solute molecule water molecule selectively permeable membrane osmosis

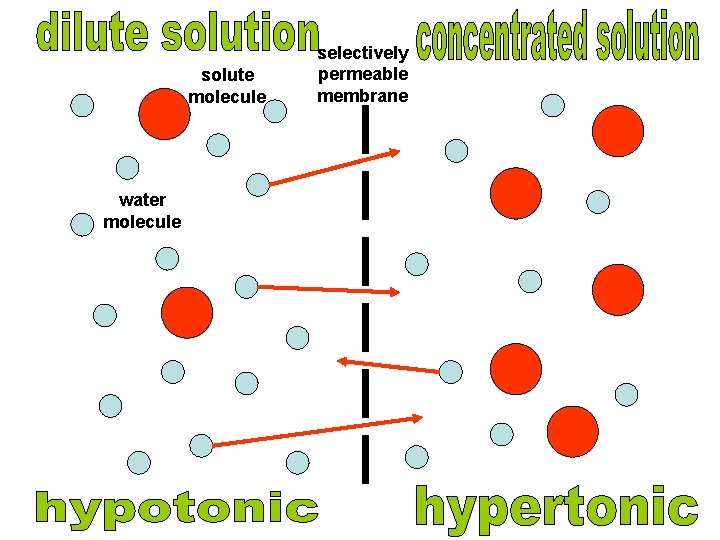
solute molecule water molecule selectively permeable membrane



osmosis animation



SOLUTE Water particles forming a hydration shell


Water potential / k. Pa -600 -1000 Size Negativity higher/ bigger lower/ smaller less negative more negative Ability to take in water less likely more likely
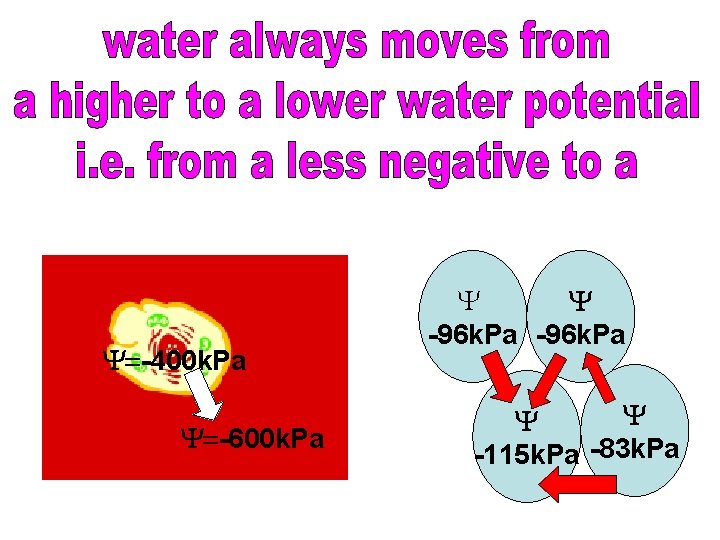
Y=-400 k. Pa Y=-600 k. Pa Y Y -96 k. Pa Y Y -115 k. Pa -83 k. Pa

This differs from water potential because water potential is affected by factors such as the available space in a cell. A turgid cell will still have the potential to take in water, as it is more concentrated than pure water, but because it is turgid it may be unable to, as there simply is no space available. That is why water potential is the tendency to take in water, it takes this into account.


