SOLUTIONS Ch 15 and 16 START WITH A













































- Slides: 54

SOLUTIONS Ch. 15 and 16

START WITH A WEB QUEST! • Take a copy of the “Web Quest” and navigate to my website. Under “Links” scroll to the bottom of the page under the “Chemistry” heading and click on the links to complete the assignment.

CHEMISTRY & YOU • Why are some sunsets red? The atmosphere contains particles of water and dust. As sunlight passes through the particles, it is scattered. However, not all wavelengths are scattered to the same extent. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

SUSPENSIONS • The difference between a solution and suspension is easily seen when the type of mixture is filtered. The small size of the solute particles in a solution allows them to pass through filter paper. The particles of a suspension can be removed by filtration.

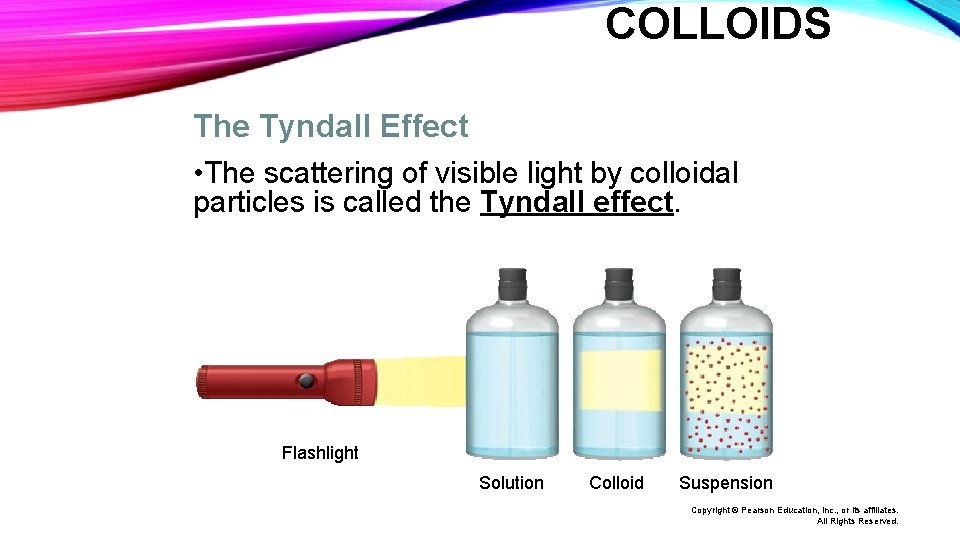
COLLOIDS The Tyndall Effect • The scattering of visible light by colloidal particles is called the Tyndall effect. Flashlight Solution Colloid Suspension Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

COLLOIDS • Suspensions also exhibit the Tyndall effect. • The particles in solutions are too small to scatter light. Flashlight Solution Colloid Suspension Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

• tyndall Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

CHEMISTRY & YOU • What would be the ideal conditions to see a red sunset? A misty or foggy evening would be ideal for seeing a red sunset. There would be a large number of particles to scatter the sunlight. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

COLLOIDS Brownian Motion Brownian motion is caused by collisions of the molecules of the dispersion medium with the small, dispersed colloidal particles. • These collisions help prevent the colloidal particles from settling. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

• Brownian Motion Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

COLLOIDS Emulsions • An emulsion is a colloidal dispersion of a liquid in a liquid. • An emulsifying agent is essential for the formation of an emulsion and for maintaining the emulsion’s stability. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

