Solution Problems A Vocabulary 1 Solution l homogeneous

Solution Problems

A. Vocabulary 1 Solution l homogeneous mixture l written Na. Cl(aq) which means Na. Cl dissolved in water 2 Solute l the substance that is being dissolved in a solution. l In chocolate milk, the chocolate is the solute. 3 Solvent l the substance that does the dissolving in a solution. l In Na. Cl(aq), water is the solvent. 4 Solubility l the amount of solute that dissolves in a given amount of solvent at a given temperature. l If a substance is soluble, it dissolves in water.

B. Using the Reference Tables 1. Table F tells if a substance dissolves (soluble) or does not dissolve (insoluble) in water Examples l l § § (NH 4)2 O § All ammonium compounds are soluble Ca. S § All sulfides are insoluble Ag. Br § All halides are soluble except with Ag+ Ba(OH)2 § All hydroxides are insoluble except Ba+2


2. Table G l Reference Table G shows how solubility changes with temperature l Each line represents a compound’s saturation point § § § On the line § Saturated—maximum solute that can dissolve Below the line § Unsaturated—not saturated; can still hold more solute Above the line “over saturated” § Supersaturated (dissolves anyway) § Precipitate (excess solute falls to the bottom of container)
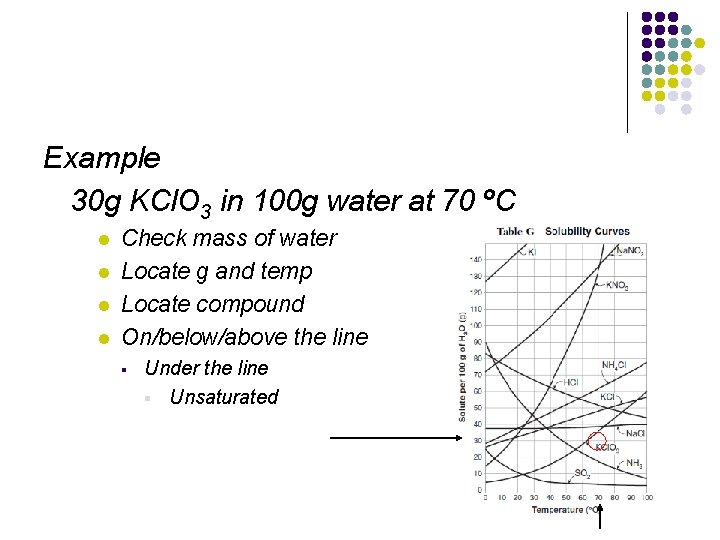
Example 30 g KCl. O 3 in 100 g water at 70 ºC l l Check mass of water Locate g and temp Locate compound On/below/above the line § Under the line § Unsaturated
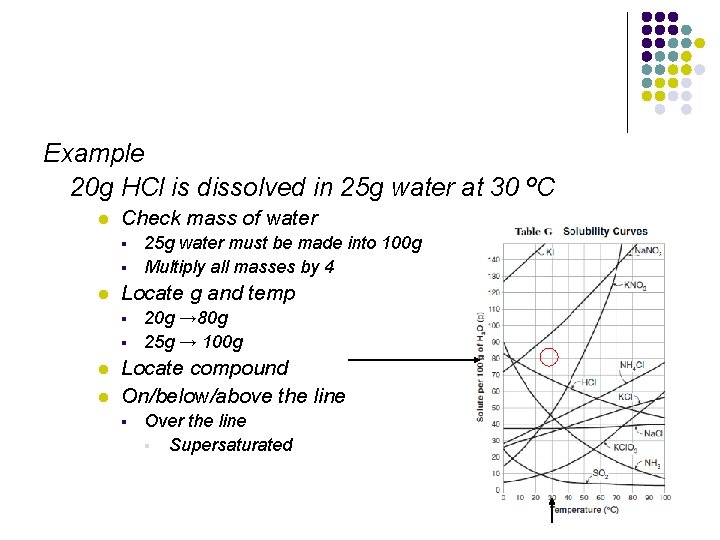
Example 20 g HCl is dissolved in 25 g water at 30 ºC l Check mass of water § § l Locate g and temp § § l l 25 g water must be made into 100 g Multiply all masses by 4 20 g → 80 g 25 g → 100 g Locate compound On/below/above the line § Over the line § Supersaturated
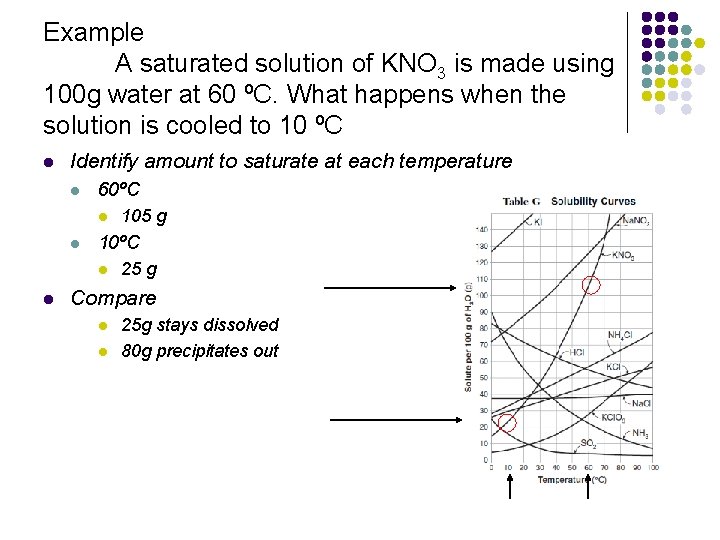
Example A saturated solution of KNO 3 is made using 100 g water at 60 ºC. What happens when the solution is cooled to 10 ºC l Identify amount to saturate at each temperature l 60ºC l l 105 g 25 g Compare l l 25 g stays dissolved 80 g precipitates out