This is the effect of pressure on the solution. A turgid palnt cell will have a high pressure on its cell wall due to the full contents of the cytoplasm and vacuole. A non-turgid cell will have less pressure. This therefore influences the ability of the cell to take in water by osmosis.
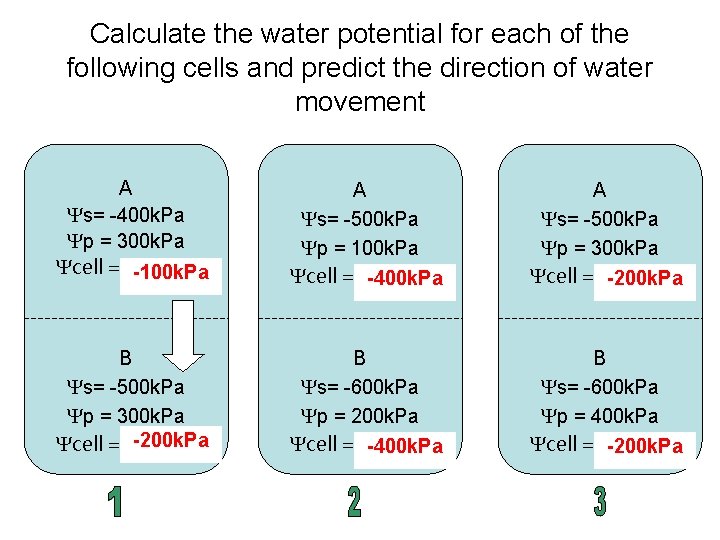
Calculate the water potential for each of the following cells and predict the direction of water movement A Ys= -400 k. Pa Yp = 300 k. Pa Ycell = ………. . -100 k. Pa A Ys= -500 k. Pa Yp = 100 k. Pa Ycell = ………. . -400 k. Pa A Ys= -500 k. Pa Yp = 300 k. Pa Ycell = ………. . -200 k. Pa B Ys= -500 k. Pa Yp = 300 k. Pa -200 k. Pa Ycell = ………. . B Ys= -600 k. Pa Yp = 200 k. Pa Ycell = ………. . -400 k. Pa B Ys= -600 k. Pa Yp = 400 k. Pa Ycell = ………. . -200 k. Pa
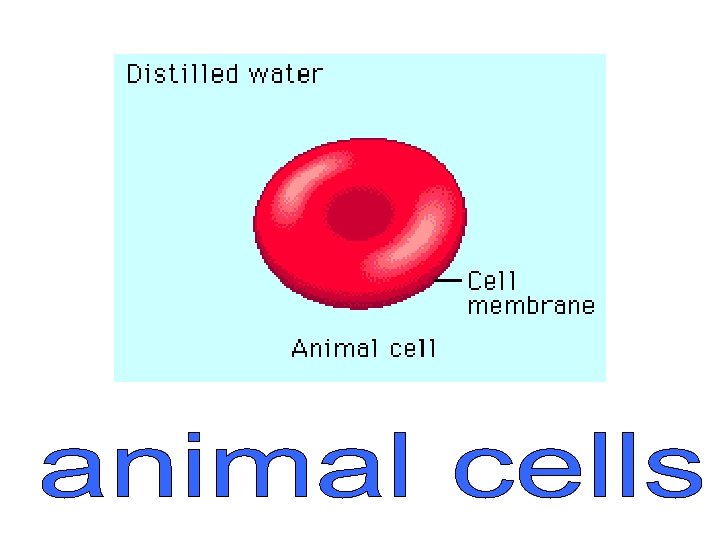

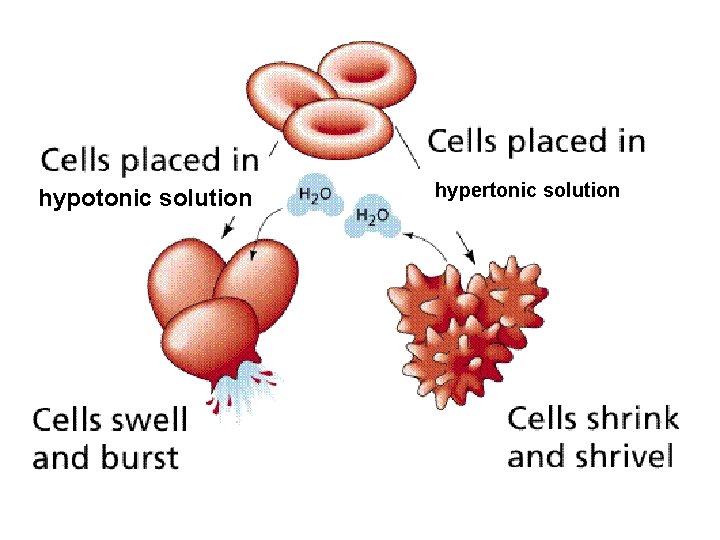
An animal cell in a hypotonic solution (more dilute) will gain water by osmosis because the water potential outside the cell will be higher (less negative) than inside. As the cytoplasm increases in volume it pushes against the cell membrane causing the pressure to increase. Eventually the cell membrane will rupture, a process called lysis or haemolysis.
hypotonic solution hypertonic solution
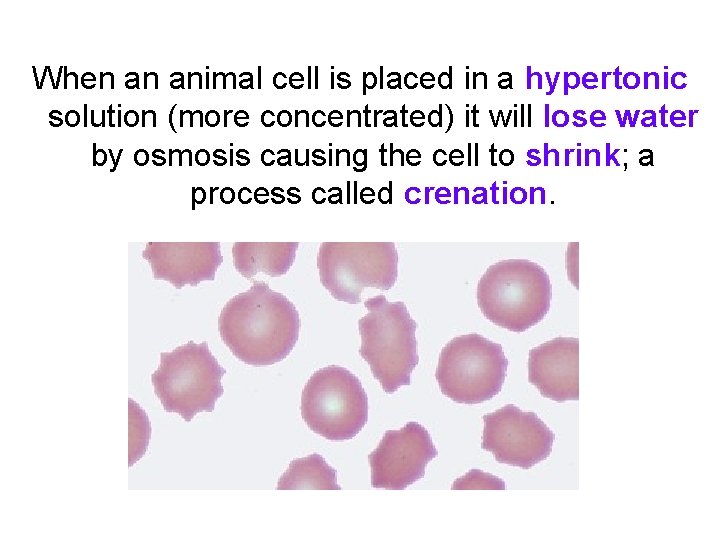
When an animal cell is placed in a hypertonic solution (more concentrated) it will lose water by osmosis causing the cell to shrink; a process called crenation.
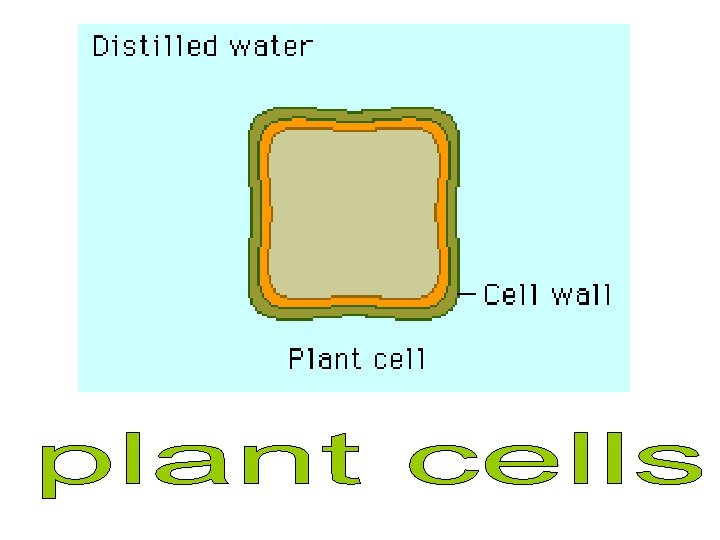

