Solutions BAFPJUC 10272020 solutions Solutions are homogeneous liquid

Solutions BA-FP-JU-C 10/27/2020

solutions Solutions are homogeneous liquid mixtures containing completely dissolved components (One-phase system). BA-FP-JU-C 10/27/2020

Solutions for ORAL dosage Advantages of solutions over a solid DF: • Much easier to swallow (i. e. Ease of taking e. g children, elderly, chronic patients). • Readily absorbed from GIT. An advantage of solutions over suspensions: • Solutions are homogenous dispersions without the need to shake the bottle. Disadvantages of solutions: • Bulky, not convenient to carry around. • Less chemically and microbiologically stable. • Drugs with unpleasant taste may not be suitable for administration as oral solutions. • Accuracy of dosage depends on the patient. BA-FP-JU-C 10/27/2020

Solutions for oral dosage The different forms of oral solutions are: • Syrups: which are concentrated, aqueous solutions of sugar or sugar-substitute. • Elixirs: which are clear, sweetened, hydroalcoholic solutions suitable for water insoluble drugs. • Linctuses: viscous liquids used in the treatment of cough. They usually contain a high proportion of sucrose, other sugars or polyhydric alcohol or alcohols. • Mixtures: pharmaceutical oral solutions and suspensions. Examples are chloral hydrate mixture. • Oral drops: are oral solutions or suspensions which are administered in small volumes, using a suitable measuring device. A proprietary example is Abidec® vitamin drops. BA-FP-JU-C 10/27/2020

Solutions for oral dosage Containers for dispensed oral solutions: • Plain, amber medicine bottles should be used, with a re-closable child-resistant closure with some exceptions. • A 5 m. L measuring spoon or an appropriate oral syringe should be supplied to the patient. Special labels and advice for dispensed oral solutions: • An expiry date should appear on the label for extemporaneously prepared solutions. Most 'official' mixtures and some oral solutions are freshly or recently prepared. • 'Official' elixirs and linctuses and manufactured products are generally more stable, unless diluted. • Diluted products generally have a shorter shelf life than the undiluted preparation. • Linctuses should be sipped and swallowed slowly without the addition of water. BA-FP-JU-C 10/27/2020

Solutions for other pharmaceutical uses Mouthwashes and Gargles • Gargles are used to relieve or treat sore throats • Mouthwashes are used on the mucous membranes of the oral cavity, rather than the throat, to refresh and mechanically clean the mouth. • Both are concentrated solutions, and usually diluted with warm water before use. • Gargles tend to contain higher concentrations of active ingredients than mouthwashes. • They may contain antiseptics, analgesics or weak astringents. The liquid is usually not intended for swallowing. BA-FP-JU-C 10/27/2020

Solutions for other pharmaceutical uses Mouthwashes and Gargles Containers for mouthwashes and gargles • An amber, ribbed bottle should be used for extemporaneously prepared solutions. Medicine bottles may be used for products which are intended to be swallowed. • Manufactured mouthwashes and gargles are usually packed in plain bottles. Special labels and advice for mouthwashes and gargles • Directions for diluting the preparations should be given to the patient. • If the preparation is not intended for swallowing, the following label is appropriate: “Not to be swallowed in large amounts ” BA-FP-JU-C 10/27/2020

Solutions for other pharmaceutical uses Nasal solutions • Most nasal preparations are solutions, administered as nasal drops or nasal sprays. • They are isotonic to nasal secretions and buffered to the normal p. H range of nasal fluids (p. H 5. 5 -6. 5) to prevent damage to ciliary transport in the nose. • The most frequent use of nose drops is as a decongestant for the common cold or to administer local steroids for the treatment of allergic rhinitis. • Nasal route may also be useful for new biologically active peptides and polypeptides which need to avoid the first pass metabolism or GI destruction. BA-FP-JU-C 10/27/2020

Solutions for other pharmaceutical uses Ear drops • They may also be referred to as otic or aural preparations. • Ear drops are solutions of one or more active ingredient which exert a local effect in the ear, e. g. by softening earwax or treating infection or inflammation. • Propylene glycol, oils, glycerol (to increase viscosity) and water may be used as vehicles. BA-FP-JU-C 10/27/2020

