Cu(NO 3)2 1. 5 L 0. 10 M . 10 mol Ag. NO 3 1 mol Cu 1 L 2 mol Ag. NO 3 63. 55 g Cu = 4. 8 g 1 mol Cu Cu Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Limiting Reactants • 79. 1 g of zinc react with 0. 90 L of 2. 5 M HCl. Identify the limiting and excess reactants. How many liters of hydrogen are formed at STP? Zn + 2 HCl 79. 1 g 0. 90 L 2. 5 M Zn. Cl 2 + H 2 ? L Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Limiting Reactants Zn + 2 HCl 79. 1 g 0. 90 L 2. 5 M 79. 1 g Zn Zn. Cl 2 + H 2 ? L 1 mol Zn 1 mol H 2 22. 4 L H 2 65. 39 g Zn 1 mol H 2 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem = 27. 1 L H 2
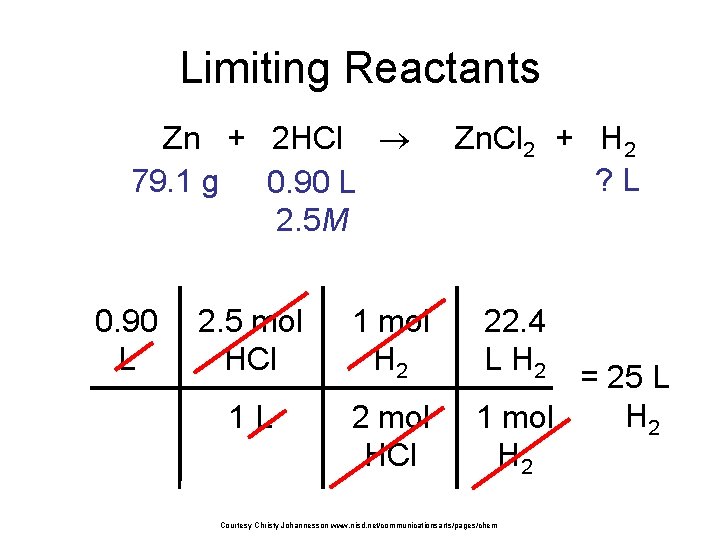
Limiting Reactants Zn + 2 HCl 79. 1 g 0. 90 L 2. 5 M 0. 90 L 2. 5 mol HCl 1 mol H 2 1 L 2 mol HCl Zn. Cl 2 + H 2 ? L 22. 4 L H 2 = 25 L H 2 1 mol H 2 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
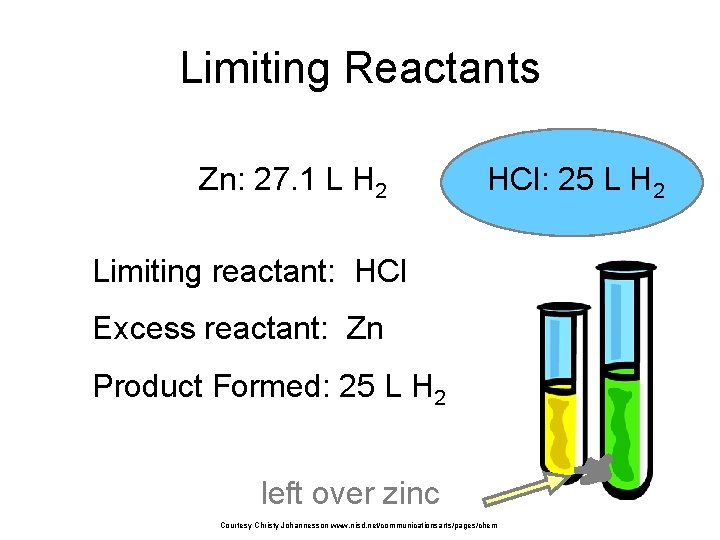
Limiting Reactants Zn: 27. 1 L H 2 HCl: 25 L H 2 Limiting reactant: HCl Excess reactant: Zn Product Formed: 25 L H 2 left over zinc Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
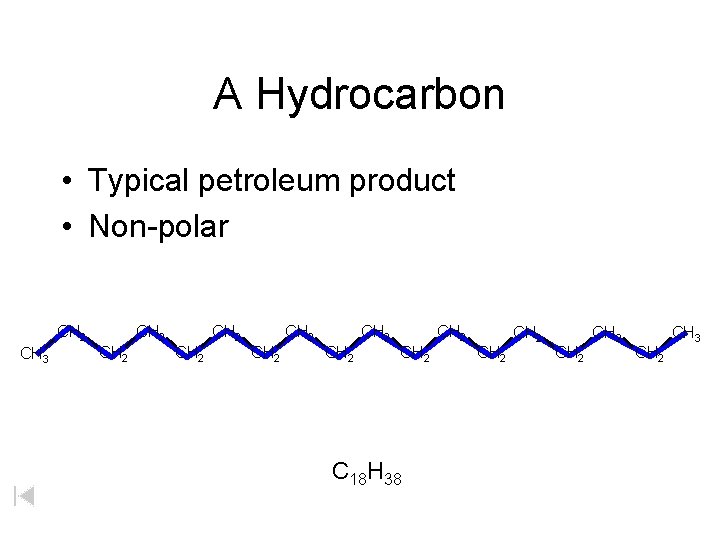