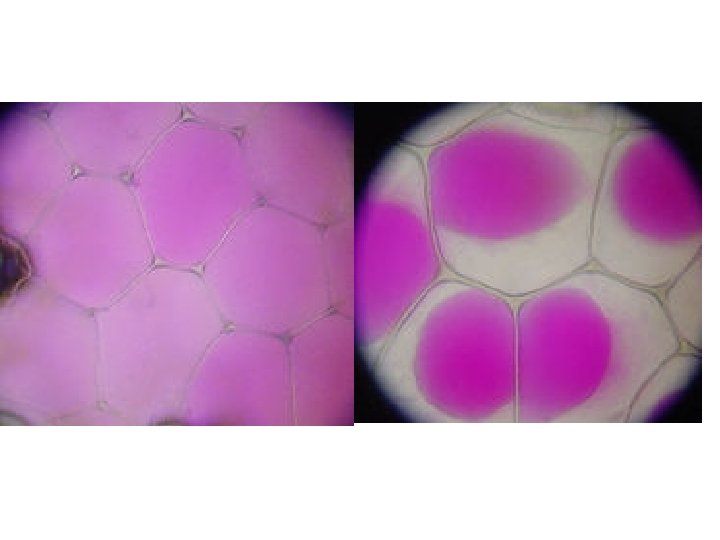
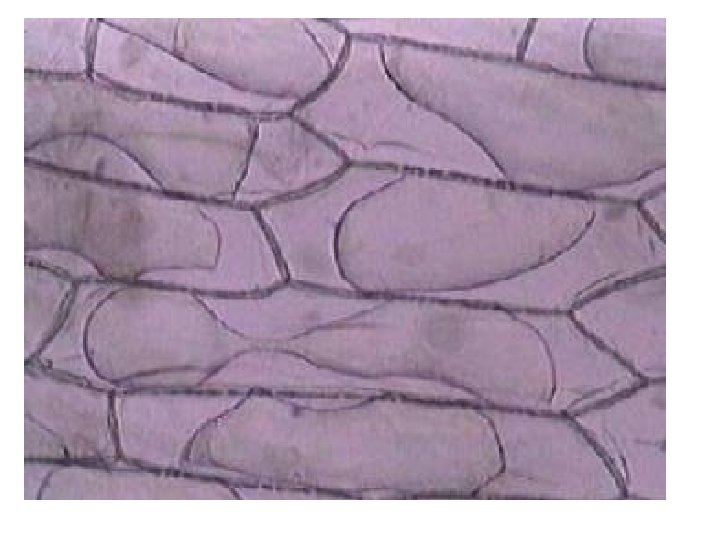
Plant cells in hypertonic solutions will lose water from the cytoplasm and vacuole by osmosis as the solution outside will have a lower water potential than inside. As the protoplasm shrinks the pressure inside the cell decreases and the membrane starts to move away from the cell wall and causes the pressure potential to decrease. When the membrane only just touches the cell wall the pressure potential is zero. This is called incipient plasmolysis. Tissue containing cells which have lost water in this way are said to be flaccid and cause a plant to wilt.