Solutions for other pharmaceutical uses Nasal and aural solutions Containers for nasal and aural preparations: • Extemporaneously: should be packed in an amber, ribbed hexagonal glass bottle (which type? ) which is fitted with a teat and dropper. • Manufactured nasal solutions may be packed in flexible plastic bottles which deliver a fine spray to the nose when squeezed, or in a plain glass bottle with a pump spray or dropper. • Manufactured ear drops are usually packed in small glass or plastic containers with a dropper. BA-FP-JU-C 10/27/2020

Solutions for other pharmaceutical uses Nasal and aural solutions Special labels and advice for nasal and aural prep: • Not to share nasal sprays or nose and ear drops in order to minimize contamination and infection. • Patients should be given advice on how to administer extemporaneously prepared nose and ear drops, accompanied by written information if possible. • Extemporaneous preparations should be labeled with the appropriate expiry date following the official monographs. • “For external use” is not an appropriate label and so ‘Not to be taken” is advised. BA-FP-JU-C 10/27/2020

Solutions for other pharmaceutical uses Enemas • Enemas are oily or aqueous solutions that are administered rectally. • They are usually anti-inflammatory, purgative, sedative or given to allow X-ray examination of the lower bowel. • Retention enemas are administered to give either a local action of the drug or for systemic absorption. They are used after defecation. • Microenemas are single-dose, small volume solutions. They are packaged in plastic containers with a nozzle for insertion into the rectum. • Large-volume (0. 5 -1 litre) enemas should be warmed to body temperature before administration. BA-FP-JU-C 10/27/2020

Solutions for other pharmaceutical uses Enemas Containers for enemas: • If extemporaneously produced, enemas are packed in amber fluted glass bottles. • Manufactured enemas will usually be packed in disposable polythene or polyvinyl chloride bags sealed to a rectal nozzle. Special labels and advice for enemas: • Patients should be advised on how to use the enema if they are self-administering and the time that the product will take to work. • The label 'For rectal use only' should be used. BA-FP-JU-C 10/27/2020

EXPRESSION OF CONCENTRATION • By percentage strength as %w/v or %v/v, • In terms of amount of drug contained in 5 m. L of vehicle. FORMULATION OF SOLUTIONS When compounding a solution: • Information on solubility and stability of each of the solutes ( drug, excipients) must be taken into account. • Chemical and physical interactions that may take place between constituents must also be taken into account. e. g. Esters of p-hydroxybenzoic acid (parabens) with some flavoring oils. BA-FP-JU-C 10/27/2020

Solubility Saturation solubility: is the maximum concentration of a solution which may be prepared at a given temperature. BA-FP-JU-C 10/27/2020

Factors affecting solubility • Polarity of the compound • p. H of the medium • Temperature of solution • Particle size may affect the rate of solution Increasing the solubility 1. Co solvency: ethanol, glycerol, propylene glycol or sorbitol are cosolvents used to increase the solubility of weak electrolytes and non-polar molecules. 2. Solubilization: surfactants such as polysorbates for oral solutions, soaps for external phenolic preparations. BA-FP-JU-C 10/27/2020

Vehicles • A vehicle is the medium which contains the ingredients of a medicine. In solutions, this is the solvent. • The choice of a vehicle depends on: ü The intended use of the preparation ü The nature and physicochemical properties of the active ingredients. Water as vehicle • Water is widely available, relatively inexpensive, palatable and non-toxic for oral use and non-irritant for external use. It is also a good solvent for many ionizable drugs. BA-FP-JU-C 10/27/2020

Vehicles Water Types of water: • Potable water is drinking water, drawn freshly from a mains supply. It should be palatable and safe for drinking, Its chemical composition may include mineral impurities which could react with drugs, e. g. the presence of calcium carbonate in hard water. • Purified water is prepared from suitable potable water by distillation, by treatment with ion-exchange materials or by any other suitable treatment method such as reverse osmosis. Distilled water is purified water that has been prepared by distillation. • Water for preparations is potable or freshly boiled and cooled purified water, which can be used in oral or external preparations which are not intended to be sterile BA-FP-JU-C 10/27/2020