• Emulsion Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

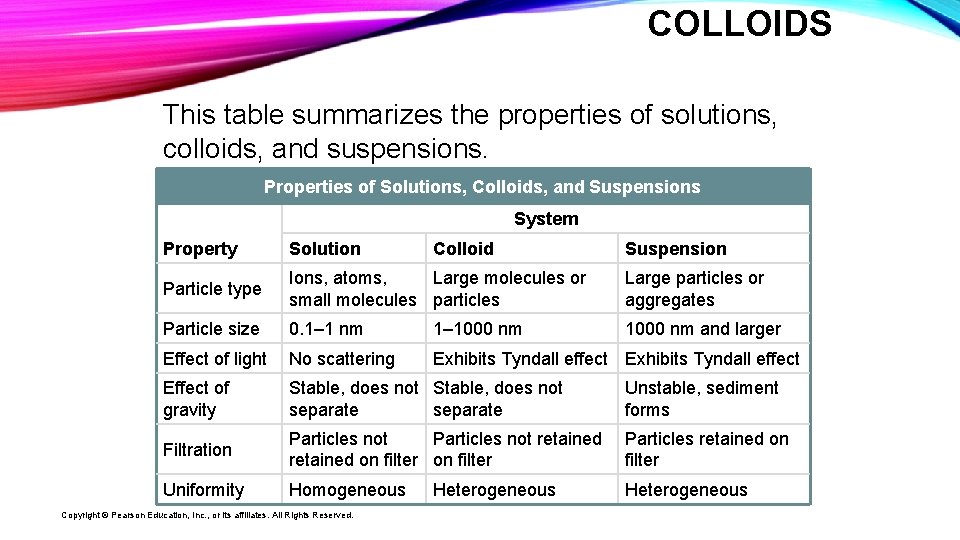
COLLOIDS This table summarizes the properties of solutions, colloids, and suspensions. Properties of Solutions, Colloids, and Suspensions System Property Solution Particle type Ions, atoms, Large molecules or small molecules particles Large particles or aggregates Particle size 0. 1– 1 nm 1– 1000 nm and larger Effect of light No scattering Exhibits Tyndall effect Effect of gravity Stable, does not separate Unstable, sediment forms Filtration Particles not retained on filter Particles retained on filter Uniformity Homogeneous Heterogeneous Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved. Colloid Heterogeneous Suspension

SUPERSATURAT • The rate at which excess solute deposits upon ED the surface of a seed crystal can be very rapid. The solution is clear before a seed crystal is added. Crystals begin to form immediately after the addition of a seed crystal. Excess solute crystallizes rapidly.

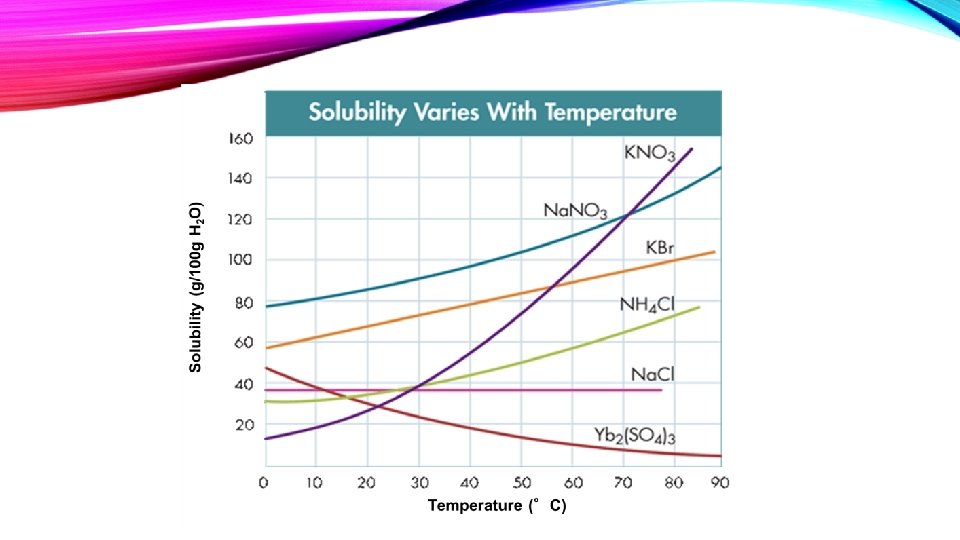
• Temperature FACTORS AFFECTING SOLUBILITY – PAGE 6 Basically, solubility increases with temperature. It is the case for most of the solvents. The situation is different for gases. With an increase of temperature they become less soluble in each other and in water, but they become more soluble in organic solvents.

SOLUTION FORMATION • Granulated sugar dissolves faster than sugar cubes, and both granulated sugar and sugar cubes dissolve faster in hot tea or when you stir the tea. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

• Polarity • In most cases solutes dissolve in solvents that have a similar polarity. Chemists use s popular saying to describe this feature of solutes and solvents. “Like dissolves like”. Non-polar solutes do not dissolve in polar solvents and visa versa.

SOLUBILITY Liquids that are insoluble in each other are immiscible. • Oil and water are examples of immiscible liquids. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

RECALL POLARITY… Many unique and important properties of water—including its high surface tension, low vapor pressure, and high boiling point—result from hydrogen bonding. Hydrogen bond Liquid water

SURFACE TENSION

• Pressure • Solid and liquid solutes • For a majority of solid and liquid solutes, pressure does not affect solubility. • Gas solutes • For gasses, Henry’s Law states that the solubility of a gas is directly proportional to the pressure of this gas. A good proof of Henry’s Law can be observed when opening a pop. When we decrease the pressure in a bottle, the gas that was dissolved in the drink bubbles out of it.

