Solutions BASIC DEFINITIONS Solution a homogeneous mixture of

Solutions
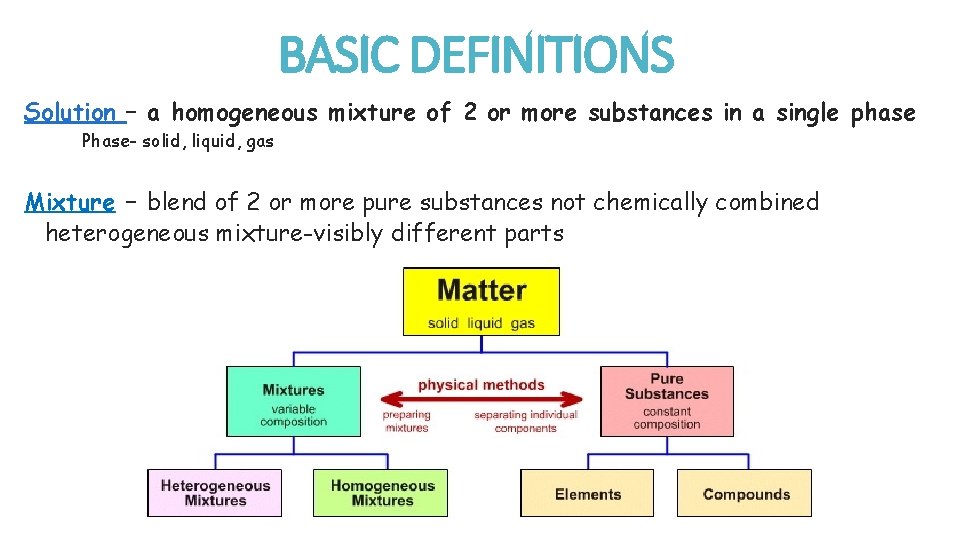
BASIC DEFINITIONS Solution – a homogeneous mixture of 2 or more substances in a single phase Phase- solid, liquid, gas Mixture – blend of 2 or more pure substances not chemically combined heterogeneous mixture-visibly different parts

A heterogeneous mixture is a mixture where the components of the mixture are not uniform. o In other words, you can see the different components that make up the mixture. A homogeneous mixture is a mixture where the components that make up the mixture are uniformly distributed throughout the mixture. The composition of the mixture is the same throughout. o In other words, you can’t see the different components that make up the mixture.
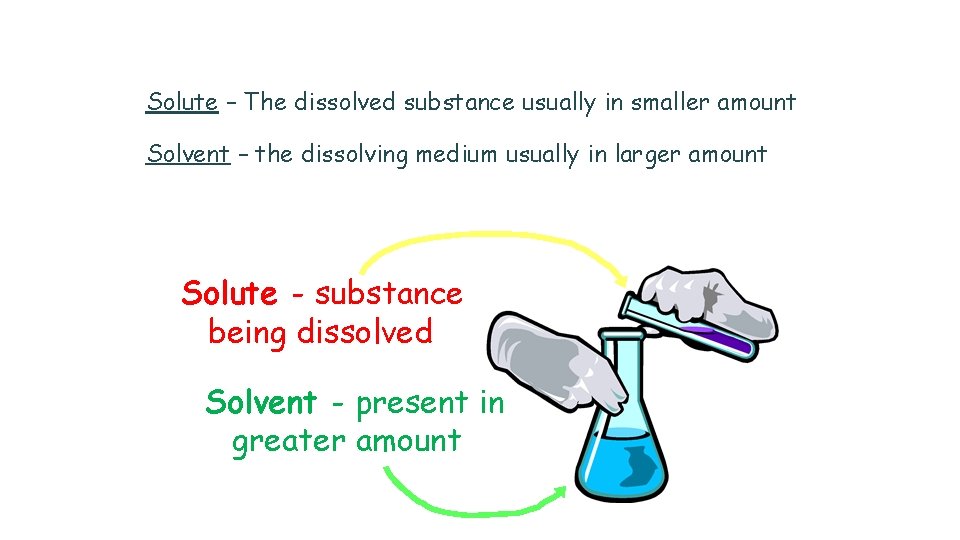
Solute – The dissolved substance usually in smaller amount Solvent – the dissolving medium usually in larger amount Solute - substance being dissolved Solvent - present in greater amount

Solubility Definition: the maximum quantity of a substance that may be dissolved in another. -The maximum amount of solute that may be dissolved in a solvent.