C Expressions of Concentration represents the amount of solute dissolved in a solution 1. Vague Terms l l Strong vs weak Concentrated vs dilute 2. Measured values l l Molarity ppm (parts per million)

3. Molarity l Represents moles of solute per 1 liter of solution Ex. What is the molarity of a solution containing 1. 2 moles Na. Cl in a 150 m. L solution? l l M = moles ÷ liters 150 m. L = 0. 150 L § l l Move decimal 3 places M = 1. 2 ÷ 0. 150 L M = 8. 0 M § Means 8 moles Na. Cl in 1 liter of solution

4. Parts per Million (ppm) l ppm = grams solute x 1, 000 grams solution Ex. 0. 0043 g of O 2 can be dissolved in 100 g water at 20ºC. Express this in terms of parts per million. l l ppm = grams solute x 1, 000 grams solution ppm = 0. 0043 x 1, 000 = 43 ppm 100. 0043

Practice Problems 1. 2. 3. 4. 5. What is the molarity of a KF solution containing 0. 32 moles in 200 m. L of solution? What is the molarity of a Na. Cl solution containing 1. 02 moles in 125 m. L of solution? What is the molarity of a Na. OH solution containing 12 g Na. OH in 50 m. L of solution? What is the ppm of a CO 2 solution containing 0. 032 grams in 200 g of solution? What is the ppm of a NH 3 solution containing 0. 0012 grams in 350 g of solution?

Dilution Problems l l Addition of water to change the concentration Two types l l Dilute an amount of solution and determine the new molarity Determine how much of a concentrated solution must be used to make a specific molarity

Dilute an amount of solution and determine the new molarity ex. What is the concentration of a solution made if 35 m. Ls of 18 M H 2 SO 4 is diluted to 250 m. Ls? l Find the original mole value l Find new molarity 0. 035 L x 18 moles = 0. 63 moles H 2 SO 4 1 L 0. 63 moles = 2. 52 M H 2 SO 4 0. 250 L

Determine how much of a concentrated solution must be used to make a specific molarity ex. How would you make 100 m. L of a 3. 0 M HCl using 12 M HCl? l How many moles do you need? l Find the volume needed of the concentrated solution 0. 100 L x 3. 0 moles = 0. 30 moles HCl 1 L 0. 30 moles x 1 L = 0. 025 L HCl 12 moles Use 25 m. Ls HCl
- Slides: 15