• Molecular Size • Larger molecules of solute have a larger molecular weight. • It is more difficult for solvent molecules to surround bigger molecules. As a general rule, larger particles are generally less soluble. • If the pressure and temperature are the same, then out of two solutes of the same polarity, the one with smaller particles is usually more soluble.

• Stirring increases the speed of dissolving • Stirring does not have an effect on the solubility of a substance, so why does stirring sugar into ice tea seem to help? • Actually, if we left the tea to stand for a long time, the sugar would dissolve. Stirring only increases the speed of the process – it increases the movement of the solvent that exposes solute to fresh portions of it, thus enabling solubility. As molecules in liquid substances are in constant motion, the process would take place anyway, but it would take more time.



PAGE 8 • Complete the Solubility and Solubility Curves Worksheet by defining the terms listed and interpreting the graph and answering all questions. • Use the Last Page in the packet also for the second solubility graph for gases.

CONCENTRATIONS AND DILUTIONS • A way of expressing concentration is called Molarity. As is clear from its name, molarity involves moles. • Molarity is the number of moles of solute dissolved in one liter of solution. • The units, therefore, are moles per liter. • Specifically, its moles of solute per liter of solution. Rather than writing out moles per liter, these units are abbreviated as M and it means moles per liter (not just moles).

• You must be very careful to distinguish between moles and molarity. “Moles” measures the amount or quantity of material you have; “Molarity” measures the concentration of that material. So, when you’re given a problem or some information that says the concentration of the solution is 0. 1 M, that means it has 0. 1 mole for every liter of solution; it does not mean that it is 0. 1 moles.

HELPFUL EQUATIONS • Molarity (M)= Moles of solute Liters of solution Dilution: M 1 V 1 = M 2 V 2 (M=molarity of solution, V=volume of solution)

MOLARIT Y • The figure below illustrates the procedure for making a 0. 5 M, or 0. 5 molar, solution. Add 0. 5 mol of solute to a 1 -L volumetric flask half filled with distilled water. Swirl the flask carefully to dissolve the solute. Fill the flask with water exactly to the 1 -L mark. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

DILUTIONS

MAKING DILUTIONS The student is preparing 100 m. L of 0. 40 M Mg. SO 4 from a stock solution of 2. 0 M Mg. SO 4. She measures 20 m. L of the stock solution with a 20 -m. L pipet. She transfers the 20 m. L to a 100 -m. L volumetric flask. She carefully adds water to the mark to make 100 m. L of solution.

EXAMPLES • A solution is made by adding 27. 5 g of calcium fluoride to enough water to make 1. 00 L. What is the concentration(molarity)? • M = Moles solute = 0. 35 mol = 0. 35 M Liters of solution 1. 00 L First convert grams to moles! 27. 5 g Ca. F 2 X 1 mole Ca. F 2 = 0. 35 mol 78. 08 g Ca. F 2

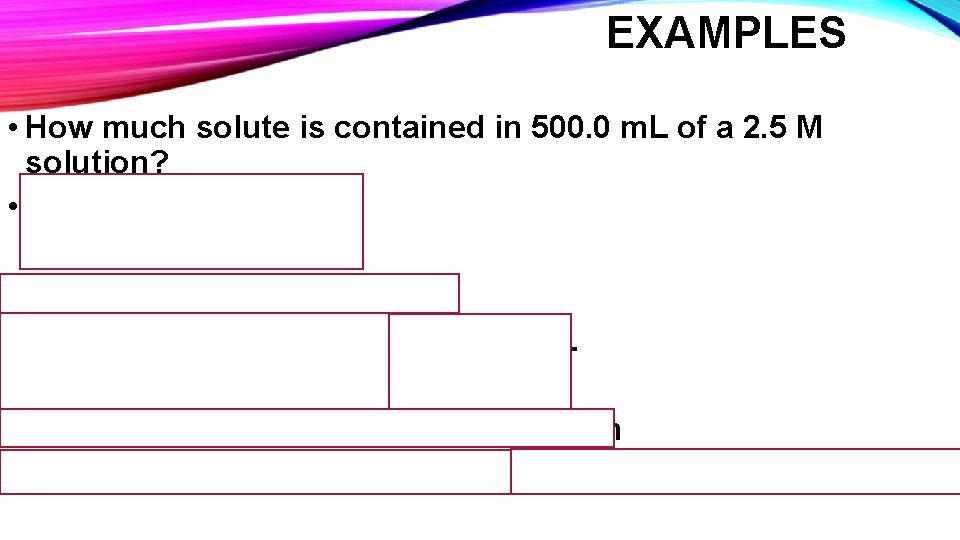
EXAMPLES • How much solute is contained in 500. 0 m. L of a 2. 5 M solution? • M = Moles solute Liters of solution First convert m. L to Liters! 500. 0 m. L X 1 L = 0. 5000 L 1000 m. L Moles solute = Molarity X Liters Solution Moles solute = 2. 5 M X 0. 5000 L = 1. 25 moles solute