A Hydrocarbon • Typical petroleum product • Non-polar CH 2 CH 3 CH 2 CH 2 CH 2 C 18 H 38 CH 2 CH 2 CH 3
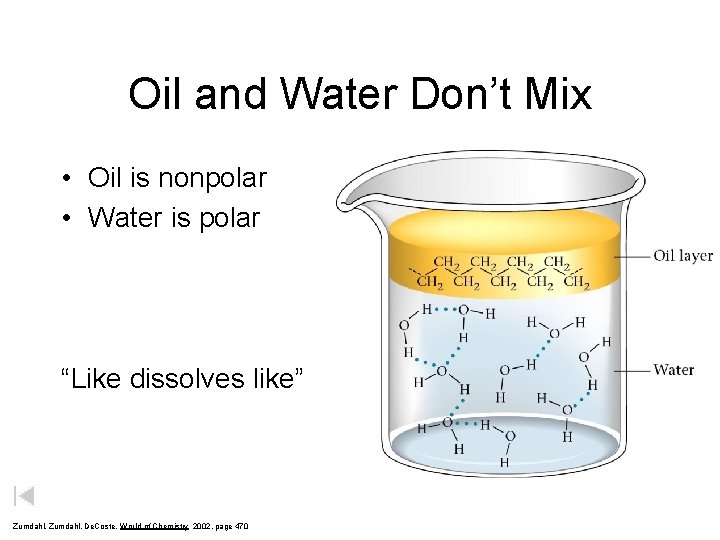
Oil and Water Don’t Mix • Oil is nonpolar • Water is polar “Like dissolves like” Zumdahl, De. Coste, World of Chemistry 2002, page 470
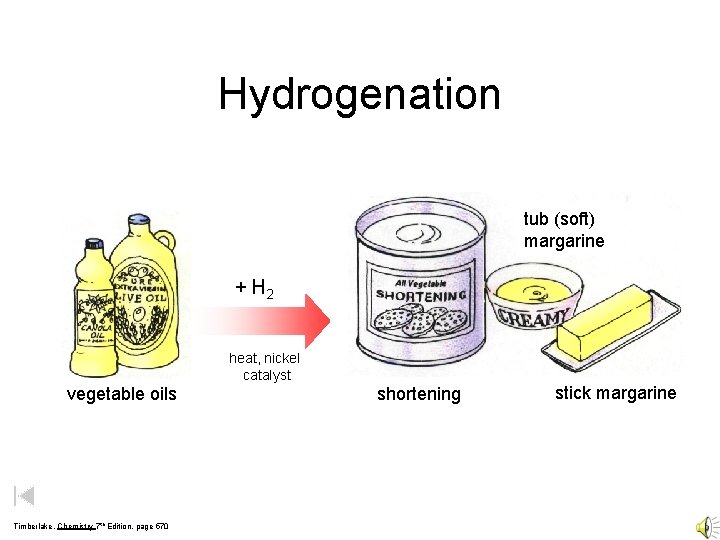
Hydrogenation tub (soft) margarine + H 2 heat, nickel catalyst vegetable oils Timberlake, Chemistry 7 th Edition, page 570 shortening stick margarine

Molecular Polarity H nonpolar molecules: -- e– are shared equally -- tend to be symmetric e. g. , fats and oils H H O polar molecules: -- e– NOT shared equally e. g. , water “Like dissolves like. ” like polar + polar = solution nonpolar + nonpolar = solution polar + nonpolar = suspension (won’t mix evenly) H–C–H H

Using Solubility Principles Chemicals used by body obey solubility principles. -- water-soluble vitamins: e. g. , vit. C -- fat-soluble vitamins: e. g. , vits. A, D Dry cleaning employs nonpolar liquids. -- polar liquids damage wool, silk -- also, dry clean for stubborn stains (ink, rust, grease) -- (tetra) perchloroethylene is in common use Cl Cl C=C Cl Cl
