Solubility Solubility of Solids l Solubility Maximum amount

Solubility

Solubility of Solids l Solubility – – Maximum amount of solid that is in a quantity of water (usually 100 g) at a certain temperature. Determined by l l l The size of the solute crystal Time and amount of stirring Temperature

Solubility of Solids l Affected by the solute-solute and solutesolvent attraction – – l Solute = the substance being dissolved Solvent = the substance that does the dissolving LIKE DISSOLVES LIKE

Polarity l Polar molecules are attracted to other polar molecules and to electrically charged particles. l Non-polar substance are attracted to other nonpolar substances l Polar and non-polar DO NOT dissolve in each other

Polarity l Water is considered a polar molecule. – – Polar Molecule: a molecule that has an uneven distribution of charge. There is a positive region and a negative region.
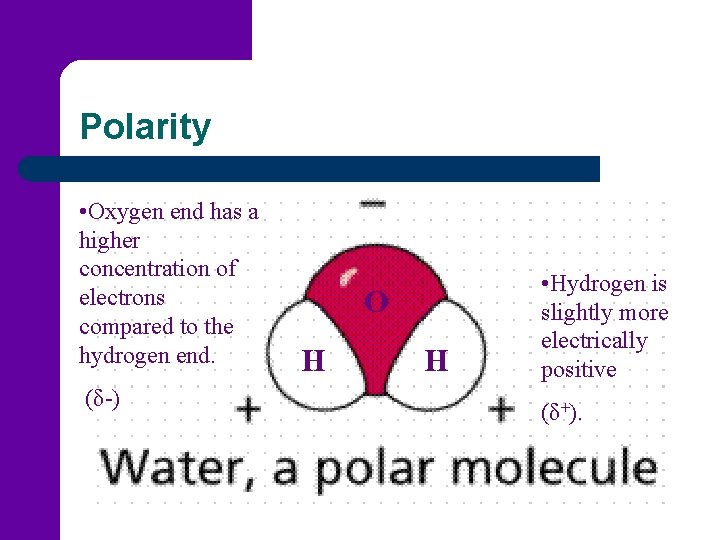
Polarity • Oxygen end has a higher concentration of electrons compared to the hydrogen end. (δ-) O H H • Hydrogen is slightly more electrically positive (δ+).

Polarity l Ionic Compounds – l are crystalline or crystal like Crystals are held together by the unlike charges (+ and –) that attract.
Polarity

Dissolving Salt l Dissolving Salt

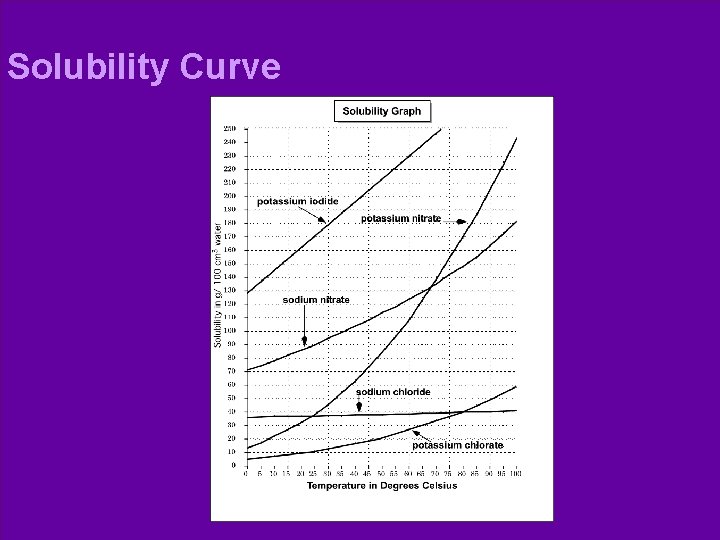
Solubility Curve l Solubility: – Units: grams 100 g H 2 O or grams. 100 m. L H 2 O