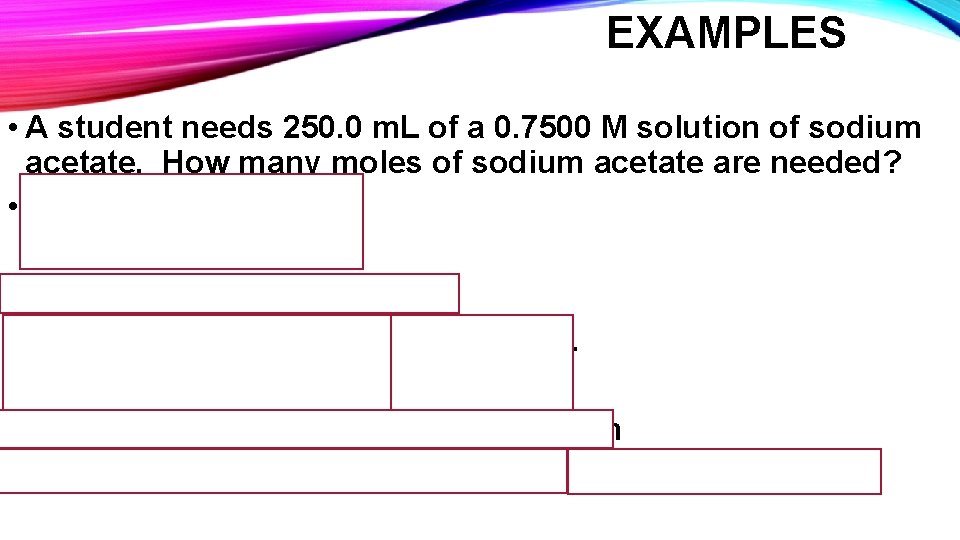
EXAMPLES • A student needs 250. 0 m. L of a 0. 7500 M solution of sodium acetate. How many moles of sodium acetate are needed? • M = Moles solute Liters of solution First convert m. L to Liters! 250. 0 m. L X 1 L = 0. 2500 L 1000 m. L Moles solute = Molarity X Liters Solution Moles solute = 0. 7500 M X 0. 2500 L = 0. 1875 moles solute

2 A PAGE 11 • 110. 0 m. L of 3. 00 M sulfuric acid has 25. 0 m. L of water ADDED to it. What is the resulting concentration of the solution? • M 1 V 1 = M 2 V 2 • Both volumes are in m. L, so it is okay – leave it. • M 1= 3. 00 M, V 1= 110. 0 m. L, M 2= ? , V 2= 110. 0 m. L+25. 0 m. L • M 2= M 1 V 1 = 3. 00 MX 110. 0 m. L = 2. 44 M V 2 135 m. L

2 C • Barium nitrate is purchased as a 17. 0 M concentration. Explain how you would prepare 500. 0 m. L of a 5. 00 M solution. • M 1 V 1 = M 2 V 2 • You will be finding the initial volume of 17. 0 M solution!! • M 1= 17. 00 M, V 1= ? , M 2= 5. 00 M, V 2= 500. 0 m. L • V 1= M 2 V 2 = 5. 00 MX 500. 0 m. L = 147 m. L M 1 17. 0 M So…. Take 147 m. L of 17. 0 M solution and add 353 m. L of water to get 500. 0 ml total 5. 00 M solution.

• Complete Molarity/Dilutions Worksheet on p age 11 and 12.