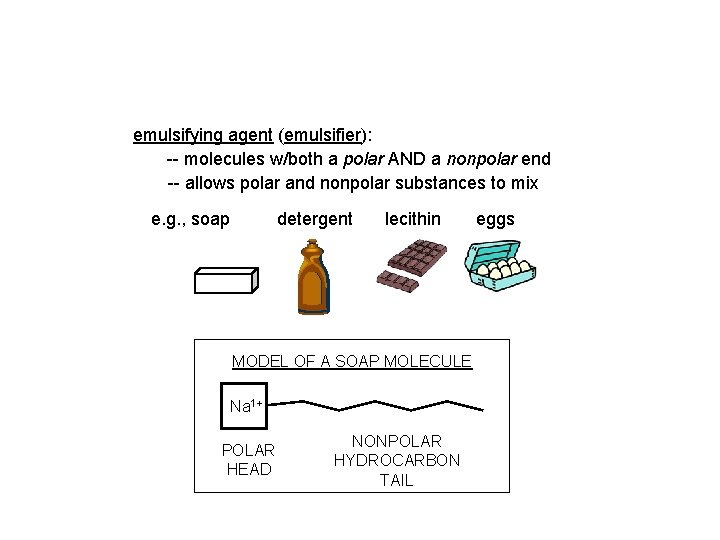
emulsifying agent (emulsifier): -- molecules w/both a polar AND a nonpolar end -- allows polar and nonpolar substances to mix e. g. , soap detergent lecithin MODEL OF A SOAP MOLECULE Na 1+ POLAR HEAD NONPOLAR HYDROCARBON TAIL eggs

Saponification Process of making soap from animal fat or vegetable oil using a base. O CH 2 – O – C – (CH 2)14 CH 3 CH 2 – OH O O CH – O – C – (CH 2)14 CH 3 + 3 Na. OH CH – OH + 3 Na+ -OC – (CH 2)14 CH 3 O CH 2 – O – C – (CH 2)14 CH 3 glyceryl tripalmitate (tripalmitin) CH 2 – OH sodium hydroxide glycerol 3 sodium palmitate (soap)
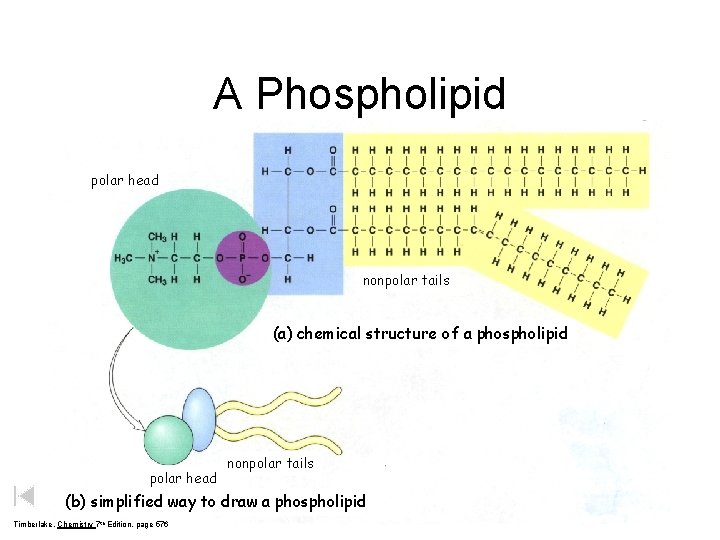
A Phospholipid polar head nonpolar tails (a) chemical structure of a phospholipid polar head nonpolar tails (b) simplified way to draw a phospholipid Timberlake, Chemistry 7 th Edition, page 576

A Model of a Cell Membrane Polar Nonpolar Cholesterol Timberlake, Chemistry 7 th Edition, page 587 Proteins Phospholipid bilayer

Cleaning Action of Soap Micelle Timberlake, Chemistry 7 th Edition, page 573

