Solutions and Solubility Solubility can be described qualitatively

Solutions and Solubility
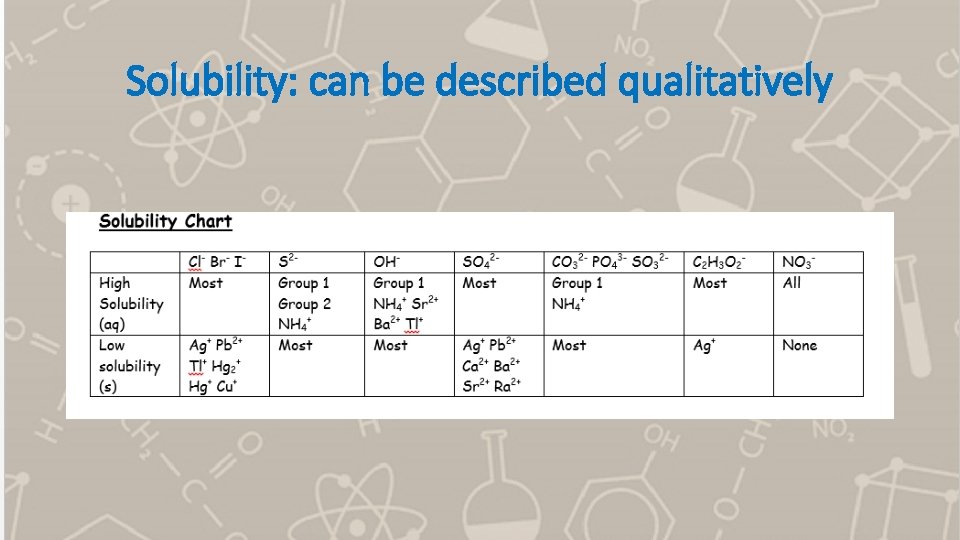
Solubility: can be described qualitatively

Solubility: can be described quantitatively • The maximum amount of solute that can be dissolved in a given volume of solution • g/100 m. L OR g/L OR mol/L (molar solubility) • Depends on the solute being dissolved AND the temperature
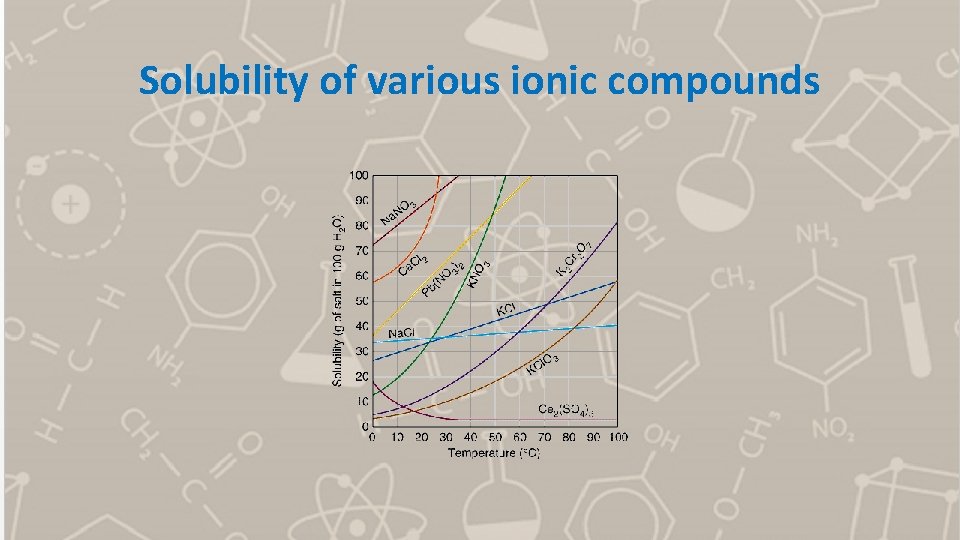
Solubility of various ionic compounds
What is the solubility of KCl. O 3 at 30 o. C a) in g/100 m. L? b) in moles/L?
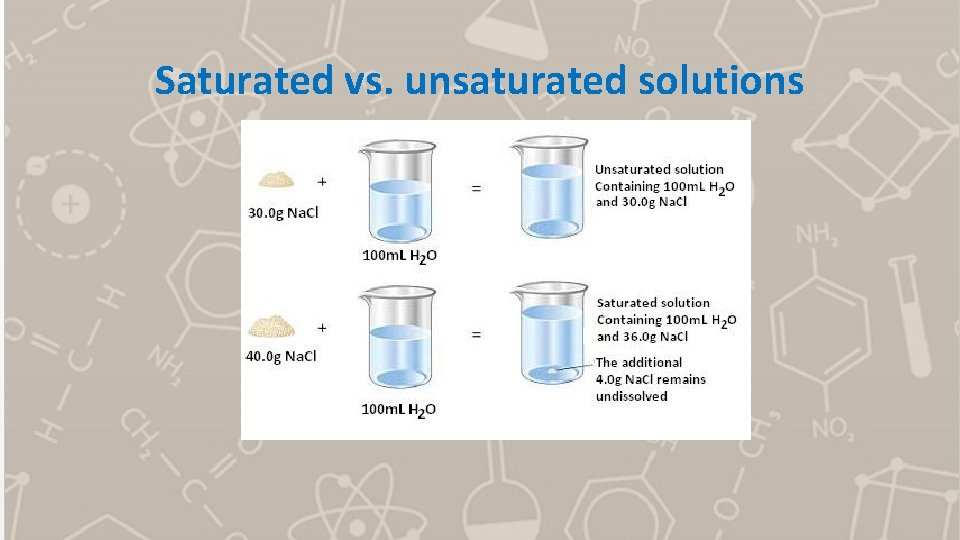
Saturated vs. unsaturated solutions
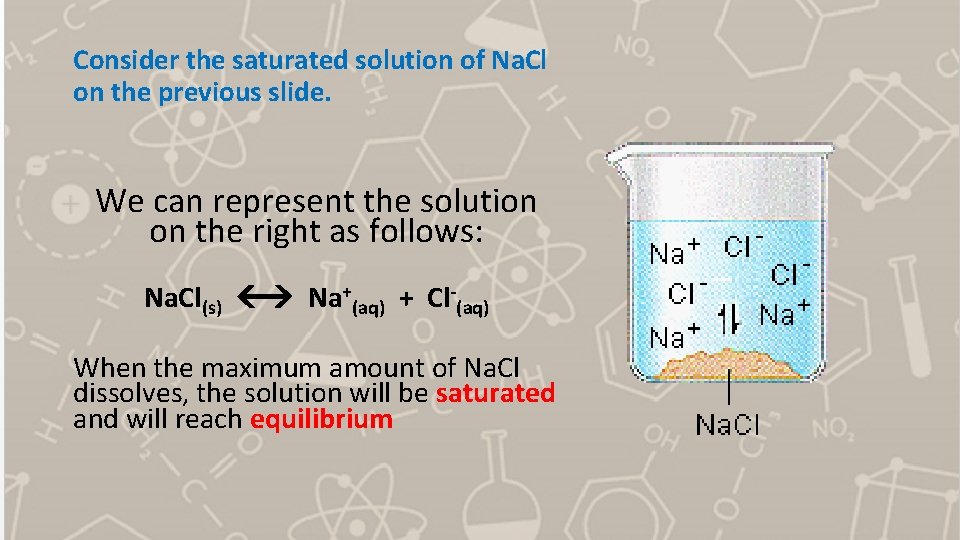
Consider the saturated solution of Na. Cl on the previous slide. We can represent the solution on the right as follows: Na. Cl(s) Na+(aq) + Cl-(aq) When the maximum amount of Na. Cl dissolves, the solution will be saturated and will reach equilibrium