COLLIGATIVE PROPERTIES • Colligative properties are those properties of solutions that depend on the number of dissolved particles in solution, but not on the identities of the solutes. For example, the freezing point of salt water is lower than that of pure water due to the presence of the salt dissolved in the water. • If the molar amounts of solute are the same AND the number of ions are the same, the freezing points will be the same. If the number of ions are different, then the one with the most particles (ions) will lower the freezing point more.

• The three commonly studied colligative properties are freezing point depression, boiling point elevation, and vapor pressure lowering.

FREEZING POINT DEPRESSION • The presence of a solute lowers the freezing point of a solution relative to that of the pure solvent. For example, pure water freezes at 0ºC; if one dissolves 10 grams of sodium chloride in 100 grams of water, the freezing point goes down to -5. 9ºC. If one uses sucrose, C 12 H 22 O 11, instead of sodium chloride, 10 grams in 100 grams of water gives a solution with a freezing point of -0. 56ºC. The reason that the salt solution has a lower freezing point than the sugar solution is that there are more particles in 10 grams of sodium chloride than in 10 grams of sucrose.


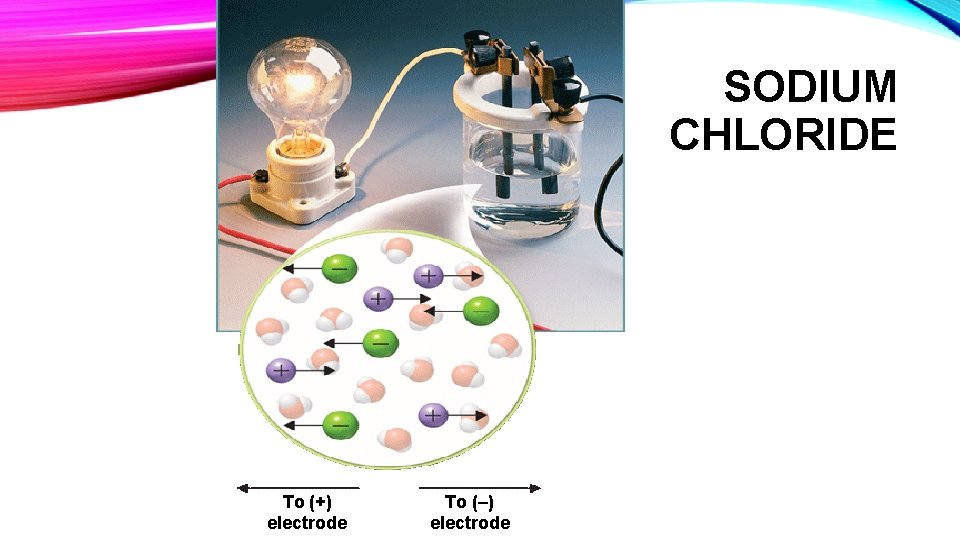
• Sodium chloride has almost six times as many sodium chloride formula units as there are sucrose molecules in a gram of sucrose. In addition, each sodium chloride formula unit comes apart into two ions (a sodium cation and a chloride anion) when dissolved in water. Sodium Chloride is therefore an electrolyte and will conduct electricity when dissolved in water.

SODIUM CHLORIDE To (+) electrode To (–) electrode

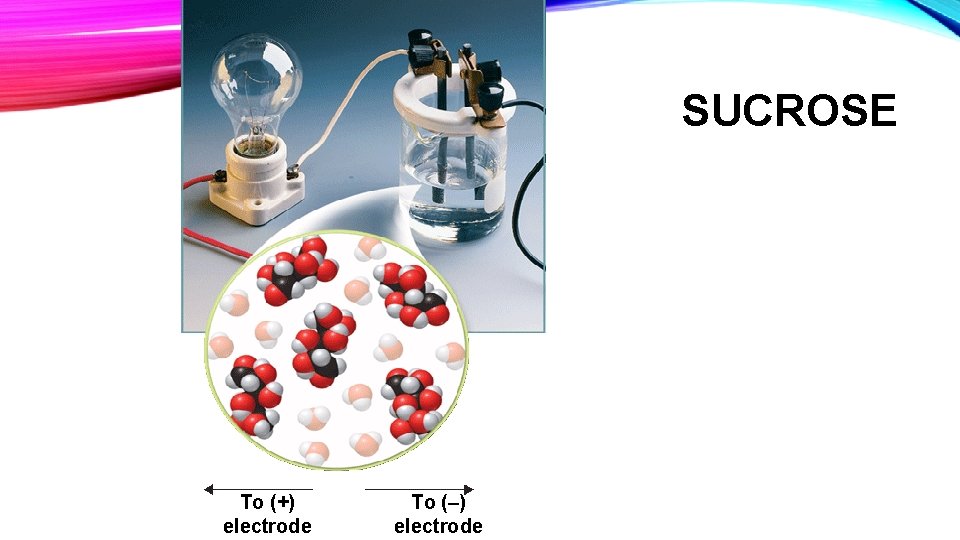
• Sucrose is a nonelectrolyte, which means that the solution contains whole sucrose molecules. In predicting the freezing point of a solution, one must consider not only the number of formula units present, but also the number of ions that result from each formula unit, in the case of ionic compounds. • Ex: Ca. Cl 2 will make more ions per formula unit than Na. Cl and therefore if there are the same number of moles of each in solution, Ca. Cl 2 will lower the freezing point more than Na. Cl.