SOAP -- made from animal and vegetable fats vs. DETERGENT -- made from petroleum -- works better in hard water Hard water contains minerals w/ions like Ca 2+, Mg 2+, and Fe 3+ that replace Na 1+ at polar end of soap molecule. Soap is changed into an insoluble precipitate (i. e. , soap scum). micelle: a liquid droplet covered w/soap or detergent molecules

Solvation “Like Dissolves Like” NONPOLAR

Solvation u Soap / Detergent • polar “head” with long nonpolar “tail” • dissolves nonpolar grease in polar water micelle
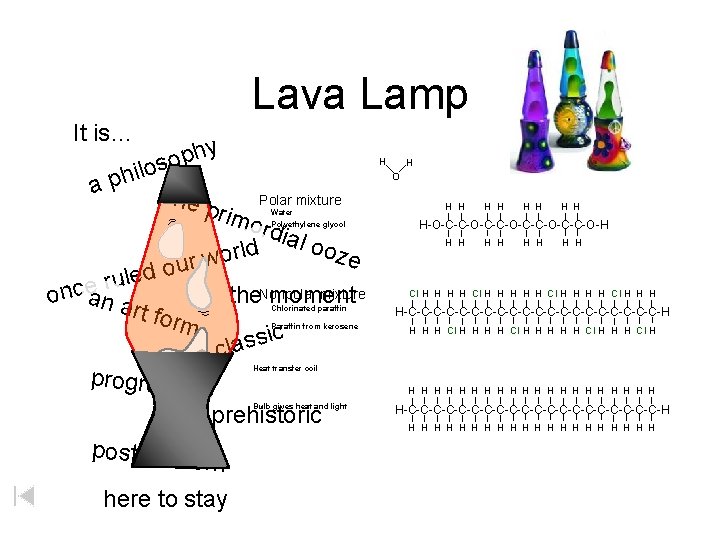
Lava Lamp It is… hy p o s o l i h ap the prim Polar mixture ordi al o d oze l r o w r u o d e l ru mixture the. Nonpolar moment once an a rt fo rm sic s a l ac progressi ve prehistoric post-mode rn here to stay Water Polyethylene glycol Chlorinated paraffin Paraffin from kerosene H H O H H H H H-O-C-C-O-C-C-O-H H H H H Cl H H H H-C-C-C-C-C-C-C-C-C-C-H H H H Cl H H H Cl H Heat transfer coil Bulb gives heat and light H H H H H H-C-C-C-C-C-C-C-C-C-C-H H H H H H
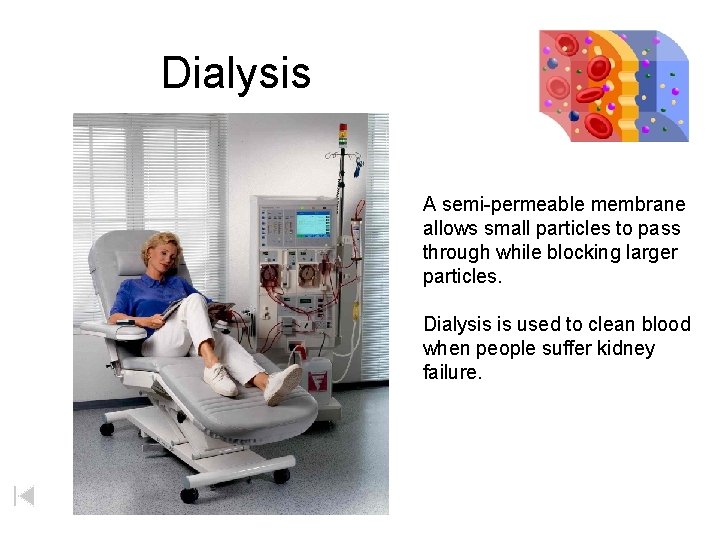
Dialysis A semi-permeable membrane allows small particles to pass through while blocking larger particles. Dialysis is used to clean blood when people suffer kidney failure.