Solubility Curve l Characteristic of substance: – l Each substance has a different maximum amount that will dissolve. Solubility Curve – Shows a solution in which the solvent contains as much solute as possible at a certain temperature.
Solubility Curve
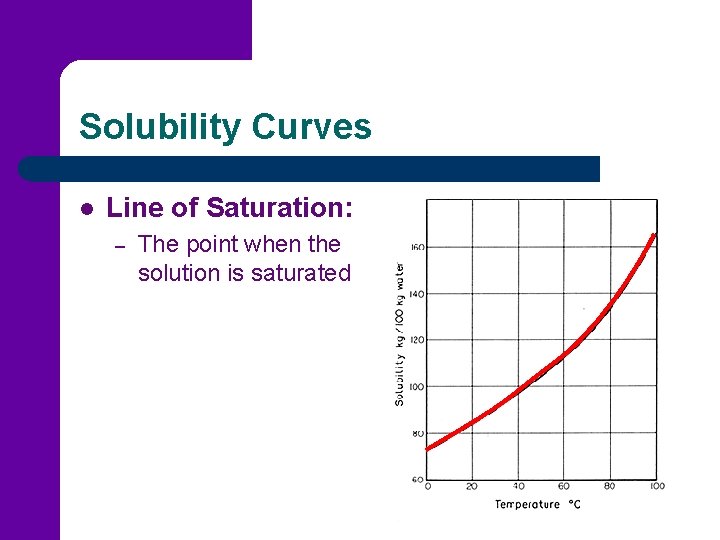
Solubility Curves l Line of Saturation: – The point when the solution is saturated
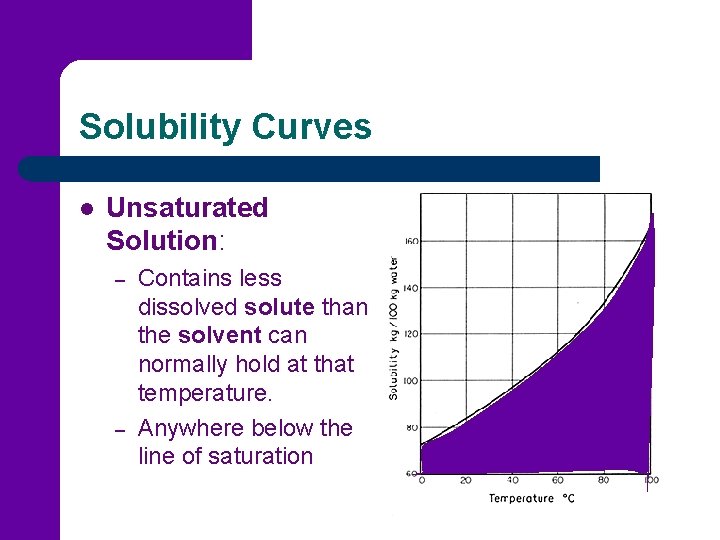
Solubility Curves l Unsaturated Solution: – – Contains less dissolved solute than the solvent can normally hold at that temperature. Anywhere below the line of saturation
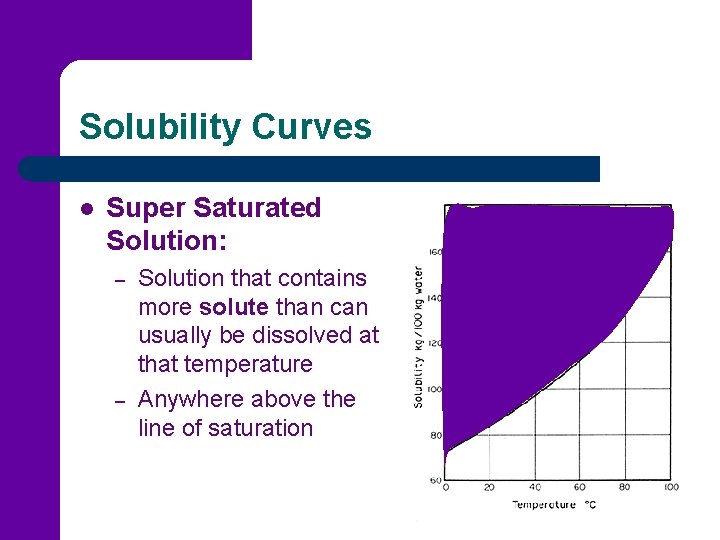