Recall how Na. Cl dissolves in polar water

Equilibrium plays a more important role with insoluble or slightly soluble compounds Consider Ca. F 2 which is considered to be a slightly soluble compound. Write the equation that represents a saturated solution of Ca. F 2. Write the equilibrium expression. Notice that the solid is not included in the expression. We no longer call this Keq but Ksp (solubility product constant).

Write the equation for saturated solutions of the following compounds and look up their Kspvalue. Ba. SO 4 Pb. I 2 Sr 3(PO 4)2
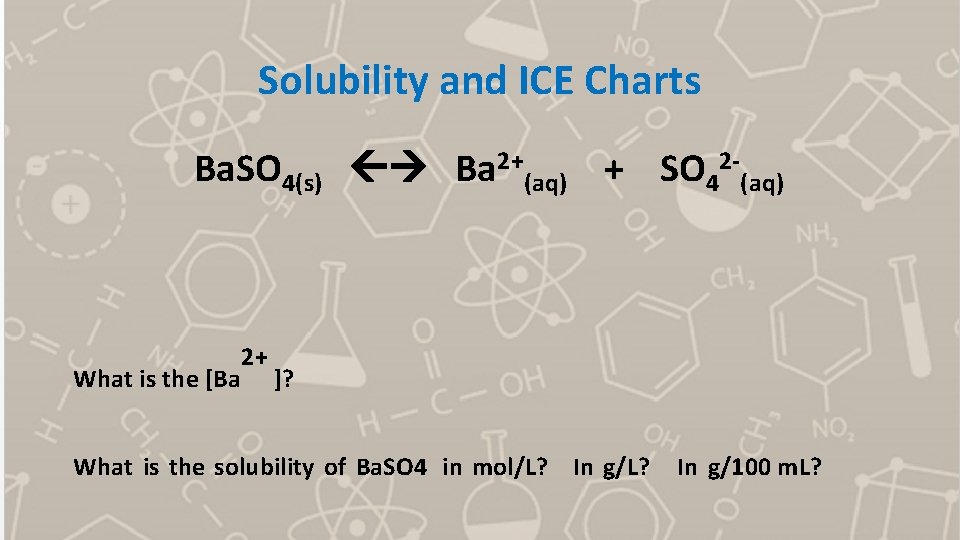
Solubility and ICE Charts Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) What is the [Ba 2+ ]? What is the solubility of Ba. SO 4 in mol/L? In g/L? In g/100 m. L?

Solubility and ICE Charts What is the molar solubility of Pb. I 2 ? Sr 3(PO 4)2 ?
- Slides: 12