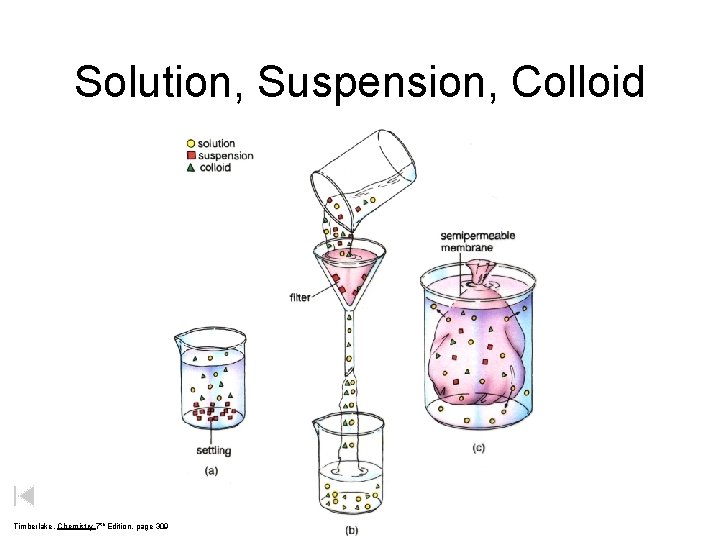
Solution, Suspension, Colloid Timberlake, Chemistry 7 th Edition, page 309
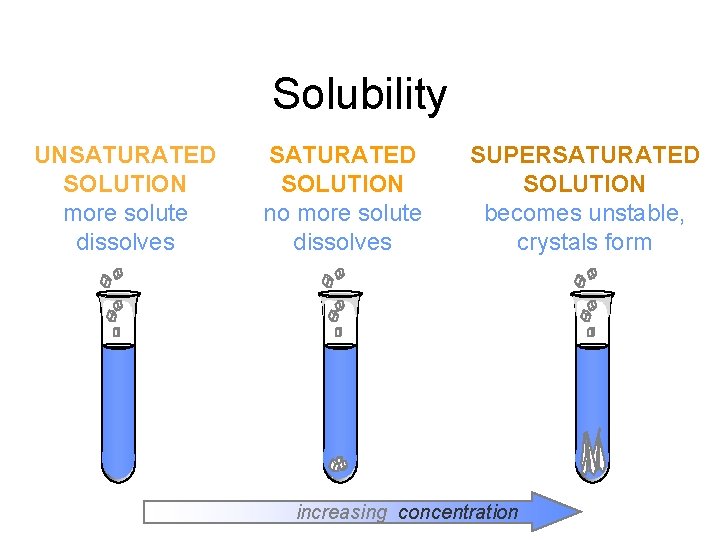
Solubility UNSATURATED SOLUTION more solute dissolves SATURATED SOLUTION no more solute dissolves SUPERSATURATED SOLUTION becomes unstable, crystals form increasing concentration

Solubility vs. Temperature for Solids 140 KI 130 Solubility Table shows the dependence of solubility on temperature Solubility (grams of solute/100 g H 2 O) 120 Na. NO 3 110 gases solids 100 KNO 3 90 80 HCl 70 60 NH 3 KCl 50 40 30 Na. Cl 20 10 KCl. O 3 SO 2 0 Le. May Jr, Beall, Robblee, Brower, Chemistry Connections to Our Changing World , 1996, page 517 NH 4 Cl 10 20 30 40 50 60 70 80 90 100

Solubility maximum grams of solute that will dissolve in 100 g of solvent at a given temperature varies with temperature based on a saturated solution

H 2 3 4 5 6 7 1 Li He Be B C N O F 2 Ne 3 4 Na Mg 5 Al 6 Si 7 P 8 S 9 Cl 10 Ar 13 14 15 16 Cu Zn Ga Ge As Se 17 Br 18 Kr 36 Xe 11 K 12 Ca Sc Ti V Cr Mn Fe Co Ni 19 20 Rb