Vehicle Water • Water for injections is pyrogen-free distilled water, sterilized immediately after collection and used for parenteral products. • Aromatic waters are near-saturated aqueous solutions of volatile oils or other aromatic or volatile substances, and are often used as a vehicle in oral solutions. Some have a mild carminative action. Aromatic waters are usually prepared from a concentrated ethanolic solution, in a dilution of 1 part to 39 parts with water. • Chloroform water is used as an antimicrobial preservative and also adds sweetness to preparations. Ø P. S. In general, chloroform can be available as: ü ü Chloroform BP = 100% v/v Concentrated chloroform water BPC = 10% v/v Single-strength chloroform water = Chloroform water BP= 0. 25% v/v Double-strength chloroform water= 0. 5% v/v BA-FP-JU-C 10/27/2020

Other vehicles used in pharmaceutical solutions • Syrup BP is a solution of 66. 7% sucrose in water. Promotes dental decay and is unsuitable for diabetic patients. Sweetening agents as mannitol, sorbitol, xylitol, etc. can replace the sucrose to give 'sugarfree' solvents. • Alcohol rarely used for internal and mainly external • Glycerol (glycerin) may be used alone as a vehicle in some external preparations. It is viscous and miscible both with water and alcohol. It may be added as a stabilizer and sweetener in internal preparations. In concentrations above 20% v/v it acts as a preservative. • Propylene glycol is a less viscous liquid and a better solvent than glycerol. • Oils Bland oils such as fractionated coconut oil and arachis oil may be used for fat soluble compounds, e. g. calciferol. • Acetone and solvent ether used as cosolvents in external preparation (limitations of ether). BA-FP-JU-C 10/27/2020

Preservations of solutions • Why preservation? • Preservatives are added to the formulation to reduce or prevent microbial growth. Chloroform: • Chloroform is the most widely used in oral extemporaneous preparations although there are disadvantages to its use. Also used for external preparations • Use in general is limited to a chloroform content of 0. 5% (w/w or w/v). • For oral solutions, chloroform at a strength of 0. 25% v/v will usually be incorporated as Chloroform Water BP. BA-FP-JU-C 10/27/2020

Preservations of solutions Other preservatives for oral solutions: • Benzoic acid at a strength of 0. 1% w/v • Sorbic acid • Para-hydroxybenzoate esters (Parabens). • Ethanol • Syrup: Syrups can be preserved by the maintenance of a high concentration of sucrose 65% w/w. • Some of the alternative preservatives have p. H-dependent activity. Preservatives for external preparations: • Chlorocresol (0. 1% w/v) • Chlorbutanol (0. 5% w/v) • Parabens BA-FP-JU-C 10/27/2020

Flavouring agents • Why? • Selection of flavours: ü To mask particular taste types, e. g. a fruit flavour helps to disguise an acid taste. ü The age of the patient should be taken into account when selecting a flavour, as children will tend to enjoy fruit or sweet flavours. ü Some flavours are associated with particular uses, e. g. peppermint is associated with antacid preparations. ü The flavour and colour should also complement each other. • Extemporaneous medicines tend to use natural flavours added as juices (raspberry ), extracts (liquorice), spirits (lemon and orange), syrups (blackcurrant), tinctures (ginger) and aromatic waters (anise and cinnamon). BA-FP-JU-C 10/27/2020

Sweetening agents • Sugars, including glucose and sucrose. • Sucrose enhances the viscosity of liquids and also gives a pleasant texture in the mouth. • ‘Sugar free’ solutions include sweetening agents such as sorbitol, mannitol, xylitol, saccharin and aspartame as alternatives to sugar (i. e. sucrose, fructose and glucose). BA-FP-JU-C 10/27/2020

Colouring agents • ü ü ü Colouring agents are added to pharmaceutical preparations to Enhance the appearance of a preparation, Increase the acceptability of a preparation to the patient. Colour is also useful to give a consistent appearance. Colours are often matched to the flavour of a preparation. Colours can give distinctive appearances to some medicines. • Colouring agents should be: ü Non-toxic. ü Free of any therapeutic activity themselves. • Types of colouring agents: ü Natural colourants: derived from animals or plalnts (e. g. carotenoids, chlorophylls, caramel, cochineal, saffron and red beetroot extract). ü Mineral pigments: such as iron oxide are not often used in solutions due to their low water solubility. ü Synthetic organic dyes such as the azo compounds. • Colours appear in pharmaceutical formulae less often now, especially in children's medicines. • In the EU, A EUROPEAN LIST OF COLOURING AGENTS HAVE THE DISIGNATION ‘E’ with numbers between 100 and 180. BA-FP-JU-C