Solubility Curves l Super Saturated Solution: – – Solution that contains more solute than can usually be dissolved at that temperature Anywhere above the line of saturation
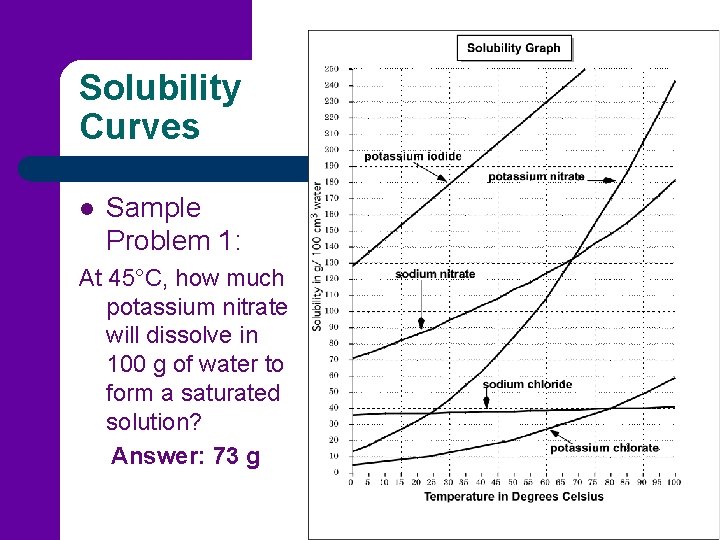
Solubility Curves l Sample Problem 1: At 45°C, how much potassium nitrate will dissolve in 100 g of water to form a saturated solution? Answer: 73 g
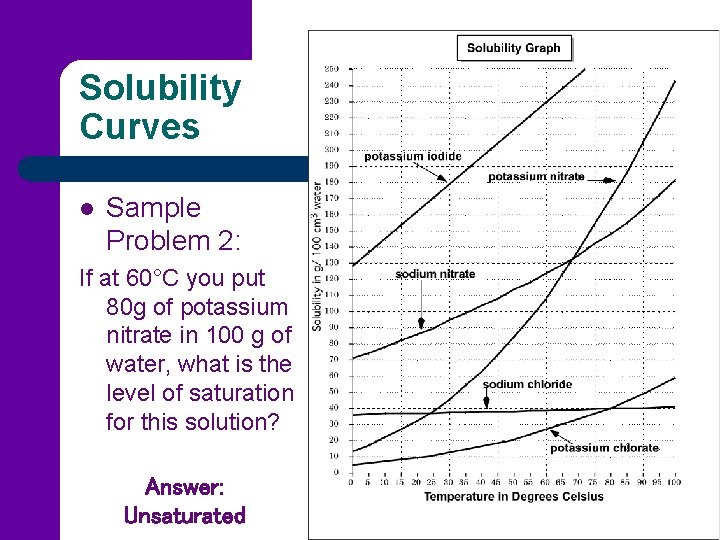
Solubility Curves l Sample Problem 2: If at 60°C you put 80 g of potassium nitrate in 100 g of water, what is the level of saturation for this solution? Answer: Unsaturated
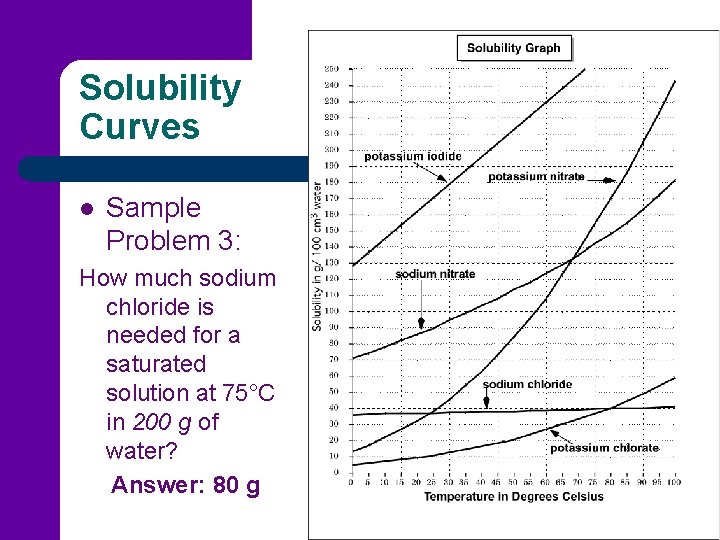
Solubility Curves l Sample Problem 3: How much sodium chloride is needed for a saturated solution at 75°C in 200 g of water? Answer: 80 g
- Slides: 18