Sr 21 Y 22 23 24 25 26 27 28 29 30 31 Zr Nb Mo Tc Ru Rh Pd Ag Cd In 32 33 34 Sn Sb Te 35 I 37 38 Cs Ba 39 40 Hf 46 47 48 49 Pt Au Hg Tl 50 Pb 51 Bi 52 Po 53 54 At Rn 78 82 83 84 85 55 Fr 87 56 Ra 88 * W 41 Ta 42 W 43 44 45 Re Os Ir 72 73 74 75 76 77 Rf Db Sg Bh Hs Mt 79 80 81 Hydrogen Bonding 86 H 2 O H 2 Te 0 -100 -200 Zumdahl, De. Coste, World of Chemistry 2002, page 443 Boiling Points of Covalent Hydrides 100 104 105 106 107 108 109 Boiling point (o. C) 1 H 2 S H 2 Se 2 3 4 Period of X (H 2 X) 5
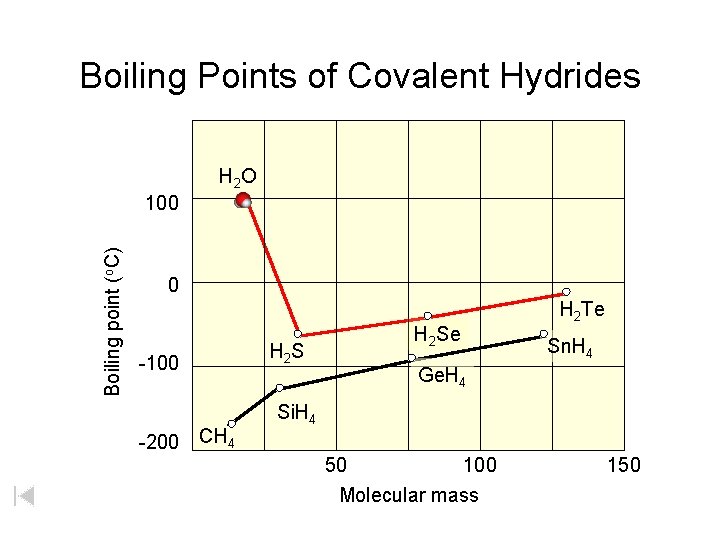
Boiling Points of Covalent Hydrides H 2 O Boiling point (o. C) 100 0 -100 -200 CH 4 H 2 S H 2 Se H 2 Te Sn. H 4 Ge. H 4 Si. H 4 50 100 Molecular mass 150
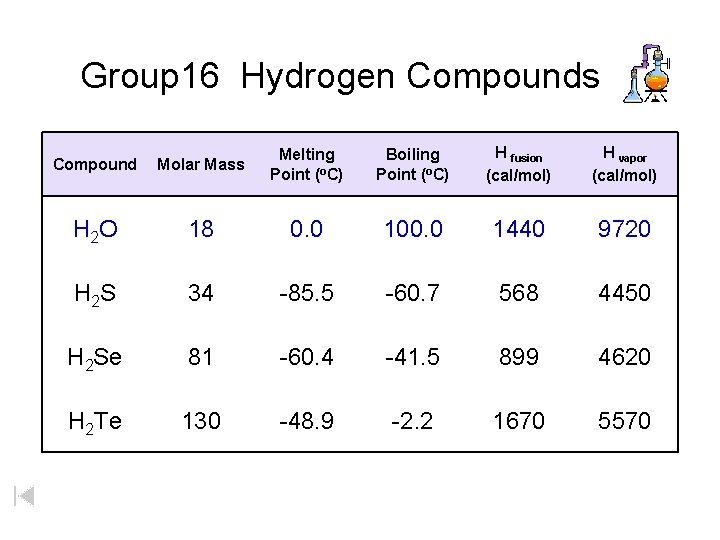
Group 16 Hydrogen Compounds Compound Molar Mass Melting Point (o. C) Boiling Point (o. C) H fusion (cal/mol) H vapor (cal/mol) H 2 O 18 0. 0 100. 0 1440 9720 H 2 S 34 -85. 5 -60. 7 568 4450 H 2 Se 81 -60. 4 -41. 5 899 4620 H 2 Te 130 -48. 9 -2. 2 1670 5570
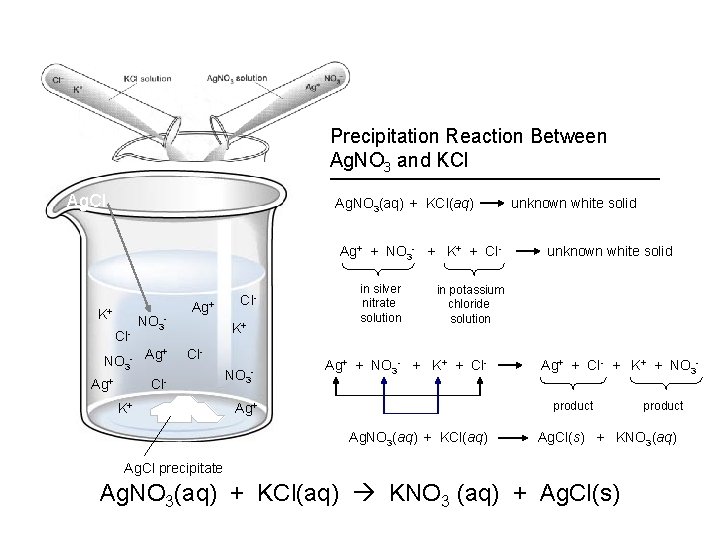