Stabilizers • Antioxidants may be used where ingredients are liable to degradation by oxidation, e. g. in oils. • Those which are added to oral preparations include: ü Ascorbic acid ü Citric acid ü Sodium meta bisulphite ü sodium sulphite. • These are odourless, tasteless and non-toxic. BA-FP-JU-C 10/27/2020

Viscosity enhancing agents • Such as syrups, they improve viscosity, palatability, and ease pourability. • Other thickening agents can also be used. BA-FP-JU-C 10/27/2020

Examples on solution preparations • Ammonium and Ipecacuanha Mixture BP. Mitte 100 ml. Master formula 100 ml Ammonium bicarbonate 200 mg 2 g Liquorice liquid extract 0. 5 ml Ipecacuanha tincture 0. 3 ml Concentrated camphor water 0. 1 ml Concentrated anise water 0. 05 ml 0. 5 ml Double strength chloroform water 5 ml 50 ml Water to 10 ml to 100 ml BA-FP-JU-C 10/27/2020

Example on solution preparations • • • Action and uses. Formulation notes. Method of preparation. Shelf life and storage. Advice and labeling. BA-FP-JU-C 10/27/2020

Rx 10 ml Sodium Bicarbonate Ear Drops BP. Master formula Sodium bicarbonate Glycerol Water 5 g 30 ml to 100 ml BA-FP-JU-C For 10 ml 500 mg 3 ml to 10 ml 10/27/2020

• 200 ml Diamorphine linctus Master formula for 200 ml Diamorphine hydrochloride Oxymel Glycerol Compound tartrazine solution Syrup 3 mg 1. 25 ml 120 mg 50 ml 0. 06 ml to 5 ml 2. 4 ml to 200 ml BA-FP-JU-C 10/27/2020
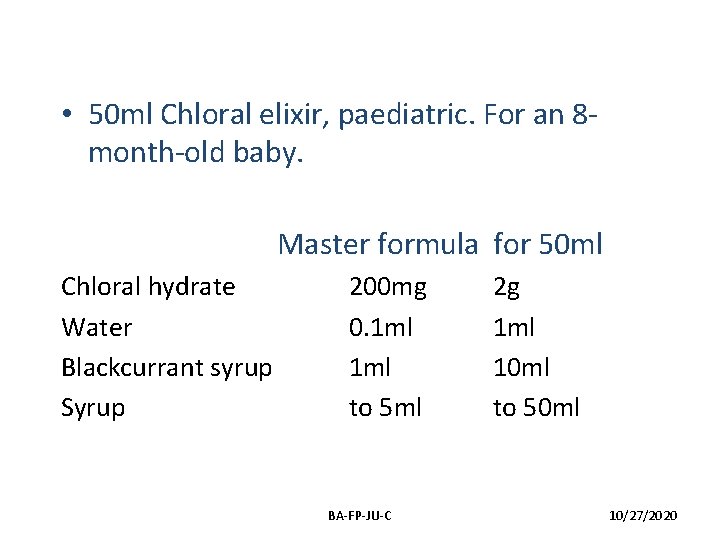
• 50 ml Chloral elixir, paediatric. For an 8 month-old baby. Master formula for 50 ml Chloral hydrate Water Blackcurrant syrup Syrup 200 mg 0. 1 ml to 5 ml BA-FP-JU-C 2 g 1 ml 10 ml to 50 ml 10/27/2020

Rx Compound Sodium Chloride Mouthwash BP. Mitte 500 ml Sodium chloride Sodium bicarbonate Concentrated peppermint emulsion Double strength chloroform water Water BA-FP-JU-C Master formula 1. 5 g 1 g 2. 5 ml 50 ml to 100 ml For 500 ml 7. 5 g 5 g 12. 5 ml 250 ml to 500 ml 10/27/2020
- Slides: 33