Solubility vs Temperature 180 160 O 3 KI
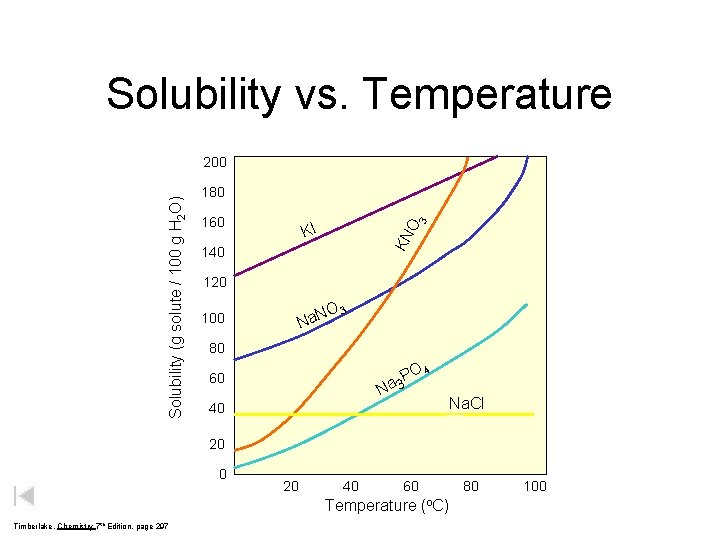
Solubility vs. Temperature 180 160 O 3 KI KN Solubility (g solute / 100 g H 2 O) 200 140 120 O Na. N 100 3 80 O 4 P a 3 60 N Na. Cl 40 20 40 60 Temperature (o. C) Timberlake, Chemistry 7 th Edition, page 297 80 100
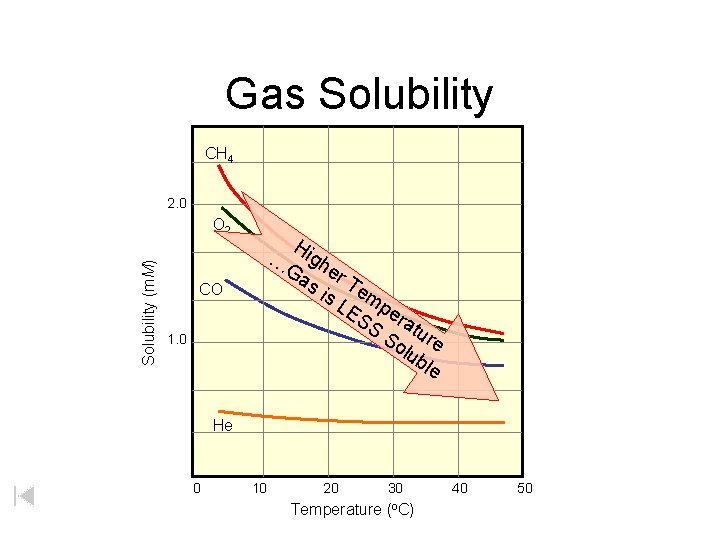
Gas Solubility CH 4 2. 0 Solubility (m. M) O 2 Hi g Ga her s i Te s L mp ES er S atu So re lub le … CO 1. 0 He 0 10 20 30 Temperature (o. C) 40 50
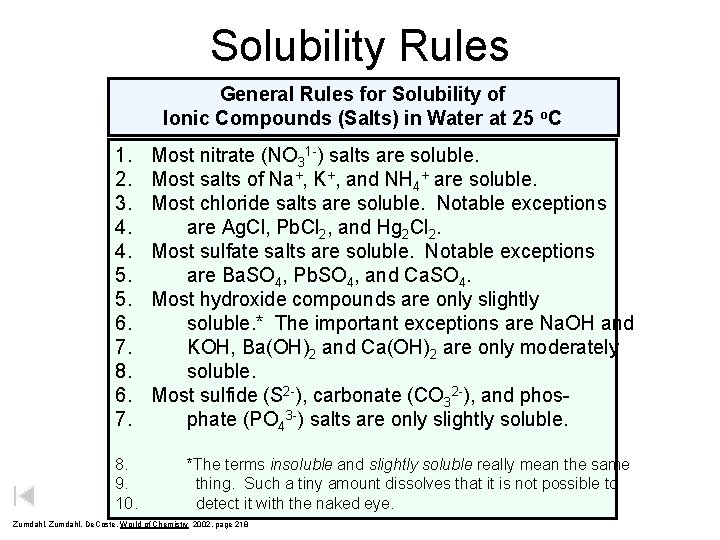
Solubility Rules General Rules for Solubility of Ionic Compounds (Salts) in Water at 25 o. C 1. 2. 3. 4. 4. 5. 5. 6. 7. 8. 9. 10. Most nitrate (NO 31 -) salts are soluble. Most salts of Na+, K+, and NH 4+ are soluble. Most chloride salts are soluble. Notable exceptions are Ag. Cl, Pb. Cl 2, and Hg 2 Cl 2. Most sulfate salts are soluble. Notable exceptions are Ba. SO 4, Pb. SO 4, and Ca. SO 4. Most hydroxide compounds are only slightly soluble. * The important exceptions are Na. OH and KOH, Ba(OH)2 and Ca(OH)2 are only moderately soluble. Most sulfide (S 2 -), carbonate (CO 32 -), and phosphate (PO 43 -) salts are only slightly soluble. *The terms insoluble and slightly soluble really mean the same thing. Such a tiny amount dissolves that it is not possible to detect it with the naked eye. Zumdahl, De. Coste, World of Chemistry 2002, page 218

Solubility of Common Compounds Soluble compounds NO 3 - salts Na+, K+, NH 4+ salts Cl-, Br-, I- salts Except for Ag+, Hg 22+, Pb 2+ those containing SO 42 - salts Except for 2+, Pb 2+, Ca 2+ Ba those containing Insoluble compounds S 2 -, CO 32 -, PO 43 - salts OH 1 - salts Zumdahl, De. Coste, World of Chemistry 2002, page 218 Except for those containing Na+, K+, Ca 2+
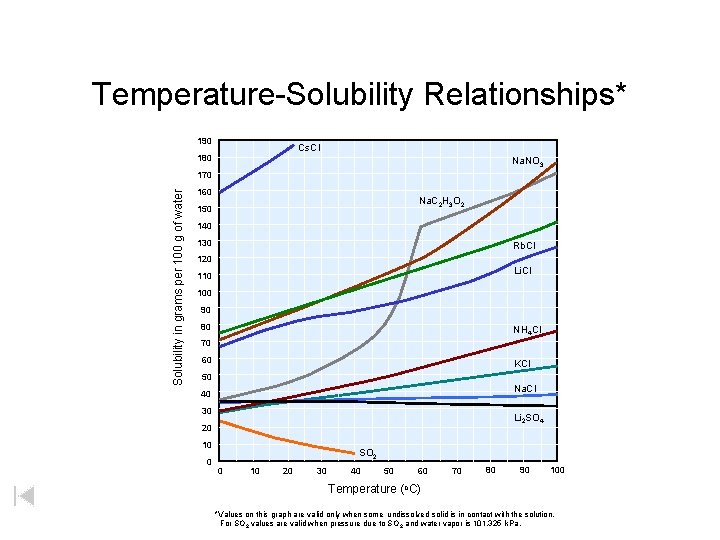
Temperature-Solubility Relationships* 190 Cs. Cl 180 Na. NO 3 Solubility in grams per 100 g of water 170 160 Na. C 2 H 3 O 2 150 140 130 Rb. Cl 120 Li. Cl 110 100 90 80 NH 4 Cl 70 60 KCl 50 Na. Cl 40 30 Li 2 SO 4 20 10 0 SO 2 0 10 20 30 40 50 60 70 80 90 100 Temperature (o. C) *Values on this graph are valid only when some undissolved solid is in contact with the solution. For SO 2 values are valid when pressure due to SO 2 and water vapor is 101. 325 k. Pa.
- Slides: 5