Precipitation Reaction Between Ag. NO 3 and KCl Ag. NO 3(aq) + KCl(aq) Ag+ + NO 3 - + K+ + Cl- K+ Cl- NO 3 - + NO 3 - Ag Ag+ ? Cl. K+ Cl- Cl. K+ NO 3 - in silver nitrate solution unknown white solid in potassium chloride solution Ag+ + NO 3 - + K+ + Cl- Ag+ + Cl- + K+ + NO 3 product Ag+ Ag. NO 3(aq) + KCl(aq) product Ag. Cl(s) + KNO 3(aq) Ag. Cl precipitate Ag. NO 3(aq) + KCl(aq) KNO 3 (aq) + Ag. Cl(s)
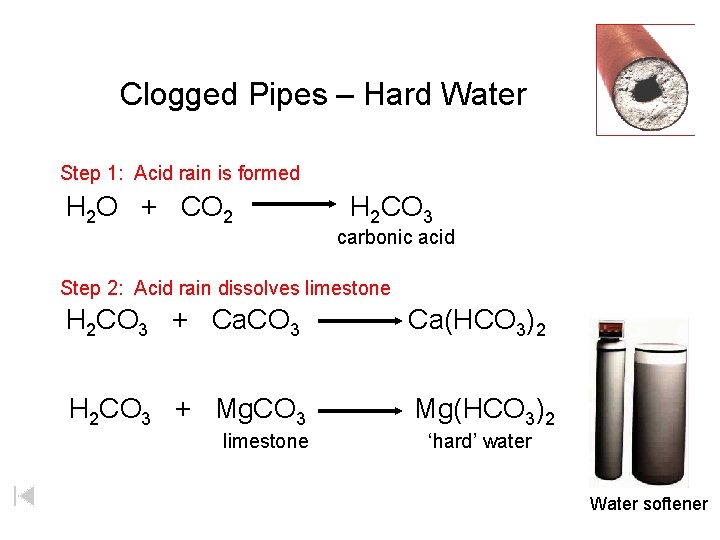
Clogged Pipes – Hard Water Step 1: Acid rain is formed H 2 O + CO 2 H 2 CO 3 carbonic acid Step 2: Acid rain dissolves limestone H 2 CO 3 + Ca. CO 3 Ca(HCO 3)2 H 2 CO 3 + Mg. CO 3 Mg(HCO 3)2 limestone ‘hard’ water Water softener

Pipes develop Scales Step 3: Hard water is heated and deposits scales Ca(HCO 3)2 D Ca. CO 3(s) + H 2 O + CO 2 scales on pipes Mg(HCO 3)2 D Mg. CO 3(s) + H 2 O + CO 2 1 gram = 1/7000 th pound 300 gallons / day with 10 grain water supply

No More Hard Water Scale No More Ugly Hard Water Spotting Protects Plumbing and Appliances Saves Money on Cleaning Products
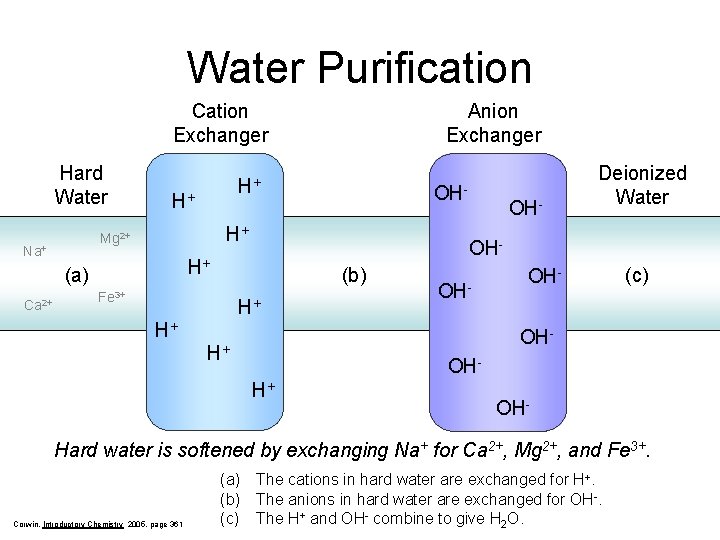