As water continues to be lost, the membrane tears away from the cell wall, only remaining attached at the plasmodesmata. The cells are described as plasmolysed and the cells are killed.


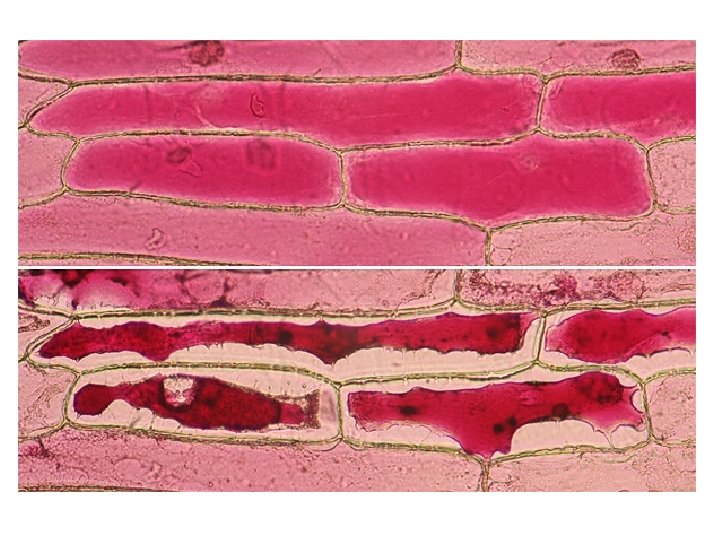
Plant cells in hypotonic solutions will increase in volume as water moves into the cytoplasm and vacuole by osmosis. This pushes the cell membrane against the cell wall. Pressure builds up but the strong cell wall pushes back onto the cell contents, preventing it from bursting. The cell is firm and described as turgid. Turgidity is important in plant support.
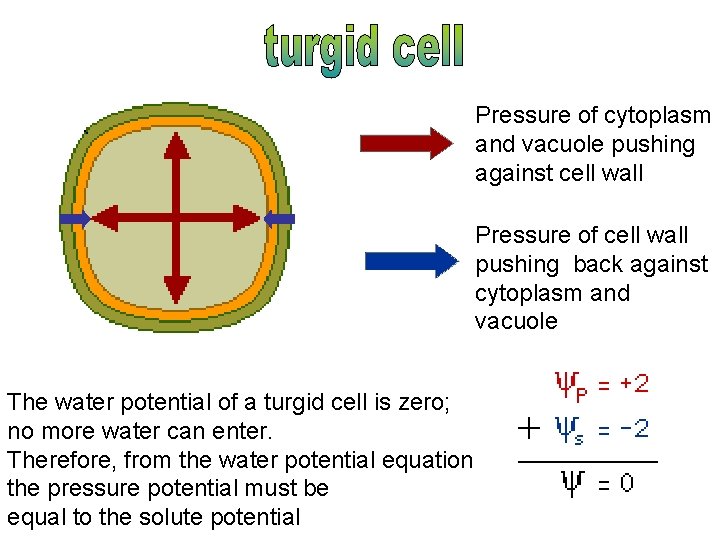
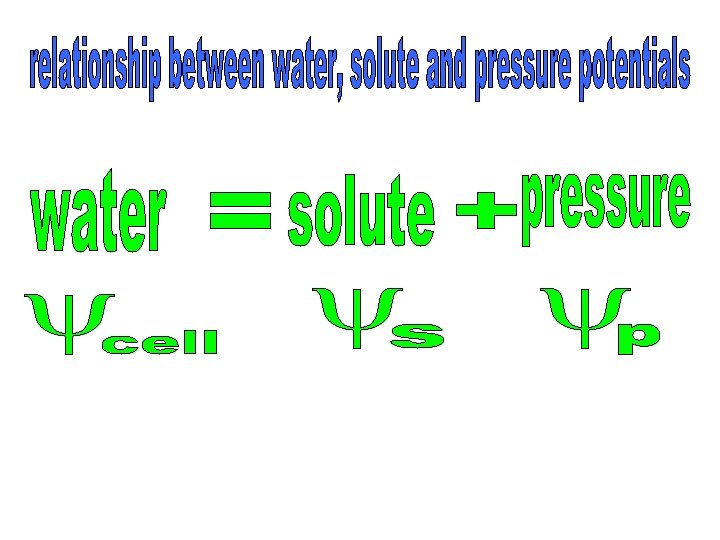
Pressure of cytoplasm and vacuole pushing against cell wall Pressure of cell wall pushing back against cytoplasm and vacuole The water potential of a turgid cell is zero; no more water can enter. Therefore, from the water potential equation the pressure potential must be equal to the solute potential

The pressure potential at incipient plasmolysis is zero; the wall no longer pushes back on the cytoplasm and vacuole. Therefore, from the water potential equation the solute potential must be equal to the water potential.
- Slides: 30