SUCROSE To (+) electrode To (–) electrode

CACL 2 WILL LOWER THE BP MORE IF EQUAL MOLES

BOILING POINT ELEVATION • The boiling point of a solution is higher than that of the pure solvent. Accordingly, the use of a solution, rather than a pure liquid, in antifreeze serves to keep the mixture from boiling in a hot automobile engine (Antifreeze also keeps the water from freezing during winter because of freezing point depression. )

BOILING POINT ELEVATION • As with freezing point depression, the effect depends on the number of solute particles present in a given amount of solvent, but not the identity of those particles. • If 10 grams of Na. Cl are dissolved in 100 g of water, the boiling point of the solution is 101. 7ºC.

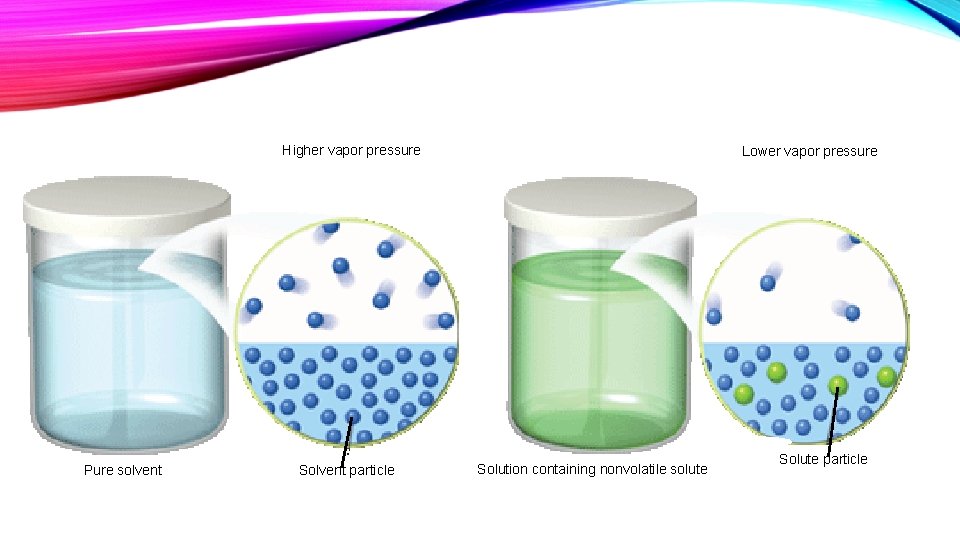
VAPOR PRESSURE LOWERING • The vapor pressure of a liquid is the equilibrium pressure of gas molecules from that liquid (i. e. , the results of evaporation) above the liquid itself. • At room temperature, the vapor pressure of pure water is about 20 Torr. • If, instead of pure water, an aqueous solution is placed in the glass, the equilibrium pressure will be lower than it would be for pure water.

• Raoult’s law states that the vapor pressure of the solvent over the solution is proportional to the fraction of solvent molecules in the solution; • That is, if two-thirds of the molecules are solvent molecules, the vapor pressure from the solvent is approximately two-thirds of what it would be for the pure solvent at that temperature.

Higher vapor pressure Pure solvent Solvent particle Lower vapor pressure Solution containing nonvolatile solute Solute particle

• One consequence of this lowering of vapor pressure may be observed in a spilled can of soda. As the water evaporates, the soda becomes more sugar and less water, until the vapor pressure of the water is so low that it barely evaporates. As a result, the spilled soda remains sticky for a long time. Contrast this behavior with that of a water spill.

• Complete Colligative Properties Worksheet on page 16