Water Purification Cation Exchanger Hard Water H+ OH- H+ (b) Fe 3+ OH- Deionized Water OH- H+ (a) Ca 2+ H+ Mg 2+ Na+ Anion Exchanger H+ OH- (c) OH- H+ OH- Hard water is softened by exchanging Na+ for Ca 2+, Mg 2+, and Fe 3+. Corwin, Introductory Chemistry 2005, page 361 (a) The cations in hard water are exchanged for H+. (b) The anions in hard water are exchanged for OH-. (c) The H+ and OH- combine to give H 2 O.

Which ions are removed from hard water to produce soft water? Na+(aq) + H (resin) Cl-(aq) + (resin) OH Na (resin) Cl + + H +(aq) OH -(aq) Notice that the ion exchange resin produces both hydrogen ions and hydroxide ions which can readily combine to give water. H+(aq) + OH- (aq) H 2 O (aq) The net result is that the resin removes all ions from water passing through the deionizing system. Hard water is softened by exchanging Na+ for Ca 2+, Mg 2+, and Fe 3+. Corwin, Introductory Chemistry 2005, page 361
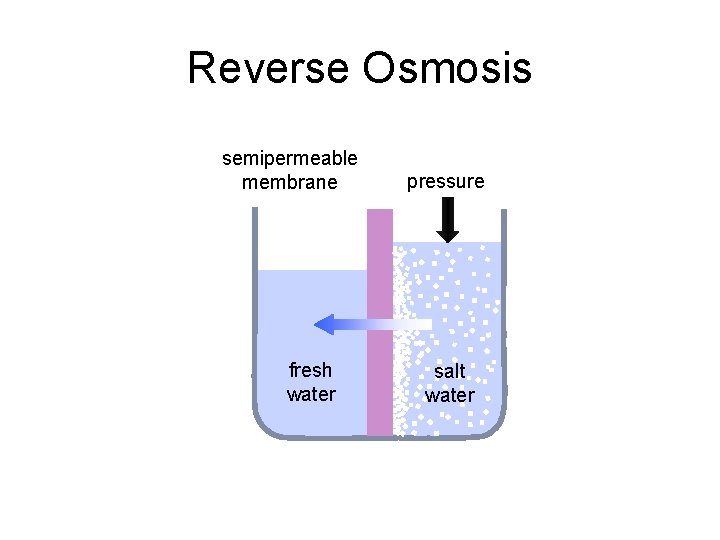
Reverse Osmosis semipermeable membrane fresh water pressure salt water
- Slides: 104