FACTORS AFFECTING SOLUBILITY 1. Surface Area – increasing the surface area gives molecules more places to interact causing the solute to dissolve faster 2. Agitation/Stirring – stirring gives molecules more opportunities to interact – faster dissolving 3. Temperature – Adding heat adds energy to molecules = faster dissolving 4. “Like dissolves like” – polar solutes dissolve in polar solvents or Nonpolar solutes dissolve in Nonpolar solvents

Solutes Change Solvents The amount of solute in a solution determines how much the physical properties of the solvent are changed. Lowering the Freezing Point The freezing point of a liquid solvent decreases when a solute is dissolved in it. Ex. Pure water freezes at 320 F (00 C), but when salt is dissolved in it, the freezing point is lowered. This is why people use salt to melt ice. Raising the Boiling Point The boiling point of a solution is higher than the boiling point of the solvent. Therefore, a solution can remain a liquid at a higher temperature than its pure solvent. Ex. The boiling point of pure water is 2120 F (1000 C), but when salt is dissolved in it, the boiling point is higher. This is why it takes salt water longer to boil than fresh water.

CONCENTRATION The strength of a solution MOLARITY (M) Moles of solute per Liter of solution

SAMPLE PROBLEM A You have 3. 50 L of solution that contains 90. 0 g of sodium chloride, Na. Cl. What is the molarity of that solution?

SAMPLE PROBLEM A You have 3. 50 L of solution that contains 90. 0 g of sodium chloride, Na. Cl. What is the molarity of that solution? Step 1: Outline what you know. M = ? Mol / L mol = 90. 0 g L = 3. 50 L

SAMPLE PROBLEM A You have 3. 50 L of solution that contains 90. 0 g of sodium chloride, Na. Cl. What is the molarity of that solution? Step 2: Convert any units necessary. mol = 90. 0 g Na. Cl x 1 mol Na. Cl 58. 44 g Na. Cl = 1. 54 mol Na. Cl

SAMPLE PROBLEM A You have 3. 50 L of solution that contains 90. 0 g of sodium chloride, Na. Cl. What is the molarity of that solution? Step 3: Plug into the equation and solve. M = ? Mol / L mol = 1. 54 mol L = 3. 50 L

SAMPLE PROBLEM A You have 3. 50 L of solution that contains 90. 0 g of sodium chloride, Na. Cl. What is the molarity of that solution? Step 3: Plug into the equation and solve. M = 1. 54 mol 3. 50 L = 0. 44 mol / L

SAMPLE PROBLEM B You have 0. 8 L of a 0. 5 M HCl solution. How many moles of HCl does this solution contain?

SAMPLE PROBLEM B You have 0. 8 L of a 0. 5 M HCl solution. How many moles of HCl does this solution contain? Step 1: Outline what you know. M = 0. 5 Mol / L mol = ? mol L = 0. 8 L

SAMPLE PROBLEM B You have 0. 8 L of a 0. 5 M HCl solution. How many moles of HCl does this solution contain? Step 2: Convert any units necessary. All units correct! Moving on…

SAMPLE PROBLEM B You have 0. 8 L of a 0. 5 M HCl solution. How many moles of HCl does this solution contain? Step 3: Plug into the equation and solve. M = 0. 5 Mol / L mol = ? mol L = 0. 8 L

SAMPLE PROBLEM B You have 0. 8 L of a 0. 5 M HCl solution. How many moles of HCl does this solution contain? Step 3: Plug into the equation and solve. HINT- Cross multiply!!! (0. 8 L) 0. 5 M = 1 X 0. 8 L (0. 8 L)

SAMPLE PROBLEM B You have 0. 8 L of a 0. 5 M HCl solution. How many moles of HCl does this solution contain? Step 3: Plug into the equation and solve. mol = 0. 4
- Slides: 19